UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
☒
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended December 31, 2025
OR
☐
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period
from to
Commission file number: 001-40492
| | Femasys Inc. | |
| | (Exact Name of Registrant as Specified in its Charter) | |
| Delaware | | 11-3713499 |
| (State or other jurisdiction of incorporation or organization) | | (IRS Employer Identification No.) |
| 3950 Johns Creek Court, Suite 100 Suwanee, GA 30024 | | (770) 500-3910 |
| (Address of principal executive offices, including zip code) | | (Registrant’s telephone number, including area code) |
Securities registered pursuant to Section 12(b) of the Act:
| | | | | |
| Title of each class | | Trading Symbol(s) | | Name of each exchange on which
registered |
| Common stock, par value $0.001 | | FEMY | | NASDAQ Capital Market |
Securities Registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Exchange Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | |
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ |
| | | Emerging growth company | ☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of June 30, 2025 was $29,865,186.
As of March 30, 2026, there were 60,390,686 shares of the registrant’s $0.001 par value common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement for the Registrant’s 2026 Annual Meeting of Stockholders are incorporated by reference in Part III of this annual report on Form 10-K. The definitive proxy statement will be filed with the U.S. Securities and Exchange Commission within 120 days after the end of the fiscal year covered by this annual report on Form 10-K.
FEMASYS INC.
FORM 10-K
| | | |
| |
TABLE
OF CONTENTS
|
|
| |
|
|
| |
|
1
|
|
|
|
5
|
|
Item 1.
|
|
5
|
|
Item 1A.
|
|
30 |
|
Item 1B.
|
|
78
|
|
Item 2.
|
|
78
|
|
Item 3.
|
|
79
|
|
Item 4.
|
|
79
|
|
|
|
80 |
|
Item 5.
|
|
80 |
|
Item 6.
|
|
80 |
|
Item 7.
|
|
81 |
|
Item 7A.
|
|
89
|
|
Item 8.
|
|
90 |
|
Item 9.
|
|
126 |
|
Item 9A.
|
|
126 |
|
Item 9B.
|
|
127 |
|
Item 9C.
|
|
127 |
|
|
|
127 |
|
Item 10.
|
|
127 |
|
Item 11.
|
|
127 |
|
Item 12.
|
|
127 |
|
Item 13.
|
|
127 |
|
Item 14.
|
|
127 |
|
|
|
128 |
|
Item 15.
|
|
128 |
|
Item 16.
|
|
130 |
| |
|
132 |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts contained in this Annual Report on Form 10-K are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. Forward-looking statements include, but are not limited to, statements concerning:
• our ability to obtain additional financing to fund commercialization of our products and fund our operations;
• our ability to obtain additional financing to fund the U.S. clinical development of our U.S. product candidate FemBloc® permanent birth control;
• our ability to obtain U.S. Food and Drug Administration (FDA) approval for our U.S. product candidate, FemBloc, for permanent birth control;
• our ability to successfully grow sales of FemaSeed® intratubal insemination in the U.S.;
• our ability to successfully grow sales internationally;
• estimates regarding the total addressable market for our products and U.S. product candidate;
• competitive companies and technologies in our industry;
• our business model and strategic plans for our products, U.S. product candidate, technologies and business, including our implementation thereof;
• commercial success and market acceptance of our products and U.S. product candidate;
• our ability to achieve and maintain adequate levels of coverage or reimbursement for FemBloc or any future product candidates, and our products we seek to commercialize;
• our ability to accurately forecast customer demand for our products and U.S. product candidate, and manage our inventory;
• our ability to build, manage and maintain our direct sales and marketing organization, and to market and sell our FemaSeed intratubal insemination product, FemBloc permanent birth control system, and women-specific medical product solutions in markets in and outside of the United States;
• our ability to establish, maintain, grow or increase sales and revenues;
• our expectations about market trends;
• our ability to continue operating as a going concern;
• the ability of our clinical trials to demonstrate safety and effectiveness of our U.S. product candidate, FemBloc, and other positive results;
• our ability to enroll subjects in the clinical trial for our U.S. product candidate, FemBloc, in order to advance the development thereof on a timely basis;
• our ability to manufacture our products and U.S. product candidate, if approved, in compliance with applicable laws, regulations and requirements and to oversee third-party suppliers, service providers and vendors in the performance of any contracted activities in accordance with applicable laws, regulations and requirements;
• our ability to hire and retain our senior management and other highly qualified personnel;
• FDA or other U.S. or foreign regulatory actions affecting us or the healthcare industry generally, including healthcare reform measures in the United States and international markets;
• the timing or likelihood of regulatory filings and approvals or clearances;
• our ability to establish and maintain intellectual property protection for our products and U.S. product candidate and our ability to avoid claims of infringement; and
• the volatility of the trading price of our common stock.
The forward-looking statements in this Annual Report on Form 10-K are only predictions and are based largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this Annual Report on Form 10-K and are subject to a number of known and unknown risks, uncertainties and assumptions, including those described under the sections in this Annual Report on Form 10-K entitled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in this Annual Report on Form 10-K. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. You should read this Annual Report on Form 10-K and the documents that we reference in this Annual Report on Form 10-K and have filed with the U.S. Securities and Exchange Commission (SEC) as exhibits hereto completely and with the understanding that our actual future results may be materially different from any future results expressed or implied by these forward-looking statements. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
Risk Factor Summary
The following is a summary of the principal risk factors associated with an investment in our common stock.
Risks Related to Our Financial Position and Need for Additional Capital
• We have incurred significant operating losses since inception, and we expect to incur operating losses in the future.
• We need substantial additional funding and may be unable to raise equity capital or debt financing when needed.
• There is substantial doubt about our ability to continue as a going concern.
• Our financial results may fluctuate significantly.
• Our ability to use our net operating losses and research and development credit carryforwards to offset future taxable income, if any, may be subject to certain limitations.
• We have received deficiency notices from Nasdaq relating to our non-compliance with Nasdaq's continued listing requirements and our common stock could become subject to delisting from Nasdaq if we fail to regain compliance.
Risks Related to Discovery and Development
• Enrollment and retention of subjects in clinical trials is an expensive and time-consuming process.
• Our current U.S. product candidate is in late-stage development.
• We are substantially dependent on the FDA’s permission to market our FemBloc system.
• The clinical development process required to obtain regulatory approvals is lengthy and expensive with uncertain outcomes.
• Interim, “topline,” and preliminary data from our clinical trials that we announce or publish from time to time may change as more data become available.
• Our products may fail to gain increased market acceptance.
• Our FemaSeed intratubal insemination solution may fail to gain market acceptance.
• If we are unable to achieve and maintain adequate levels of coverage or reimbursement for our FemBloc permanent birth control solution, our commercial success may be severely hindered.
• Third-party payors and healthcare practitioners who do not cover or use our permanent birth control solution or other women’s healthcare devices may require additional clinical data prior to adopting or maintaining coverage of our FemBloc system.
• The training required for healthcare practitioners to use our FemBloc permanent birth control solution could reduce the market acceptance of our U.S. product candidate.
• Some of our competitors have longer operating histories and more established products or greater resources than we do.
• Our long-term growth depends on our ability to enhance our solutions, expand our indications and develop and commercialize additional products.
• Our results of operations could be materially harmed if we are unable to accurately forecast customer demand and manage our inventory.
• We manufacture and assemble components for our products and U.S. product candidate, and a loss or degradation in performance of our manufacturing capabilities could have a material adverse effect on our business.
• We rely on a limited number of third-party suppliers for components for our products and U.S. product candidate.
• Performance issues, service interruptions or price increases by our shipping carriers could adversely affect our business.
• We have limited experience marketing and selling our women-specific medical product solutions.
• We plan to rely on our own direct sales force in North America to market our women-specific medical products.
• We plan to rely on distribution partners outside of North America to market our women-specific medical products.
• We face the risk of product liability claims that could be expensive.
• If the quality of our solutions does not meet the expectations of healthcare practitioners or patients, then our brand and reputation or our business could be adversely affected.
Risks Related to Managing Growth and Employee Matters
• We face risks related to health epidemics and outbreaks.
• Failure of a key information technology system, process or site could have an adverse effect on our business.
• Our facilities could become damaged or inoperable.
• Our ability to maintain our competitive position depends on our ability to attract and retain highly qualified talent.
• We will need to grow the size of our organization, and we may experience difficulties in managing this growth.
Risks Related to Government Regulation
• Our products and operations are subject to extensive government regulations.
• We may not receive the necessary regulatory approvals, classifications, or clearances to grow our business.
• Modifications to our U.S. product candidate if FDA approved may require us to obtain new Premarket Approval (PMA) approval or approvals of a PMA supplement.
• Failure to comply with post-marketing regulatory requirements could subject us to enforcement actions.
• Our products must be manufactured in accordance with federal and state regulations.
• If treatment guidelines for permanent birth control or other women’s healthcare treatments change or the standard of care evolves, we may need to redesign and seek new marketing authorization from the FDA for one or more of our products.
• There may be misuse or off-label use of our products in the marketplace.
• Our products may cause or contribute to adverse medical events or be subject to failures or malfunctions.
• If we do not obtain and maintain international regulatory registrations or approvals for our products, we will be unable to market and sell our products outside of the United States.
• Legislative or regulatory reforms in the United States or the European Union may make it more difficult and costly for us to obtain regulatory clearances or approvals for our products.
• Our business involves the use of hazardous materials.
Risks Related to Intellectual Property Matters
• If we are unable to adequately protect our intellectual property rights, our competitive position could be harmed.
• Obtaining and maintaining patent protection depends on compliance with various governmental requirements.
• Litigation or other proceedings or third-party claims of intellectual property infringement could require us to spend significant time and money.
• We may be unable to enforce our intellectual property rights throughout the world.
• Third parties may assert that our employees or consultants have wrongfully used or disclosed confidential information or misappropriated trade secrets.
• Recent changes in U.S. patent laws could diminish the value of patents in general.
• Patent terms may be inadequate to protect our competitive position on our products.
Risks Related to Our Common Stock
• Our directors, officers and principal stockholders have significant voting power.
• We incur significant costs as a result of being a public company.
• We are obligated to develop and maintain proper and effective internal controls over financial reporting.
