ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with the unaudited condensed consolidated financial statements and notes thereto included elsewhere in this Quarterly Report on Form 10-Q, or Form 10-Q, and the audited consolidated financial statements and notes thereto for our fiscal year ended September 30, 2025 included in our Annual Report on Form 10-K for that fiscal year, which is referred to as our 2025 Form 10-K. Please refer to our note regarding forward-looking statements on page 2 of this Form 10-Q, which is incorporated herein by this reference.
The Enanta name and logo are our trademarks. This Form 10-Q also includes trademarks, trade names and service marks of other persons. All other trademarks, trade names and service marks appearing in this Form 10-Q are the property of their respective owners.
Overview
We are a biotechnology company that uses our robust, chemistry-driven approach and drug discovery capabilities to discover and develop small molecule drugs for virology and immunology indications.
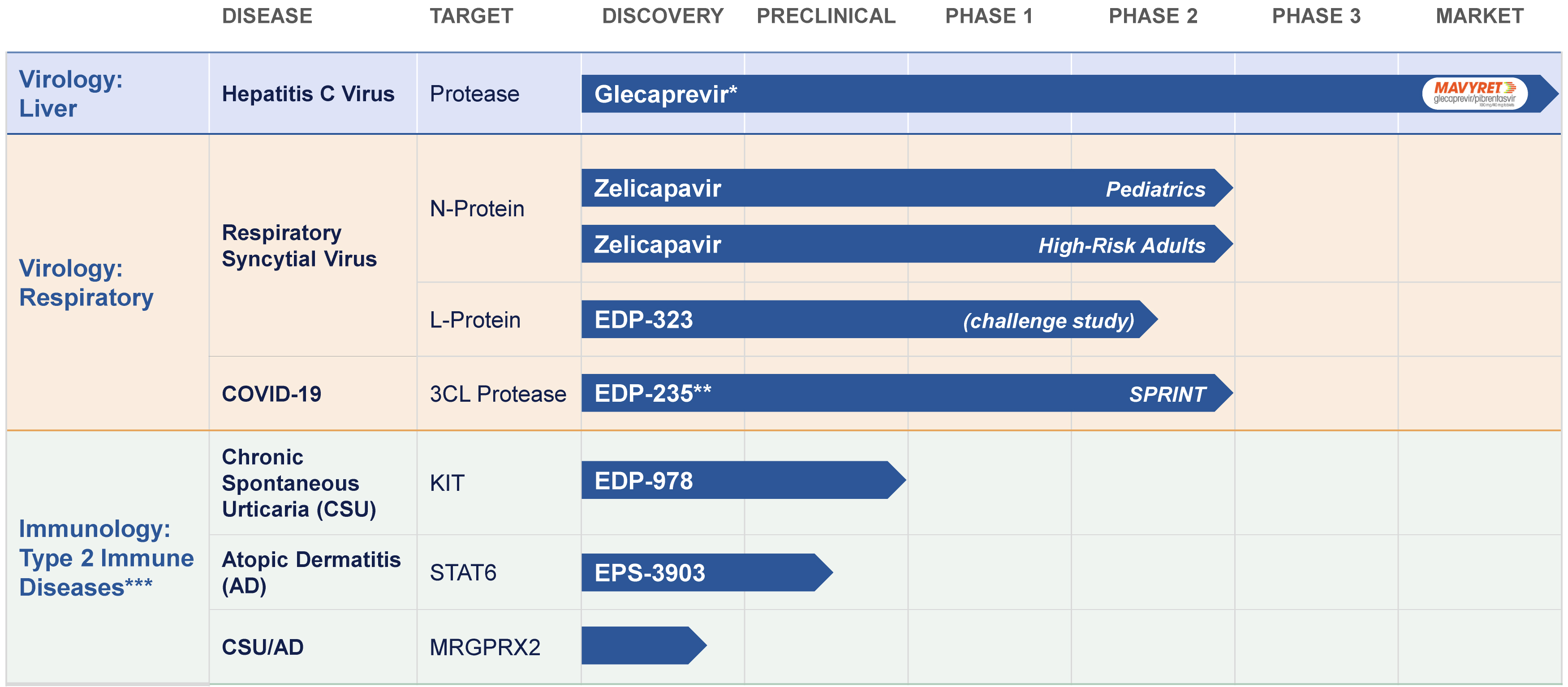
Virology:
We discovered glecaprevir, the second of two antiviral protease inhibitors developed through our collaboration with AbbVie for the treatment of acute or chronic infection with hepatitis C virus, or HCV. Glecaprevir is co-formulated as part of AbbVie’s leading brand of direct-acting antiviral, or DAA, combination treatment for HCV, which has been marketed under the tradenames MAVYRET® (U.S.) and MAVIRET® (ex-U.S.) (glecaprevir/pibrentasvir) since 2017 for the treatment of chronic HCV. MAVYRET® was also approved as the first and only treatment for acute HCV infection in June 2025.
Our active development programs in virology are focused on respiratory syncytial virus, or RSV, the most common cause of bronchiolitis and pneumonia and a leading cause of U.S. hospitalization in young children and a significant cause of respiratory illness in older adults. Populations at high risk for severe RSV infection include infants and young children, adults older than 65 years of age, and those with comorbidities such as chronic heart or lung disease. Recent CDC estimates suggest a significant RSV burden in the U.S., with up to 6.5 million outpatient visits, 350,000 hospitalizations and 23,000 deaths annually.
We also have clinical-stage programs in virology for SARS-CoV-2, the virus that causes COVID-19, and Hepatitis B virus, or HBV, the most prevalent chronic hepatitis.
Immunology:
In immunology, we are designing and developing highly potent and selective, oral small molecule inhibitors for the treatment of type 2 inflammatory disease by targeting key mechanisms of the immune response. An overactive response is a primary driver of a number of inflammatory diseases for which there is an enduring unmet need including atopic dermatitis, or AD, urticarias, asthma, prurigo nodularis, or PN, chronic rhinosinusitis with nasal polyps, or CRSwNP, as well as some forms of chronic obstructive pulmonary disease, or COPD, and other conditions. Based on industry reports, by 2030 the market is projected to be approximately $5 billion for urticaria, $30 billion for AD and $35 billion for the combined market of asthma, COPD, CRSwNP, and PN.
Our initial immunology targets involve the following mechanisms of immune response:
•KIT, a receptor tyrosine kinase, critical for regulating mast cell survival and activation, including release of potent inflammatory mediators such as histamine, which is a primary driver of inflammation and implicated in multiple allergic diseases;
•STAT6, a transcription factor uniquely responsible for interleukin-4, or IL-4, and interleukin-13, or IL-13, cell signaling, which drives a type 2 dominant phenotype and downstream inflammation; and
•MRGPRX2, a non-canonical G-Protein-Coupled-Receptor (GPCR) expressed predominantly on mast cells, which upon activation triggers degranulation and release of inflammation mediating components, leading to an inflammatory response that is a driver in multiple allergic diseases.
These mechanisms are implicated, along with others, in several diseases, and it is not uncommon for an efficacious treatment for one disease to be tested and approved for other immunology indications. In addition, these mechanisms are orthogonal approaches that may provide additive or complementary benefit if used in combination. We currently plan to focus our initial immunology drug development, proof-of-concept efforts on the following disease indications:
•Urticaria, including chronic spontaneous urticaria, or CSU, a severely debilitating, chronic inflammatory skin disease manifested by hives, angioedema, which is swelling of soft tissues, or both, but with no identified triggers, which has an estimated global prevalence of between 0.5% – 1% of the population, resulting in approximately 1.75–3.5 million people