• Our disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
Business Overview
We are a leading biomedical innovator, addressing significant unmet needs in women’s health worldwide, with a broad patent-protected portfolio of disruptive, accessible, in-office therapeutic and diagnostic products. The Company is a U.S. manufacturer that has received global regulatory approvals for its product portfolio, which is currently being commercialized in the U.S. and key international markets. FemaSeed® Intratubal Insemination, a groundbreaking first-step infertility treatment delivering sperm directly to the site of conception, is U.S. FDA-cleared and approved in Europe, United Kingdom (UK), Canada, Israel, Australia and New Zealand. A peer-reviewed publication of positive data from its pivotal clinical trial of FemaSeed demonstrated effectiveness and safety with high satisfaction from both patients and practitioners. FemSperm®, which includes setup, preparation and analysis kits designed to expand our infertility portfolio and, for the first time, enables gynecologists to perform in-office sperm preparation and analysis for use with FemaSeed. FemVue®, a companion diagnostic for fallopian tube assessment via ultrasound, is U.S. FDA-cleared and approved in Europe, UK, Canada, Japan, Israel, Australia and New Zealand. FemVue Controlled is US FDA-cleared diagnostic device and is the next-generation design integrating features of FemVue and FemChec® technologies into a single platform, enabling multiple clinical uses within one solution, including confirmation of tubal patency prior to use with FemaSeed. FemCerv®, an endocervical tissue sampler for cervical cancer diagnosis, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel and New Zealand.
FemBloc® permanent birth control is a revolutionary first-in-class non-surgical solution, which involves minimally-invasive placement of a patented delivery system for precise delivery of our proprietary synthetic tissue adhesive (blended polymer) into both fallopian tubes simultaneously. Over time, the blended polymer fully degrades and produces nonfunctional scar tissue to permanently block the fallopian tubes in a safe and natural approach. This is in stark contrast to centuries-old surgical sterilization with reported risks that include infection, minor or major bleeding, injury to nearby organs, anesthesia-related events, and even death. Along with the various surgical risks, some patients may not qualify as good surgical candidates due to obesity or medical comorbidities. The FemBloc non-surgical approach has the potential to offer a safe and effective, more accessible in-office alternative with fewer risks, contraindications, and substantially lower cost than the surgical alternative. A peer-reviewed publication of positive data from its initial clinical trials of FemBloc has demonstrated compelling effectiveness and five-year safety with high satisfaction from both patients and practitioners. In March 2025, we announced Conformité Européene (CE) mark certification under European Union (“EU”) Medical Device Regulation (“MDR”) as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, for FemBloc. In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. The Company received FDA approval in November 2025 of its investigational device exemption (“IDE”) supplement to move forward to the final phase of the pivotal clinical trial (clinicaltrials.gov: NCT05977751) for U.S. approval. In March 2026, we announced
the initiation of enrollment in this final phase. FemChec, a companion diagnostic product for FemBloc’s ultrasound-based confirmation test, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel, Australia and New Zealand. FemCath® for selective fallopian tube evaluation is U.S. FDA-cleared and approved in Europe, Canada and Israel for selective fallopian tube evaluation. The Company is a woman-founded and led company with an expansive, internally created intellectual property portfolio with approximately 228 issued patents globally, in-house chemistry, manufacturing, and controls (CMC) and device manufacturing capabilities and proven ability to develop products with commercialization efforts underway. Our suite of products and U.S. product candidate addresses what we believe are multi-billion dollar global market segments in which there has been little advancement for many years, helping women avoid pharmaceutical solutions, implants and surgery that can be expensive and expose women to harm.
The following table summarizes our current products and product candidate pipeline:
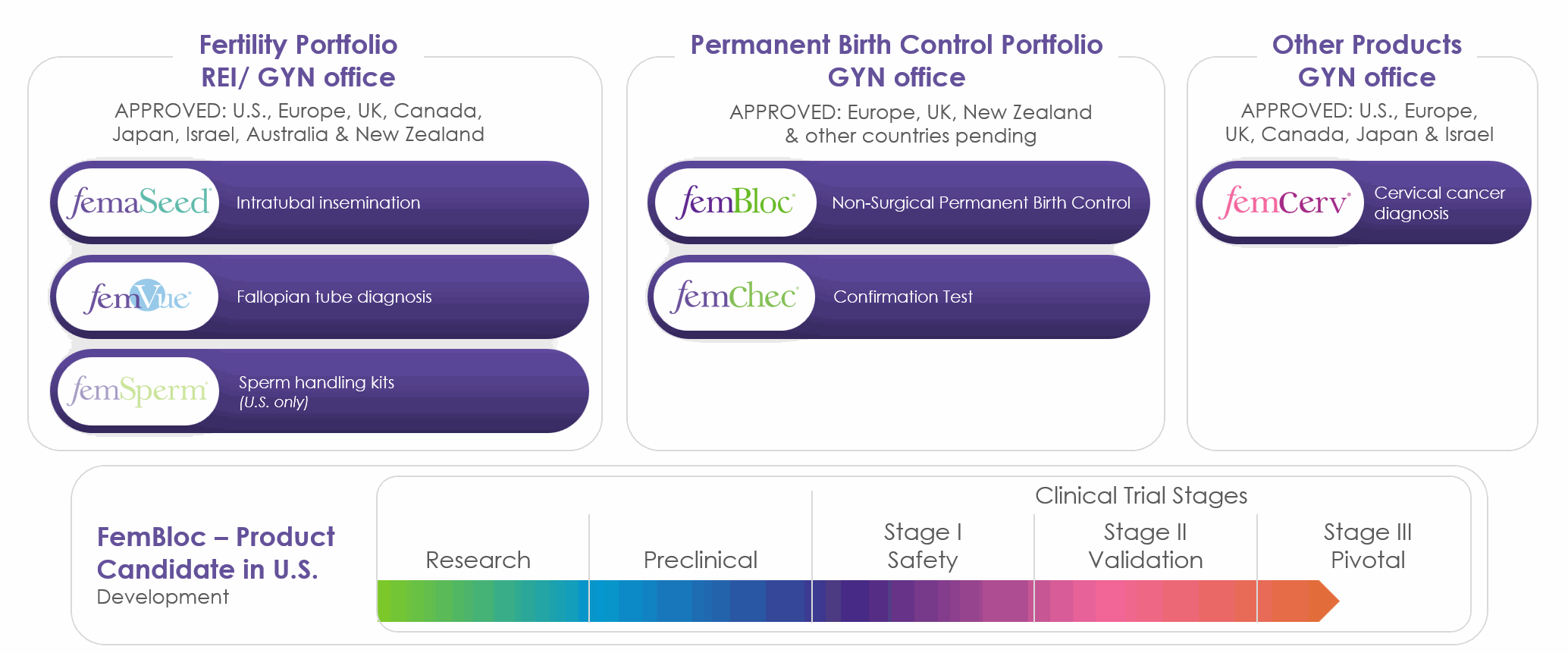
FemaSeed – Our Artificial Insemination Therapeutic Solution, FemVue – Our Companion Diagnostic for Tubal Evaluation and FemSperm – Our Sperm Handling Solution. FemaSeed, our FDA-cleared innovative advancement in artificial insemination is designed to enhance fertilization by precisely delivering sperm into the fallopian tube, the natural site of conception. It offers a safe, accessible and cost-effective first-line therapeutic option for infertile women, men and couples seeking pregnancy through insemination. FemaSeed offers a revolutionary alternative to intrauterine insemination (IUI), enabling healthcare professionals to expand their practice services with a more effective approach as demonstrated in the pivotal trial (NCT0468847) for low male sperm count. It serves as an affordable, less burdensome and lower-risk first step before IVF. FemaSeed is U.S. FDA-cleared and approved in Europe, UK, Canada and Israel. Our first-line therapeutic infertility solution, FemaSeed Intratubal Insemination (ITI), is complemented by our diagnostic companion product, FemVue, the first FDA-cleared product that creates natural saline and air contrast for a safe, reliable, real-time evaluation of the fallopian tubes using ultrasound. When combined with a uterine cavity assessment, it provides a comprehensive exam in the comfort of the gynecologist’s office. Since FemaSeed infertility treatment requires at least one open fallopian tube, FemVue is an essential companion diagnostic. FemVue is U.S. FDA-cleared with approvals in Europe, UK, Canada, Japan and Israel. FemVue can be used with our FDA-cleared and marketed FemCath device, which allows for selective evaluation of the fallopian tube. We believe FemVue offers significant advantages over other existing approaches, including being able to provide ultrasound evaluation of a woman’s fallopian tubes as part of an existing diagnostic infertility assessment. The FemSperm product family includes setup, insemination preparation and analysis kits, to expand our infertility portfolio and, for the first time, enable gynecologists to perform in-office sperm preparation and analysis for use with our first-step infertility treatment, FemaSeed Intratubal Insemination.
In April 2021 we received an IDE approval from the FDA that allowed us to initiate a pivotal trial for the FemaSeed device. The first subject was enrolled in July 2021. In October 2022, we announced an updated study design for the pivotal trial to focus on couples experiencing male factor infertility, an underserved patient segment. In April 2023 we received approval to sell FemaSeed in Canada. In September 2023 we announced 510(k) clearance from the FDA for FemaSeed for ITI. The clinical trial was still ongoing at the time of receiving U.S. regulatory clearance from FDA, however, the study was concluded with enrollment completed in November 2023. Topline results of the clinical trial were announced in March 2024. In November 2024, we announced a peer-reviewed publication of positive data from its pivotal trial in the Journal of Gynecology & Reproductive Medicine (JGRM), a leading journal covering gynecology and reproductive medicine. The publication entitled, “FemaSeed Directional Intratubal Artificial Insemination for Couples with Male-Factor or Unexplained Infertility Associated with Low Male Sperm Count,” includes positive data from the pivotal trial (Clinicaltrials.gov NCT04968847). The trial met its primary endpoint with a pregnancy rate per subject of 26.3% (95%CI: 13.4‒43.1%; n=10/38) and 17.5% per cycle (95%LCB: 7.6%, 95%CI: 5.7‒29.4%; n=10/57), which was significantly higher than the performance goal of 7% based on the historical control (one-sided P=0.041). Safety reports were consistent with IUI. The vast majority of subjects stated they would probably or definitely recommend FemaSeed, and investigator satisfaction was similarly high.
Targeted intratubal insemination of washed spermatozoa using the FemaSeed ITI device is a safe artificial insemination technique that demonstrated high effectiveness for couples with male-factor/unexplained infertility associated with low male sperm count. Delivery of washed spermatozoa directly into the utero-tubal ostium and fallopian tube without catheterization likely increases sperm-oocyte interaction, suggestive of improved efficiency over conventional intrauterine insemination particularly for male-factor infertility. In March 2024, we announced the first commercial use of FemaSeed. In September 2024, we announced strategic distribution partnerships for FemaSeed and FemVue in Spain. In October and December 2024 and March 2025, we announced partnerships with prominent infertility center conglomerates, Boston IVF, HRC Fertility and CNY Fertility, respectively. In August and September of 2025, we announced the availability through customized products and selected partnerships, the FemSperm family of product kits.
FemBloc – Our
Permanent Birth Control Solution and FemChec – Our Companion Diagnostic for
Tubal Occlusion Confirmation. FemBloc is our revolutionary first-of-its-kind non-surgical
approach, which involves minimally-invasive placement of a patented delivery
system for precise delivery of our proprietary synthetic tissue adhesive
(blended polymer) into both fallopian tubes simultaneously. Over time, the
blended polymer fully degrades and produces nonfunctional scar tissue to
permanently block the fallopian tubes in a safe and natural approach. This is
in stark contrast to centuries-old surgical sterilization with reported risks
that include infection, minor or major bleeding, injury to nearby organs,
anesthesia-related events, and even death. Along with the various surgical risks,
some patients may not qualify as good surgical candidates due to obesity or
medical comorbidities. The FemBloc non-surgical approach has the potential to
offer a safe and effective, more accessible in-office alternative with fewer
risks, contraindications, and substantially lower cost than the surgical alternative.
Our non-surgical permanent birth control solution, FemBloc, is complemented by
our diagnostic companion product, FemChec, an FDA-cleared product that creates
natural saline and air contrast that is delivered in a controlled manner for a
safe, reliable, real-time evaluation of the fallopian tubes using ultrasound
for confirmation of procedure success often by the same healthcare
practitioner. Since FemBloc required that both fallopian tubes are blocked for
use as permanent birth control, FemChec is an essential companion diagnostic.
FemChec is U.S. FDA-cleared with approvals in Europe and Canada. FemChec can be
used with our FDA-cleared and marketed FemCath device, which allows for
selective evaluation of each fallopian tube. Blocked fallopian tubes are necessary
for successful permanent birth control, and FemChec offers significant
advantages over other existing approaches, including use of existing
ultrasound.
In June 2023 we received an IDE approval from the FDA to allow us to initiate the first phase of the pivotal trial for the FemBloc system followed by a confirmation test with FemChec (Clinicaltrials.gov NCT05977751). The first subject was enrolled in August 2023. The FINALE [Prospective Multi-Center Trial for FemBloc INtratubal Occlusion for TranscervicAL PErmanent Birth Control] pivotal trial is a prospective, multi-center, open-label, single-arm study design with primary endpoint of pregnancy rate, which is to be analyzed once 401 women have relied on FemBloc for one year for permanent birth control. In addition, the study is designed as a roll-in beginning with enrollment of 50 women for a clinical readout primarily of preliminary safety data prior to enrolling the remaining subjects. An interim analysis of clinical data endpoints is planned once 300 women have relied on FemBloc.
In February 2025, we announced a peer-reviewed publication of positive data from our initial clinical trials of FemBloc permanent birth control in the Journal of Gynecology & Reproductive Medicine (JGRM), a leading journal covering gynecology and reproductive medicine. The publication entitled, “FemBloc Non-Surgical Permanent Contraception for Occlusion of the Fallopian Tubes” includes positive data from three initial clinical trials (Clinicaltrials.gov NCT03067272, NCT03433911, and NCT04273594). The pregnancy rate for FemBloc subjects, who met trial eligibility and were determined bilaterally occluded after a confirmation test with FemChec three months post-FemBloc was 0% (95%UCB: 0.057; n=0/51). This is significantly lower than the performance goal of 6% based on the historical control, surgical sterilization (one-sided p-value=0.0426). Safety reports were consistent with those typically observed for intrauterine transcervical procedures, with no on-going safety concerns through five years. There were no reports of serious adverse events (n=0/229). The vast majority of subjects stated they would probably or definitely recommend FemBloc, and investigator satisfaction was similarly high.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. FemBloc has the potential to offer significant advantages over the only existing option, surgical sterilization (i.e., tubal ligation or “having her tubes tied”), including a significant cost savings at likely half the overall cost and a confirmation test to ensure procedure success. In October 2025, we announced the commencement of a post-market surveillance (PMS) clinical study for FemBloc, in accordance with the European Union Medical Device Regulation (EU MDR 2017/745) and supported by numerous thought leaders throughout the region. FemBloc is a procedure that can be completed in a healthcare practitioner’s office, with no anesthesia, no incisions or cannulation, no specialty skill set or capital equipment and minimal pain and recovery time, and no residual implant remaining in the patient’s body after the tissue in-growth develops. We believe there are also significant advantages over other temporary or reversible methods that women may be using in lieu of the surgical tubal ligation option, as FemBloc does not use hormones or leave a long-term implant behind. In November 2025, we announced FDA IDE approval for the final phase of the FINALE pivotal trial based on the successful completion of the initial phase of the trial. In March 2026, we announced the initiation of enrollment in this final phase.
Additional Women’s Health Solutions. Our FemCerv product, an endocervical tissue sampler for cervical cancer diagnosis, is U.S. FDA-cleared and approved in Europe, UK, Canada, and Israel. FemCerv is a tissue sampling device for endocervical curettage that can be used to obtain a comprehensive sample of cervical cells and tissue circumferentially with sample containment within the device to minimize contamination. We sponsored a post-market study of FemCerv where subjects found the procedure to be relatively pain-free and the sample obtained was complete for analysis, which we believe may aid in reliable diagnosis. Our FemCerv product is utilized by gynecologists and will be a focus for our direct United States salesforce in the future as our commercial efforts remains focused on the infertility line of products.
Our Team
We are a woman-founded, woman-led biomedical company, with a team of experienced biotechnology and medical device developers. Our founder and Chief Executive Officer, Kathy Lee-Sepsick, has over 30 years of entrepreneurial and executive experience in the medical technology field with approximately 228 patents issued globally. Dov Elefant, our Chief Financial Officer, has over 30 years of experience leading public and private biotech companies throughout various stages of financing. Dr. James Liu, MD, our Chief Medical Officer, has over 40 years of practicing as a reproductive endocrinologist and expertise in medical affairs and clinical strategy development. Jeremy Sipos, our Chief Technology Officer, has over 20 years of research and development and engineering experience in the medical device industry. Our experienced leadership team with concentrated development and execution expertise has an unwavering commitment to advancing women’s health. We have raised over $170 million since inception from both institutional and strategic investors, including Medtronic and executives from leading life science companies and our initial public offering, or IPO, in June 2021.
Our Intellectual Property and Production Capabilities
We have designed and developed the proprietary methods utilized in our women’s health solutions so that they are protected by patents, know-how, and trade secrets. Each product and U.S. product candidate in our portfolio is covered by both design and utility patents in the U.S. and significant ex-U.S. markets. As of December 31, 2025, we owned 41 issued U.S. patents and 187 issued foreign patents, 15 pending U.S. patent applications and 41 pending foreign patent applications. These issued patents, and any patents granted from such applications, are expected to expire between 2026 and 2049, without taking potential patent term extensions or adjustments into account.
All of our products are manufactured or assembled at our facility, and manufacturing activities are conducted to ensure compliance with the FDA and other international governing bodies, and good manufacturing practices with significant CMC and device manufacturing infrastructure in compliance with QSR. We have passed numerous manufacturing audits, including those by the FDA and international notified bodies.
Our Strategy
Our goal is to become a global leader in women’s health providing safe and effective solutions that have the potential to disrupt and grow the market segments that they address. To achieve this goal and to contribute to our future success and growth, we are pursuing the following strategies:
Address unmet clinical needs in multiple large markets for women. Our initial focus is on critical areas of unmet need in reproductive health, which is a growing challenge for women that is not optimally addressed with existing therapies. Two ends of the spectrum (infertility treatment with artificial insemination and contraception with permanent birth control) represent large, growing total addressable market opportunities. Patients who wish to control their risk of pregnancy are often utilizing temporary or reversible options or are choosing the only permanent option that bears surgical risk and expense. We believe our FemBloc system has the potential to offer the first non-surgical, non-implant option performed exclusively in the provider’s office without the risk of general anesthesia and incisions, which would potentially allow a doctor to perform multiple procedures in the same room. We estimate that the U.S. market for the FemBloc system may be over $20 billion with an immediate addressable market of over $3 billion annually. We consider those electing surgery for permanent birth control annually to be our immediately addressable market. On the other end of the spectrum, patients who are struggling to become pregnant or are seeking assistance to become pregnant (e.g., same sex couples or single women) are often referred to highly specialized healthcare practitioners for treatment with age-old technology. We believe our FemaSeed product has the potential to offer a first-line approach with local delivery of sperm directly to the fallopian tube where conception occurs. We estimate the immediately addressable U.S. market for FemaSeed, FemVue, a companion diagnostic product, and FemSperm, for in-office handling of sperm by the gynecologist, may be over $1 billion.
Execute on our clinical program to achieve FDA approval to advance our FemBloc system for use together with FemChec, our companion diagnostic device for ultrasound confirmation, as the preferred option for permanent birth control for women. We have studied FemBloc in three earlier clinical trials, each pursuant to an FDA-approved IDE evaluating safety in 228 subjects in total. In February 2025, we announced a peer-reviewed publication of positive data from our initial clinical trials of FemBloc permanent birth control in the Journal of Gynecology & Reproductive Medicine (JGRM), a leading journal covering gynecology and reproductive medicine. The publication entitled, “FemBloc Non-Surgical Permanent Contraception for Occlusion of the Fallopian Tubes” includes positive data from three initial clinical trials (Clinicaltrials.gov NCT03067272, NCT03433911, and NCT04273594). The pregnancy rate for FemBloc subjects, who met trial eligibility and were determined bilaterally occluded after a confirmation test three months post-FemBloc was 0% (95%UCB: 0.057; n=0/51). This is significantly lower than the performance goal of 6% based on the historical control, surgical sterilization (one-sided p-value=0.0426). Safety reports were consistent with those typically observed for intrauterine transcervical procedures, with no on-going safety concerns through five years. There were no reports of serious adverse events (n=0/229). The vast majority of subjects stated they would probably or definitely recommend FemBloc, and investigator satisfaction was similarly high.
In June 2023 we received FDA approval of our IDE to evaluate the safety and efficacy of FemBloc, our non-surgical, non-implant, in-office solution for permanent birth control in a pivotal clinical trial. In August 2023, we announced the initiation of enrollment in the first phase of the FINALE [Prospective Multi-Center Trial for FemBloc INtratubal Occlusion for TranscervicAL PErmanent Birth Control] pivotal trial. This prospective, multi-center, open-label, single-arm study design includes pregnancy rate as the primary endpoint, which will be analyzed once 401 women have used FemBloc for one year for permanent birth control. In addition, the study is designed as a roll-in beginning with enrollment of 50 women for a clinical readout primarily of preliminary safety data prior to enrolling the remaining subjects. An interim analysis of clinical data endpoints is planned once 300 women have used FemBloc for permanent birth control for one year. Follow-up will continue annually for five years post-market. In November 2025, we announced FDA IDE approval for the final phase of the FINALE pivotal trial based on the successful completion of the initial phase of the trial. In March 2026, we announced the initiation of enrollment in this final phase.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In October 2025, we announced the commencement of a post-market surveillance (PMS) clinical study for FemBloc, in accordance with the European Union Medical Device Regulation (EU MDR 2017/745) and supported by numerous thought leaders throughout the region. This PMS study is a key component of Femasys’ long-term commitment to monitor the safety and performance of its products in real‑world settings while complying with the rigorous post-market requirements introduced under the MDR framework (Annex XIV Part B).
Execute on our commercial strategy to market our FemaSeed product for use together with FemVue, our companion diagnostic, and FemSperm - our Sperm Handling Solution, as the first-line option for infertility treatment and build a commercialization infrastructure with a specialized direct sales and marketing team. In April 2023 we received approval to sell FemaSeed in Canada. In September 2023, we announced 510(k) clearance from the FDA for FemaSeed for Intratubal Insemination (ITI). We concluded the clinical trial that had been ongoing when we received 510(k) clearance from the FDA, with enrollment completed in November 2023. In March 2024, topline data were announced, followed by the data published in the Journal of Gynecology & Reproductive Medicine (JGRM) in November 2024. The publication titled, “FemaSeed directional intratubal artificial insemination for couples with male-factor or unexplained infertility associated with low male sperm count,” includes positive data from the pivotal trial (Clinicaltrials.gov NCT04968847). The trial met its primary endpoint with a pregnancy rate per subject of 26.3% (95%CI: 13.4‒43.1%; n=10/38) and 17.5% per cycle (95%LCB: 7.6%, 95%CI: 5.7‒29.4%; n=10/57), which was significantly higher than the performance goal of 7% based on the historical control (one-sided P=0.041). Safety reports were consistent with IUI. The vast majority of subjects stated they would probably or definitely recommend FemaSeed, and investigator satisfaction was similarly high. In March 2024, we announced the first commercial use of FemaSeed. In September 2024, we announced the strategic distribution partnerships for FemaSeed and FemVue in Spain. In October and December 2024 and March 2025, we announced partnerships with prominent infertility center conglomerates, Boston IVF, HRC Fertility, and CNY Fertility, respectively. FemVue is U.S. FDA-cleared with approvals in Europe, UK, Canada, Japan and Israel. In March 2024, we announced the first commercial use of FemaSeed. In September 2024, we announced the strategic distribution partnerships for FemaSeed and FemVue in Spain. In October and December 2024 and March 2025, we announced partnerships with prominent infertility center conglomerates, Boston IVF, HRC Fertility, and CNY Fertility, respectively. The FemSperm product family includes setup, insemination preparation and analysis kits, to expand our infertility portfolio and, for the first time, enable gynecologists to perform in-office sperm preparation and analysis for use with our first-step infertility treatment, FemaSeed Intratubal Insemination.
Continuously innovate to introduce additional product offerings for women. We intend to continue to invest in research and development activities focused on improvements and enhancements to our FemaSeed and FemBloc system and other existing products, and additional women-specific medical products. We have designed and developed proprietary methods utilized in our women’s health solutions and have protected these internally conceived advancements by patents, know-how, and trade secrets. Our team has demonstrated the ability to achieve marketing authorizations and clearances in the U.S., Europe, Canada, Japan, UK and Israel, and to manufacture in accordance with FDA and other international governing bodies. Availability of the additional product offerings will expand our suite of solutions for reproductive health and women’s health in general over time with the goal of addressing aspects of care that have had negligible advancement over decades to create improved patient care and improved healthcare practitioner treatment options.
Penetrate the addressable markets by promoting patient and practice awareness. It is estimated in the U.S. alone over 10 million women (https://www.cdc.gov/nchs/nsfg/key_statistics/i-keystat.htm) are infertile and approximately 40-50% of all infertility is attributed to male factor (Kumar et al., 2015) likely due to the over 50% decline in male sperm count worldwide (Levine et al., 2023). Only a little over half of women proceed with some form of intervention and only a very small proportion undergo more advanced technologies. We believe the major factor that influences this light penetration of the market is the cost and burden of the existing technologies despite the familiarity of intrauterine insemination and in-vitro fertilization (IVF) as options. We intend to increase healthcare practitioner awareness through engagement and continued publication of scientific data in peer reviewed journals. Additionally, we intend to engage women and couples suffering from infertility or who wish to undergo insemination for pregnancy through direct patient outreach. On the other end of the spectrum, it is estimated in the U.S. alone, approximately 1.2 million women elect surgical tubal ligation (Martinez, 2024) and 500,000 men elect vasectomy (Ostrowski, et al., 2018) annually for permanent birth control. There are another 12 million women who utilize a non-permanent birth control option (Daniels, et al., 2020), many of whom we believe may prefer a permanent option if it were non-surgical. We believe the major factor that influences this light penetration of the market is the limitations of existing technology despite the likely familiarity of surgical tubal ligation as an option. Like our infertility portfolio, we plan to increase healthcare practitioner awareness prior to direct patient outreach.
From the outset, we spent significant time understanding the unmet needs of patients and healthcare practitioners through patient and healthcare practitioner surveys and early engagement of healthcare practitioners and key opinion leaders to properly position our solutions. We have established an initial commercial infrastructure following the clearance of our FemaSeed product. Our already commercially available FemVue device is being marketed along with the FemaSeed product to the same target healthcare practitioner: the reproductive endocrinologist. We intend to focus the significant majority of our sales and marketing efforts in North America since we believe that initially nearly 90% of the potential annual global FemaSeed/FemVue/FemSperm sales would be generated in this market. Our priority in the U.S. is to target existing FemVue customers followed by gynecologists with the new FemSperm product line in non-covered states, and reproductive endocrinologists in high volume areas. We have hired a sales force with strong expertise in gynecology, of approximately 10 sales representatives for our infertility products and plan to increase the sales force as necessary for the FemBloc system, to the target healthcare practitioner: the gynecologist. Our already commercially available FemCerv device will be marketed along with the FemBloc product to the same target healthcare practitioner: the gynecologist. In addition, we plan to continue to expand our in-house manufacturing capabilities as we scale to meet the demand and introduce new products while evaluating potential suppliers to assess the viability of outsourcing portions of our manufacturing and assembly processes to ensure significant growth, profitability and operating leverage.
Expand gynecologists’ practice capabilities by diversifying products and services to include artificial insemination with FemaSeed. There are a limited number of gynecological practices performing infertility services and treatment today, but we believe this has the potential to grow over time, in particular with the introduction of FemSperm, enabling sperm handling for use with FemaSeed. FemaSeed is designed to be an in-office infertility procedure that can be done by a gynecologist or advanced practice provider (i.e., nurse, physician assistant) as applicable using his or her existing skillset, expanding the number of gynecological practices that can offer effective fertility services to their patients without needing to refer them to an infertility specialist. We plan to use our gynecologic sales force for FemBloc, if approved, to introduce those healthcare practitioners that have adopted FemaSeed as the two product portfolios share a similar delivery platform.
The Current Market Landscape
For permanent birth control, tubal ligation, an invasive surgical procedure requiring implants, incisions, hospitalization and general anesthesia, has been offered for decades, so risks are known. It is performed either immediately after cesarean delivery or via laparoscopic procedures, which has notable disadvantages and risk of complications. The most significant morbidity arising from surgical tubal ligation is associated with the use of electrical energy and inadvertent thermal damage to the bowel. Introduction of surgical instruments into the abdominal cavity carries substantial risk of injury to intra-abdominal organs and blood vessels, with approximately 1% of all procedures resulting in unintended further major surgery. In addition, anesthesia risk, bleeding, bowel damage, and long recovery times are inherent complication risks of surgical tubal ligation. Temporary and reversible contraceptive methods, such as birth control and intrauterine devices (IUDs), are being used by women long-term as a compromise by women who are unwilling to undergo a surgical sterilization procedure because of the surgical risks, not wanting an incision, or to be exposed to risk of anesthesia. Some may be contraindicated for surgical sterilization because of obesity or medical conditions. Long-term use of hormonal birth control, including IUDs, have drawbacks as well. Hormonal birth control is associated with health risks, such as an increased risk of breast cancer and blood clots, and device-based birth control can result in uterine perforation and increased risks of pelvic inflammatory disease and ectopic pregnancy. Previously available non-surgical methods utilizing permanent implants for closing the fallopian tubes have been removed from the market due to safety or intellectual property infringement issues, and thus the only currently available permanent birth control option is surgical tubal ligation.
For artificial insemination, traditional intrauterine insemination is an undirected procedure delivering sperm into the uterine cavity that has been offered for decades. IUI continues to be offered as a first-line treatment option in spite of its low success rate due to its low cost and ease, with a short learning curve and minimal equipment requirements. Although current IUI devices address the unfavorable environment sperm would encounter in the vagina and cervix by placing sperm into the uterine cavity, the biology of sperm transport is complex and of the millions of sperm inseminated in the uterus, almost all fail to reach the fallopian tubes. In contrast to the unfavorable environment of the uterus, the fallopian tubes act as a reservoir for traveling sperm and is the location of conception. Sperm count is declining at an accelerated pace globally by greater than 50% with an increasing proportion of men having sperm counts below any given threshold for sub-fertility or infertility. This substantial and persistent decline is now recognized as a significant public health concern. (Levine et al., 2023). In vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI) are highly effective treatments with reported pregnancy rates of approximately 25% for male factor infertility, however, these approaches are associated with significant cost and clinical risks (Ravitsky et al., 2019). Many infertile women and couples are unwilling to undergo treatment mostly due to financial reasons as services are provided by referral to an infertility center. IVF is extremely expensive, costing as much as $15,000 to $30,000 per IVF cycle according to Forbes Health, 2021 (with cycle effectiveness usually only around 25%), and often not covered by insurance. Genetic testing of the eggs or embryo, also known as preimplantation genetic testing (PGT), is an often optional cost up to $10,000 or more if disorders are tested. IVF/ICSI is also physically and emotionally demanding for the patient, with an increased risk of ectopic pregnancy and miscarriage. The American Society for Reproductive Medicine provided guidance (committee opinion, 2021) promoting singleton gestation to reduce the risk of multiple pregnancies. There are over ten million women in the United States known to be infertile and only approximately 200,000 IVF cycles completed per year, indicating that IVF is not a realistic solution available to most women or couples. Our FemaSeed product establishes a new category within artificial insemination: a localized, directional delivery of sperm directly into the fallopian tube (intratubal insemination), precisely where conception occurs. We believe this in-office, cost-effective solution can become a first-line treatment for infertility, specifically when male factor is involved, increasing access to infertility treatment for women, couples and the LGBTQ community. The FemSperm product line activates the gynecologist for sperm handling for use with FemaSeed, eliminating the referral to an infertility specialist.
The Reproductive Health Opportunity
There are an estimated 78 million reproductive aged women in the United States alone (World Health Organization, 2025). We intend to offer comprehensive solutions for preventing pregnancy and achieving pregnancy for women, providing cost-effective and safe solutions while avoiding surgery. During their childbearing years, most women will want to control their risk of pregnancy. Additionally, there are many women who wish to become pregnant that are unable to do so. According to a Centers for Disease Control and Prevention (CDC) report, the ability to plan when to be pregnant and how many pregnancies to initiate has been called one of the ten great public health achievements in the twentieth century. Many women, who spend an average of three years seeking to become pregnant and thirty years avoiding pregnancy, are not satisfied with the current methods for preventing unwanted pregnancies and achieving pregnancy (Guttmacher Institute, 2019).
Approximately 1.2 million women undergo surgical tubal ligation each year in the United States alone (Martinez, 2024), with an average cost of approximately $6,000 per procedure (Planned Parenthood, 2019). The total cost is estimated to be higher with the increase in surgical operating room costs. However, there are over an estimated 12 million women who remain on a non-permanent birth control option long-term (National Center for Health Statistics, 2015), which we believe is due to there being only a surgical permanent contraceptive option available to women. In addition, 500,000 men undergo a vasectomy procedure every year (Ostrowski et al., 2018). While the 1.7 million women and their partners annually who want to permanently prevent pregnancy represent our initial near-term market opportunity, we believe these numbers do not reflect the true demand for permanent birth control, as many do not want to submit to invasive surgical procedures such as vasectomies and tubal ligations. The market for female permanent birth control is large and growing, and we believe the market opportunity in the U.S. alone could expand to exceed $20 billion with a safe and effective in-office option as women shift from temporary or reversible contraceptive alternatives to more permanent solutions.
The overall decline in birth rates in the United States and globally has resulted in aging populations that present serious challenges for the global economy and economic stability. In the United States alone, it is estimated by the Centers for Disease Control and Prevention that over ten million women desire pregnancy but are unable to achieve pregnancy (National Survey of Family Growth, 2019). Only a little over half of these women proceed with some form of intervention, and only a very small proportion undergo more advanced assisted reproductive technologies such as IVF at a referral infertility center. Approximately 40-50% of infertility cases are due to a male factor (Kumar et al., 2015), which may be the result of the greater than 50% decline worldwide in male sperm count (Levine et al., 2023). Although IUI, an artificial insemination option, is the oldest technique in reproductive medicine and a well-accepted first-line treatment method for couples with unexplained infertility, mild male factor infertility, sexual dysfunction, and cervical factor infertility, its success rates remain relatively low. However, for couples with low total motile sperm count, treatment with highly effective IVF/ ICSI is advised given the comparatively reduced success rates for IUI. Alternative methods to IUI have not been advanced to meet the continuous demand for safe and effective first-line alternatives that are considerably less costly and less invasive than more advanced assisted reproductive options. No methods have been introduced to the primary gynecologists to handle the sperm required for use in artificial insemination procedures, including FemaSeed. The market for assisted reproduction is large and growing, and we believe the market in the United States alone could exceed $1 billion with a safe and effective novel first-line approach as women move to seek care for the treatment of infertility.
Clinical Development
Overview of Clinical Programs. We are developing a growing body of compelling clinical evidence for our intrauterine directional delivery products and U.S. product candidate.
Our Permanent Birth Control Solution – FemBloc and ultrasound confirmation with FemChec
Clinical Trials
In June 2023 we received FDA approval of our IDE to evaluate the safety and efficacy of FemBloc, our non-surgical, non-implant, in-office solution for permanent birth control in a pivotal clinical trial. In August 2023, we announced the initiation of enrollment in the first phase of the FINALE [Prospective Multi-Center Trial for FemBloc INtratubal Occlusion for TranscervicAL PErmanent Birth Control] pivotal trial. This prospective, multi-center, open-label, single-arm study design includes pregnancy rate as the primary endpoint, which will be analyzed once 401 women have used FemBloc for one year for permanent birth control. In addition, the study is designed as a roll-in beginning with enrollment of 50 women for a clinical readout primarily of preliminary safety data prior to enrolling the remaining subjects. An interim analysis of clinical data endpoints is planned once 300 women have used FemBloc for permanent birth control for one year. Follow-up will continue annually for five years post-market. In November 2025, we received FDA approval of our IDE for the final phase of the FINALE pivotal trial based on successful completion of the initial phase. In March 2026, we announced initiation of enrollment in this final phase.
In February 2025, we announced a peer-reviewed publication of positive data from its initial clinical trials of FemBloc permanent birth control in the Journal of Gynecology & Reproductive Medicine (JGRM), a leading journal covering gynecology and reproductive medicine. The publication entitled, “FemBloc Non-Surgical Permanent Contraception for Occlusion of the Fallopian Tubes” includes positive data from three initial clinical trials (Clinicaltrials.gov NCT03067272, NCT03433911, and NCT04273594). The pregnancy rate for FemBloc subjects, who met trial eligibility and were determined bilaterally occluded after a confirmation test three months post-FemBloc was 0% (95%UCB: 0.057; n=0/51). This is significantly lower than the performance goal of 6% based on the historical control, surgical sterilization (one-sided p-value=0.0426). Safety reports were consistent with those typically observed for intrauterine transcervical procedures, with no on-going safety concerns through five years. There were no reports of serious adverse events (n=0/229). The vast majority of subjects stated they would probably or definitely recommend FemBloc, and investigator satisfaction was similarly high.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In October 2025, we announced the commencement of a post-market surveillance (PMS) clinical study for FemBloc, in accordance with the European Union Medical Device Regulation (EU MDR 2017/745) and supported by numerous thought leaders throughout the region. This PMS study is a key component of Femasys’ long-term commitment to monitor the safety and performance of its products in real‑world settings while complying with the rigorous post-market requirements introduced under the MDR framework (Annex XIV Part B).
Products
Our Artificial Insemination Solution.
510(k) Clearance for FemaSeed Intratubal Insemination Device
FemaSeed, our FDA-cleared innovative advancement in artificial insemination, is designed to enhance fertilization by precisely delivering sperm into the fallopian tube, the natural site of conception. It offers a safe, accessible and cost-effective first-line therapeutic option for infertile women, men and couples seeking pregnancy through insemination. FemaSeed offers a revolutionary alternative to IUI, enabling healthcare professionals to expand their practice services with a more effective approach as demonstrated in the pivotal trial (NCT0468847) for low male sperm count. It serves as an affordable, less burdensome and lower-risk first step before IVF. FemaSeed is U.S. FDA-cleared and approved in Europe, UK, Canada and Israel.
We have studied FemaSeed pursuant to an FDA-approved IDE received in April 2021 to evaluate safety and efficacy in the LOCAL [Prospective Multi-Center LOCALized Directional Insemination Trial for Artificial Insemination] pivotal trial. In October 2022, we announced an updated study design for the pivotal trial, which focused on couples experiencing male factor infertility. The clinical trial was still ongoing at the time of receiving U.S. regulatory clearance from FDA, however, the study concluded with enrollment completed in November 2023. Topline results of the clinical trial were announced in March 2024. The data was published in the Journal of Gynecology & Reproductive Medicine (JGRM) in November 2024. The publication titled, “FemaSeed directional intratubal artificial insemination for couples with male-factor or unexplained infertility associated with low male sperm count,” includes positive data from the pivotal trial (Clinicaltrials.gov NCT04968847). The trial met its primary endpoint with a pregnancy rate per subject of 26.3% (95%CI: 13.4‒43.1%; n=10/38) and 17.5% per cycle (95%LCB: 7.6%, 95%CI: 5.7‒29.4%; n=10/57), which was significantly higher than the performance goal of 7% based on the historical control (one-sided P=0.041). Safety reports were consistent with IUI. The vast majority of subjects stated they would probably or definitely recommend FemaSeed, and investigator satisfaction was similarly high. Targeted intratubal insemination of washed spermatozoa using the FemaSeed ITI device is a safe artificial insemination technique that demonstrated high effectiveness for couples with male-factor/unexplained infertility associated with low male sperm count. Delivery of washed spermatozoa directly into the utero-tubal ostium and fallopian tube without catheterization likely increases sperm-oocyte interaction, suggestive of improved efficiency over conventional intrauterine insemination particularly for male-factor infertility. In March 2024, we announced the first commercial use of FemaSeed. In September 2024, we announced the strategic distribution partnerships for FemaSeed and FemVue in Spain. In October and December 2024 and March 2025, we announced partnerships with prominent infertility center conglomerates, Boston IVF, HRC Fertility and CNY Fertility, respectively.
510(k) Clearance for Intrauterine Device for Selective Tubal Evaluation
FemCath Cornual Balloon Catheter, an innovative advancement in selective tubal evaluation, designed similar to FemaSeed but for a different indication, is authorized for marketing in the U.S., Europe and Canada. A post-market study in 23 subjects (45 tubes) was conducted with the product and FemVue Saline-Air device to assess fallopian tubes selectively by infusion of saline-air contrast. 89% had contrast observed entering the proximal portion of the fallopian tube combined with sustained flow or exit into the peritoneal cavity. This is the same intended location of delivery for the FemaSeed product. There were no serious adverse events reported. We began limited market release of the FemCath product in December 2022, and it is being utilized in the pivotal trial for FemBloc, as part of the confirmation test with FemChec.
Post-Market Information
FemVue Saline - Air device, a contrast-generating product, is authorized for marketing in the United States, Europe, UK, Canada, Japan and Israel. There have been multiple publications and abstracts presented with clinical evidence in support of FemVue, a diagnostic companion product to our artificial insemination solution. It has been concluded that tubal patency assessment with FemVue is comparable to fluoroscopic hysterosalpingogram (HSG), and it appears to be a convenient and well-tolerable method that may be performed alongside conventional ultrasound and uterine cavity assessment as part of an infertility evaluation. Other publications have stated that FemVue is an accurate test for diagnosing tubal occlusion, performs similarly to a fluoroscopic HSG and could potentially replace fluoroscopic HSG.
Our Endocervical Tissue Sampler.
Post-Market Information
FemCerv, a next generation endocervical tissue sampler for cervical cancer diagnosis, is U.S. FDA-cleared with approvals in Europe, UK, Canada and Israel. We sponsored a post-market study for FemCerv in 112 patients undergoing further evaluation of an abnormal cervical tissue result. It was observed that FemCerv provided samples were reported as adequate when evaluated histologically in 94% of the patients. Healthcare practitioners reported that 95% of the patients experienced mild or no discomfort during the FemCerv procedure and 92% of the healthcare practitioners reported the FemCerv device as easy to insert. There were no adverse events reported. We began limited market release in the U.S. in September 2022; however, the commercial efforts are currently focused on the infertility portfolio.
Manufacturing
We have developed and implemented the infrastructure required to manufacture and distribute finished medical devices, including a robust medical device quality management system, which meets the requirements of the FDA Quality System Regulation, and is certified to Medical Device Single Audit Program (MDSAP) and International Organization for Standardization (ISO) 13485:2016. We currently manufacture or assemble all products and U.S. product candidate and source components from contract suppliers. We believe that we currently have sufficient capacity to meet clinical program demands, product supply and launch requirements for the FemaSeed and FemBloc products, both in the U.S. and in select international countries, and believe that we will be able to scale up our capacity relatively quickly with modest capital investment. We believe our manufacturing capacity is sufficient to meet global market demand for our products for the foreseeable future until potential approval by the FDA for the FemBloc system. While we plan to continue manufacturing our product and U.S. product candidate, if approved or granted marketing authorization, we will consider outsourcing arrangements for certain sub-assemblies as needed as we scale our commercial production.
We employ a rigorous supplier assessment, qualification, and selection process targeted to suppliers that meet the requirements of the FDA and the ISO and quality standards supported by internal policies and procedures. Our quality assurance process monitors and maintains supplier performance through qualification and periodic supplier reviews and audits. We are required to maintain ISO 13485 certification for medical devices sold in the European Economic Area (EEA) which requires, among other items, an implemented quality system that applies to component quality, supplier control, product design and manufacturing operations. In June 2024, we announced that we received the European Union Medical Device Regulation (EU MDR) certificate for all products in the portfolio except FemBloc, demonstrating Femasys’ compliance with the highest required regulatory standards. Subsequently in March 2025 we announced receiving EU MDR for the delivery system component of FemBloc and June 2025 for the class III blended polymer component of FemBloc. In February 2026, we announced MDSAP certification for FemBloc.
We inspect, test, and assemble our products under strict manufacturing processes supported by internal policies and procedures with significant CMC and device manufacturing infrastructure. We perform our own final quality control testing of each product and U.S. product candidate, and we have complete control over all aspects of the manufacturing process and are compliant with QSR good manufacturing practice regulations applicable to our products.
Our suppliers are managed through our supplier management program that is focused on reducing supply chain risk. Key aspects of this program include managing component inventory at the supplier and second sourcing approaches for specific suppliers. Typically, our outside vendors produce components to our specifications and in many instances to our designs. Our suppliers are audited periodically by our quality department to ensure conformity with the specifications, policies and procedures for our products. In addition, we and our suppliers are subject to periodic unannounced inspections by U.S. and international regulatory authorities to ensure compliance with quality regulations. We believe that, if necessary, alternative sources of supply would be available in a relatively short period of time and on commercially reasonable terms.
We do not have long-term supply agreements and we purchase certain components for our products on a purchase order basis. We do not currently have arrangements in place for redundant supply of certain components of our products. If our current third-party suppliers cannot perform as agreed, we may be required to replace those suppliers. Although we believe that there are several potential alternative suppliers who could provide these components, we may incur added costs and delays in identifying and qualifying any such replacement.
Finally, for our products and U.S. product candidate, we utilize third-party sterilizers to ensure these single-use products are packaged and shipped sterile for use. If our current contract sterilizer(s) cannot continue to perform as agreed, we may be required to identify and contract with another third-party contract sterilizer which may incur added costs and delays in identifying and qualifying any such replacement.
Competition
The markets in which we compete are highly competitive and in limited areas are characterized by rapid and significant technological change. To compete successfully, we need to continue to demonstrate the advantages of our products and U.S. product candidate compared to both well-established and new alternative procedures, products and technologies, and convince healthcare practitioners and other healthcare decision makers of the advantages of our products and technologies.
With respect to our permanent birth control solution, we expect to compete with surgical tubal ligation, vasectomies for women’s partners, other methods of non-permanent birth control, including devices such as IUDs, prescription drugs such as the birth control pill and injectable and implantable contraceptives and patches, and other contraceptive and birth control methods. There is no directly competing non-surgical, non-implant permanent birth control product currently on the market, or, to our knowledge, in development.
With respect to our artificial insemination solution, we expect to compete with IUI, IVF/ ICSI and fertility-enhancing pharmaceuticals currently in the market and those in clinical and preclinical development. While there is no direct competitor in our segment of the product category of artificial insemination, there are alternatives, such as IUI and IVF/ ICSI. Leading companies that produce IUI devices include Cook Medical LLC, a subsidiary of Cook Group, Inc., Cooper Surgical, Inc., MedGyn Products, Inc. and Rocket Medical LLC.
With respect to our tissue sampling product, there are other procedures used in women’s health to evaluate the cervical canal, such as the Pap test, HPV test and colposcopy, which are well established and pervasive. Companies such as Dysis Medical and Guided Therapeutics are also developing cervical tissue sampling product candidates.
Many of our competitors have access to greater resources required to develop and market a competitive product than we do. In addition, new competition and products may arise due to consolidation within the industry and other companies may develop products that could compete with our products or U.S. product candidate, and there may be product candidates in early stages of development of which we are not aware.
Sales & Marketing
FemaSeed – Our Artificial Intratubal Insemination Solution and FemSperm - our Sperm Handling Solution.
With the 510(k) clearance from the FDA and regulatory approval from Europe, UK, Canada and Israel for FemaSeed, we have recruited a direct sales force comprising approximately 10 sales representatives with strong sales backgrounds and experience in medical device sales to gynecologists for commercialization in the U.S. We intend to focus the significant majority of our direct sales and marketing efforts in North America for our infertility portfolio Secondary, we will explore distribution partners for international markets. Our initial target prior to FemSperm availability was the approximately 1,700 reproductive endocrinologists at 450 practices, who are trained and have experience performing infertility procedures. Over 50% of the practices representing approximately 60% of the assisted reproduction cycles performed are located in 8 states, and are prioritized, along with the existing FemVue customers. The introduction of the FemSperm product line expands our commercial focus to gynecologists, who are often the primary healthcare practitioners for women seeking to become pregnant. By providing a simplified sperm preparation solution designed for use within the gynecologist’s office, FemSperm has the potential to broaden access to fertility treatment earlier in the patient journey. Our initial commercialization strategy prioritizes states that do not mandate insurance coverage for in vitro fertilization (IVF), where patients may be more likely to seek lower-cost fertility treatment options prior to pursuing IVF.
We plan to engage in awareness-building activities designed to inform healthcare practitioners and patients about the clinical benefits and potential applications of our FemaSeed product in the United States. These activities are intended to increase familiarity with FemaSeed as an in-office therapeutic option for infertility treatment. Our efforts will focus initially on educating healthcare practitioners, particularly gynecologists and fertility specialists, regarding the role of FemaSeed as a potential first-line therapeutic approach for certain infertility patients and for those seeking artificial intratubal insemination as a pathway to pregnancy. We expect that increasing practitioner awareness and adoption will, in turn, support broader patient awareness and utilization. Our educational and awareness initiatives may include professional education, clinical engagement, and other programs intended to facilitate understanding of the procedure, its potential benefits, and its role within the fertility treatment pathway.
FemBloc – Our Permanent Birth Control Solution.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In October 2025, we announced the commencement of a post-market surveillance (PMS) clinical study for FemBloc, in accordance with the European Union Medical Device Regulation (EU MDR 2017/745) and supported by numerous thought leaders throughout the region. This PMS study is a key component of Femasys’ long-term commitment to monitor the safety and performance of its products in real‑world settings while complying with the rigorous post-market requirements introduced under the MDR framework.
If approved, we plan to leverage our existing U.S. commercial infrastructure supporting FemaSeed to introduce FemBloc to gynecologists who perform infertility evaluation and treatment. We expect to expand our direct sales organization in the United States and recruit representatives with experience in medical device sales and, where possible, established relationships with gynecologists. Our commercial efforts will focus primarily on North America, while we continue to utilize distribution partners for international markets. Through our direct sales organization, we plan to target approximately 40,000 gynecologists in the United States who perform gynecologic procedures and provide family planning services. Over 60% of practices representing more than 60% of reproductive-aged women are located in 13 states, which we intend to prioritize.
Based on our clinical experience to date, we believe healthcare practitioners experienced in intrauterine procedures, such as IUD placement, will require minimal training to start utilizing the FemBloc system, similar to our experience with our FemaSeed product. Additional sonographic training may be required for the healthcare practitioners or sonographers performing the ultrasound confirmation test. Based on our clinical experience to date, we believe the healthcare practitioners and sonographers will require minimal training and it can be accomplished largely through online training program. Those with FemVue experience will require minimal training for the confirmation test. We expect to begin building our sales organization in advance of potential FDA approval of the product candidate in the U.S.
We plan to conduct awareness education initiatives, highlighting the benefits of our FemBloc system in jurisdictions where we are approved to market. These activities are intended to increase familiarity among healthcare practitioners and patients with FemBloc as the first non-surgical, non-implant option for permanent birth control.
Reimbursement
In the United States, we anticipate deriving the majority of our revenue from the sale of products in our infertility portfolio, including the FemaSeed, FemVue, and FemSperm products, to gynecologists and infertility specialists. Fertility-related procedures are frequently paid directly by patients, as insurance coverage for infertility services varies significantly by state and by payor. As a result, providers commonly bill patients directly for procedures and associated products, including FemaSeed and FemVue. The prevalence of cash-pay fertility services may facilitate adoption of new procedures and technologies that offer alternative treatment options within the physician’s office setting. In some cases, when patients have applicable insurance coverage, providers may seek reimbursement from third-party payors such as private insurance companies and health maintenance organizations.
When third-party payors are involved in the United States, healthcare practitioners typically identify the procedure or service for reimbursement using Current Procedural Terminology (CPT) codes, which are a five-digit alphanumeric codes and maintained by the American Medical Association (“AMA”) and used for reporting and billing medical procedures. Long-established procedures, such as intrauterine insemination, have recognized CPT codes that facilitate billing and reimbursement. Newer procedures may initially lack established CPT codes, which can make reimbursement less straightforward even when coverage may be available. In February 2026, the AMA approved a new Category III CPT code for the FemaSeed procedure, which is expected to become effective in January 2027. Category III CPT codes are used to track emerging technologies and procedures and may support future reimbursement discussions with payors.
Outside of the United States, reimbursement processes vary significantly by country and region. In markets operating under single-payor healthcare systems, direct patient payment may still occur for fertility procedures where public healthcare budgets limit the number of fertility treatments covered. Reimbursement may therefore be obtained from a variety of sources, including government-sponsored insurance plans, private health insurance plans, or direct patient payment, depending on the applicable healthcare system.
In the United States, if approved, we expect to derive revenue from the sale of our FemBloc system primarily through gynecology practices performing in-office permanent contraception procedures. Providers typically seek reimbursement from third-party payors, including Medicare, Medicaid, private insurance companies, health maintenance organizations, and other healthcare-related organizations. Any portion of the procedure costs not covered by third-party payors, such as deductibles or co-payments, may be billed directly to the patient by the provider. As a permanent contraceptive method, we expect the FemBloc procedure may be eligible for coverage under the contraceptive coverage provisions of the Affordable Care Act (“ACA”), which require most health insurance plans to cover contraceptive services without patient cost-sharing when provided by an in-network provider.
Following potential regulatory approval, we intend to pursue Category I Current Procedural Terminology (“CPT”) codes for the FemBloc procedure. Physician reimbursement under Medicare is generally determined by the Medicare Physician Fee Schedule, where payment levels reflect the relative value assigned to the service performed in the physician office setting. We believe the clinical data generated from our FemBloc program will support our efforts to obtain appropriate coding and reimbursement. We also believe that the development of coding for our FemaSeed infertility procedure, which utilizes a similar catheter-based intrauterine delivery platform, may support familiarity with related procedures using our technology. This shared procedural platform may help facilitate physician adoption and reimbursement discussions for procedures within our women’s health product portfolio.
Outside the United States, reimbursement levels vary significantly by country and region, particularly depending on whether the country or region utilizes a single-payor healthcare system. Annual healthcare budgets in single-payor systems often determine the number of permanent birth control procedures funded in any given year. Reimbursement may be sourced from government-sponsored health programs, private insurance plans, or combinations of both. In some countries or regions, additional clinical data collection may be required, such as post market study data, before coverage and reimbursement are granted for our FemBloc system. We intend to work with payors in select countries and regions to pursue appropriate coverage and reimbursement approvals where economically viable.
Intellectual Property
Our success depends in part on our ability to obtain, maintain, protect and enforce our proprietary technology and intellectual property rights, in particular, our patent rights, preserve the confidentiality of our trade secrets, and operate without infringing the valid and enforceable patents and other proprietary rights of third parties. We rely on a combination of patent, trademark, trade secret, copyright and other intellectual property rights and measures to protect the intellectual property rights that we consider important to our business. We also rely on know-how and continuing technological innovation to develop and maintain our competitive position.
We seek to protect our proprietary rights through a variety of methods, including confidentiality agreements and proprietary information agreements with suppliers, employees, consultants and others who may have access to our proprietary information. However, trade secrets and proprietary information can be difficult to protect. While we have confidence in the measures we take to protect and preserve our trade secrets and proprietary information, such measures can be breached, and we may not have adequate remedies for any such breach. In addition, our trade secrets and proprietary information may otherwise become known or be independently discovered by competitors.
As of December 31, 2025, we owned 41 issued U.S. patents and 187 issued foreign patents, 15 pending U.S. patent applications and 41 pending foreign patent applications. Our patent portfolio includes both utility and design patents and patent applications, both in the U.S. and overseas. Issued patents have a limited term for enforcement, and the termination dates differ for utility and design patents, and from country to country. Expirations of our earlier-filed patents in the US or overseas are anticipated by several strategies. Some earlier-filed design patents were directed to devices that are no longer used or marketed. Design patents with shorter enforcement periods generally have existing utility patents, still in enforcement periods, that continue to protect the subject matter of the device. Additionally, earlier-filed utility patents may reach their termination dates, but these patents recite devices or methods of using the devices that have been supplanted by patents and patent applications for improved devices and methods of using the improved devices prior to the expiration dates earlier-filed utility patents. As part of the evolving products offered by us, products covered by utility patents that have expiration dates in the next two to three years, which generally covered the first generation devices, are being replaced by improved devices that are now protected by issued patents and patent applications, and design and manufacture efforts have developed improved devices that can be protected in a similar manner. These issued patents, and any patents granted from such applications, are expected to expire between 2025 and 2046, without taking potential patent term extensions or adjustments into account. We believe that the patents expiring in 2025 are not material to our business.
In the United States, our controlled delivery FemBloc patent portfolio includes two patent families. They include granted utility and design patents providing protection until at least 2028 and 2039, excluding any eligible patent term adjustments and extensions. One family includes design patents issued in the US and internationally. The utility patent family covering FemBloc for female permanent birth control includes multiple issued and pending US and foreign patents and applications, with an anticipated expiration of at least 2039. Our blended polymer composition patent portfolio includes one patent family. The patent family includes granted utility patents providing protection until at least 2038, and pending utility patent applications in the US and internationally.
Our FemaSeed patent portfolio includes one patent family. The patent family includes granted utility and design patents providing protection until at least 2034 and 2042. We believe that the patents expiring in 2026 are not material to our business. In January 2025, we announced a new U.S. patent covering use of FemaSeed for female infertility treatment with an anticipated expiration 2042 at the earliest.
Our FemVue patent portfolio includes five patent families, including granted utility and design patents providing protection until at least 2026 and 2028. We believe that the patents expiring in 2026 are not material to our business. The two utility patent families include pending patent applications, which if granted, could result in patents expiring in 2028, plus any eligible patent term adjustments and extensions. Our FemVue Mini patent portfolio includes two patent families. The two patent families include granted design patents providing protection until at least 2034 and 2049. The utility patent family includes two pending patent applications, which if it provides the priority for a granted patent, could result in patents expiring at least in 2045, plus any eligible patent term adjustments and extensions.
Our FemChec patent portfolio includes five patent families. The five patent families include granted utility and design patents providing protection until at least 2026, 2028, 2029, 2032, and 2046. The utility patent family includes issued patents and pending patent applications expiring in 2028 and 2031.
Our FemCerv patent portfolio includes two patent families. The two patent families include granted utility and design patents providing protection until at least 2027, 2032, and 2033. The utility patent family includes a pending patent application, which if granted, could result in a patent expiring in 2033.
Our FemEMB patent portfolio includes one patent family. The patent family includes granted utility patents providing protection until at least 2033. The utility patent family includes a pending patent application, which if granted, could result in a patent expiring in 2033.
Our syringe lock patent portfolio includes two patent families. The patent families include granted design patents, in the U.S. and internationally, providing protection until at least 2038 and 2044, and one pending design application, that if granted, could result in a patent expiring in 2038 and later.
Our Varilock patent portfolio includes two patent families, including pending utility and design patent applications that if granted, could result in a patent expiring in 2048.
Our FemCath patent portfolio includes one patent family, including a design patent that expires in 2026, plus any eligible patent term adjustments and extensions.
There can be no assurance that a pending patent application will be granted. Our material international patents and patent applications include granted design and utility patents, as applicable, with similar overview detail as with the U.S. patent application, including in Canada, China, Hong Kong, European Union, India, Japan, South Korea and Brazil.
The term of individual patents depends upon the legal term for patents in the countries in which they are granted. In most countries, including the United States, the patent term is 20 years from the earliest claimed filing date of a nonprovisional patent application in the applicable country. In the United States, a patent’s term may, in certain cases, be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the United States Patent and Trademark Office in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over a commonly owned patent or a patent naming a common inventor and having an earlier expiration date. We cannot be sure that our pending patent applications that we have filed or may file in the future will result in issued patents, and we can give no assurance that any patents that have issued or might issue in the future will protect our current or future products, will provide us with any competitive advantage, and will not be challenged, invalidated, or circumvented.
For more information regarding the risks related to our intellectual property, please see the section titled “Risk Factors—Risks Related to Our Intellectual Property.”
Government Regulations
United States
Our products are medical devices subject to extensive and ongoing regulation by the FDA under the Federal Food, Drug, and Cosmetic Act (FDCA) and its implementing regulations, as well as other federal and state regulatory bodies in the United States and comparable authorities in other countries under other statutes and regulations. The laws and regulations govern, among other things, product design and development, preclinical and clinical testing, manufacturing, packaging, labeling, storage, recordkeeping and reporting, clearance or approval, marketing, distribution, promotion, import and export and post-marketing surveillance. Failure to comply with applicable requirements may subject a device and/or its manufacturer to a variety of administrative sanctions, such as issuance of warning letters, import detentions, civil monetary penalties and/or judicial sanctions, such as product seizures, injunctions and criminal prosecution.
FDA’s Pre-market Review Requirements
Each medical device we seek to commercially distribute in the United States will require either a prior 510(k) clearance, unless it is exempt, a granted request for de novo classification, or a pre-market approval from the FDA. Medical devices are classified into one of three classes—Class I, Class II or Class III—depending on the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurance of product safety and effectiveness. Class I devices are deemed to be low risk and are subject to the general controls of the FDCA, such as provisions that relate to: adulteration; misbranding; registration and listing; notification, including repair, replacement, or refund; records and reports; and good manufacturing practices. Most Class I devices are classified as exempt from pre-market notification under section 510(k) of the FDCA, and therefore may be commercially distributed without obtaining 510(k) clearance from the FDA. Class II devices are subject to both general controls and special controls, which include performance standards, post market surveillance, patient registries and guidance documents. For most Class II devices, the manufacturer must submit to the FDA a pre-market notification requesting permission to commercially distribute the device. Devices deemed by the FDA to pose the greatest risk, such as life-sustaining, life supporting or implantable devices, or devices deemed not substantially equivalent to a previously cleared 510(k) device, are placed in Class III. A Class III device cannot be marketed in the United States unless the FDA approves the device after submission of a PMA. However, there are some Class III pre-amendment devices for which the FDA has not yet called for a PMA. These devices require a PMA only after the FDA publishes a regulation calling for PMA submissions. Prior to the PMA effective date, the manufacturer must submit a 510(k) pre-market notification and obtain clearance in order to commercially distribute these devices. The FDA can also impose sales, marketing or other restrictions on devices in order to ensure that they are used in a safe and effective manner.
510(k) Clearance Pathway
When a 510(k) clearance is required, we must submit a pre-market notification to the FDA demonstrating that our proposed device is substantially equivalent to a predicate device, which may be a previously cleared and legally marketed 510(k) device or a device that was in commercial distribution before May 28, 1976, or a device that that was de novo classified under section 513(f)(2) of the FDCA. To demonstrate substantial equivalence, the manufacturer must show that the proposed device has the same intended use as the predicate device, and it either has the same technological characteristics, or different technological characteristics and the information in the pre-market notification demonstrates that the device is as safe and effective as the predicate device and does not raise different questions of safety and effectiveness. Demonstrating substantial equivalence requires non-clinical performance data and, in some cases, clinical data. If the FDA determines that the device is not substantially equivalent to a previously cleared device, the FDA will place the device into Class III.
There are three types of 510(k)s: traditional; special; and abbreviated. Special 510(k)s are for devices that are modified by the manufacturer legally authorized to market the device, and where performance data are unnecessary, or if performance data are necessary, well-established methods are available to evaluate the change, and the performance data necessary to support substantially equivalent (SE) can be reviewed in a summary or risk analysis format. Abbreviated 510(k)s are for devices that conform to a recognized standard. The special and abbreviated 510(k)s are intended to streamline review, and the FDA intends to process special 510(k)s within 30 days of receipt. The FDA also recently established the Safety and Performance Based Pathway that is an expansion of the concept of the abbreviated 510(k) pathway for certain, well understood device types, and provides the option to use FDA-identified performance criteria to demonstrate that a device is as safe and effective as a predicate device.
De Novo Classification
Medical device types that the FDA has not previously classified as Class I, II or III are automatically classified into Class III regardless of the level of risk they pose. The Food and Drug Administration Modernization Act of 1997 established a new route to market for low to moderate risk medical devices that are automatically placed into Class III due to the absence of a predicate device, called the “Request for Evaluation of Automatic Class III Designation,” or the de novo classification procedure. This procedure allows a manufacturer whose novel device is automatically classified into Class III to request de novo classification of its medical device into Class I or Class II on the basis that the device presents low or moderate risk, and that general controls alone, or general and special controls, provide reasonable assurance of safety and effectiveness for the intended use and that the probable benefits of the device outweigh the probable risks.
The FDA has issued several guidance documents addressing the de novo classification process and the contents of de novo classification requests, but the FDA has not yet issued regulations governing the de novo classification process. On December 7, 2018, the FDA published a proposed rule to establish regulations for the de novo classification process. The proposed regulations, if finalized, are intended to provide structure, clarity and transparency on the de novo classification process, including requirements related to the format and content of de novo requests, as well as processes and criteria for accepting, granting, declining and withdrawing de novo requests.
Under the Food and Drug Administration Safety and Innovation Act (FDASIA), the FDA is required to issue an order classifying the device within 120 days following receipt of the de novo request, but in practice the time for FDA review of de novo classification requests is significantly longer. Under the Food and Drug Administration Reauthorization Act (FDARA), Congress implemented user fees for de novo classification requests and the FDA committed to performance goals for their review. If the manufacturer seeks de novo classification into Class II, the manufacturer must include a draft proposal for special controls that are necessary to provide a reasonable assurance of the safety and effectiveness of the medical device. In addition, the FDA may decline the de novo classification request if it identifies a legally marketed predicate device, or determines that general controls or general and special controls are insufficient to provide reasonable assurance of safety and effectiveness of the device, or that the probable benefits of the device do not outweigh the probable risks. Devices that are classified into class I or class II in response to a de novo classification request may be marketed and used as predicates for future pre-market notification 510(k) submissions.
Pre-market Approval Pathway
A pre-market approval application must be submitted to the FDA for all Class III devices other than preamendment Class III devices for which the FDA has not yet required a PMA. The pre-market approval application process is much more demanding than the 510(k) pre-market notification process. A pre-market approval application must be supported by extensive data, including but not limited to technical, preclinical and clinical trial data, and manufacturing and labeling information to demonstrate to the FDA’s satisfaction a reasonable assurance of safety and effectiveness of the device.
Within 45 days after submission of a PMA application, the FDA will determine whether the application is sufficiently complete to permit a substantive review and thus whether the FDA will file the application for review. The FDA has a performance goal of issuing a decision on original PMAs that do not require input from an advisory committee within 180 FDA Days, which exclude days during which an agency request for additional information is pending with the applicant. The total time for FDA review of an application generally occurs over a significantly longer period of time and can take a year, or even longer. During this review period, the FDA may request additional information or clarification of the information already provided. Also, an advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations to the FDA as to the approvability of the device. Although the FDA is not bound by the advisory panel decision and may or may not accept the panel’s recommendations, the panel’s recommendations are important to the FDA’s overall decision-making process. In addition, the FDA may conduct a preapproval inspection of the manufacturing facility to ensure compliance with the Quality System Regulation, or QSR. The agency also may inspect one or more clinical sites to assure compliance with the FDA’s regulations.
The FDA allows applicants to submit discrete sections (modules) of the PMA to the FDA for review soon after completing the testing and analysis. The FDA intends the modular review approach to provide a mechanism by which applicants may submit preclinical data and manufacturing information for review while still collecting, compiling, and analyzing the clinical data. Therefore, a modular PMA is a compilation of sections or “modules” submitted at different times that together become a complete application. Additionally, the modular approach allows the applicant to potentially resolve any deficiencies noted by the FDA earlier in the review process than would occur with a traditional PMA application.
During the PMA review, the FDA assesses whether the data and information in the PMA constitute valid scientific evidence to support a determination that there is a reasonable assurance that the device is safe and effective for its intended use(s) based on the proposed labeling. Upon completion of the PMA review, the FDA may: (i) approve the PMA, which authorizes commercial marketing with specific prescribing information for one or more indications, and which can be more limited than those originally sought; (ii) issue an approvable letter, which indicates the FDA’s belief that the PMA is approvable and states what additional information the FDA requires, or the post-approval commitments that must be agreed to prior to approval; (iii) issue a not approvable letter, which outlines steps required for approval, but which are typically more onerous than those in an approvable letter, and may require additional clinical trials that are often expensive and time consuming and can delay approval for months or even years; or (iv) deny the application. If the FDA issues an approvable or not approvable letter, the applicant has 180 days to respond, after which the FDA’s review clock is reset. If the FDA issues a PMA approval, the approval may contain post approval conditions intended to ensure the safety and effectiveness of the device, including, among other things, restrictions on labeling, promotion, sale and distribution, and collection of long-term follow-up data from patients in the clinical trial that supported a PMA or requirements to conduct additional clinical trials post-approval.
Certain changes to an approved device, such as changes in manufacturing facilities, methods, or quality control procedures, or changes in the design performance specifications, which affect the safety or effectiveness of the device, require submission and approval of a PMA supplement. Certain other changes to an approved device require the submission and approval of a new PMA, such as when the design change leads to a different intended use, mode of operation, and technical basis of operation, or when the design change is so significant that a new generation of the device will be developed, and the originally submitted data are not applicable to the change.
Clinical Trials
Clinical trials are almost always required to support pre-market approval, are often required for de novo classification, and are sometimes required for 510(k) clearance. In the United States, for significant risk devices, these trials require submission of an IDE application to the FDA. The IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing it is safe to test the device in humans and that the testing protocol is scientifically sound. The IDE must be approved in advance by the FDA for a specific number of patients at specified study sites. During the trial, the sponsor must comply with the FDA’s IDE requirements for investigator selection, trial monitoring, reporting and recordkeeping. The investigators must obtain patient informed consent, rigorously follow the investigational plan and study protocol, control the disposition of investigational devices and comply with all reporting and recordkeeping requirements. Clinical trials for significant risk devices may not begin until the IDE application is approved by the FDA. An IDE application is considered approved 30 days after it has been received by the FDA, unless the agency otherwise informs the sponsor via email prior to 30 calendar days from the date of receipt, that the IDE is approved, approved with conditions, or disapproved. In addition, the study must be approved by, and conducted under the oversight of an Institutional Review Board (IRB). An IRB is an appropriately constituted group that has been formally designated to review and monitor medical research involving subjects and which has the authority to approve, require modifications in, or disapprove research to protect the rights, safety and welfare of human research subjects. A nonsignificant risk device does not require FDA approval of an IDE; however, the clinical trial must still be conducted in compliance with abbreviated IDE requirements such as monitoring of the investigation, ensuring that the investigators obtain informed consent, and labeling and record-keeping requirements, and be approved by an IRB at the clinical trial sites. The FDA or the IRB at each site at which a clinical trial is being performed may withdraw approval of a clinical trial at any time for various reasons, including a belief that the risks to study subjects outweigh the benefits or a failure to comply with FDA or IRB requirements. Even if a trial is completed, the results of clinical testing may not demonstrate the safety and effectiveness of the device, may be equivocal or may otherwise not be sufficient to obtain approval or clearance of the product.
Sponsors of certain clinical trials of devices are required to register with clinicaltrials.gov, a public database of clinical trial information. Information related to the device, patient population, phase of investigation, study sites and investigators and other aspects of the clinical trial is made public as part of the registration.
Ongoing Regulation by the FDA
Even after a device receives clearance, grant of a de novo classification request or approval and is placed on the market, numerous regulatory requirements apply. These include:
•
establishment registration and device listing;
•
the Quality System Regulation, or QSR, which requires manufacturers, including third-party contract manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the manufacturing process;
•
labeling regulations and the FDA prohibitions against the promotion of products for uncleared or unapproved uses (“off-label” uses) and other requirements related to promotional activities, including the advertising of restricted devices;
•
medical device reporting regulations, which require that manufacturers report to the FDA if their device may have caused or contributed to a death or serious injury, or if their device malfunctioned and the device or a similar device marketed by the manufacturer would be likely to cause or contribute to a death or serious injury if the malfunction were to recur;
•
corrections and removal reporting regulations, which require that manufacturers report to the FDA field corrections or removals if undertaken to reduce a risk to health posed by a device or to remedy a violation of the FDCA that may present a risk to health; and
•
post market surveillance regulations, which apply to certain Class II or III devices when necessary to protect the public health or to provide additional safety and efficacy data for the device.
After a device receives 510(k) clearance or is de novo classified, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, will require a new clearance or possibly even a new de novo classification or PMA supplement. The FDA requires each manufacturer to make this determination initially, but the FDA can review any such decision and can disagree with a manufacturer’s determination. If the FDA disagrees with our determination not to seek a new 510(k) clearance, the FDA may retroactively require us to seek 510(k) clearance or possibly de novo classification or PMA supplement. The FDA could also require us to cease marketing and distribution and/or recall the modified device until 510(k) clearance, de novo classification, or pre-market approval is obtained. Also, in these circumstances, we may be subject to enforcement actions.
Some changes to an approved PMA device, including changes in indications, labeling or manufacturing processes or facilities, require submission and FDA approval of a new PMA or PMA supplement, as appropriate, before the change can be implemented. Supplements to a PMA often require the submission of the same type of information required for an original PMA, except that the supplement is generally limited to that information needed to support the proposed change from the device covered by the original PMA. The FDA uses the same procedures and actions in reviewing PMA supplements as it does in reviewing original PMAs.
FDA regulations require us to register as a medical device manufacturer with the FDA. Additionally, the California Department of Health Services, or CDHS, requires us to register as a medical device manufacturer within the state. Our manufacturing processes are required to comply with the applicable portions of the QSR, which cover the methods and the facilities and controls for the design, manufacture, testing, production, processes, controls, quality assurance, labeling, packaging, distribution, installation and servicing of finished devices intended for human use. As a manufacturer, our facilities, records and manufacturing processes are subject to periodically scheduled or unscheduled inspections by the FDA and the CDHS. Our failure to maintain compliance with the QSR could result in the shutdown of, or restrictions on, our manufacturing operations and the recall or seizure of our products. We have undergone and expect to continue to undergo regular QSR inspections in connection with the manufacture of our products at our facilities. Further, the FDA requires us to comply with various FDA requirements regarding labeling and promotion. Failure by us or by our suppliers to comply with applicable regulatory requirements can result in enforcement action by the FDA or state authorities, which may include any of the following sanctions:
•
warning or untitled letters, fines, injunctions, consent decrees and civil penalties; customer notifications, voluntary or mandatory recall or seizure of our products;
•
operating restrictions, partial suspension or total shutdown of production;
•
delay in processing submissions or applications for new products or modifications to existing products;
•
withdrawing PMA approvals that have already been granted; and
The Medical Device Reporting laws and regulations require us to provide information to the FDA when we receive or otherwise become aware of information that reasonably suggests our device may have caused or contributed to a death or serious injury, or has malfunctioned and the device or a similar device that we market would be likely to cause or contribute to a death or serious injury if the malfunction were to recur. In addition, the FDA prohibits an approved device from being marketed for off-label use. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability, including substantial monetary penalties and criminal prosecution.
Newly discovered or developed safety or effectiveness data may require changes to a product’s labeling, including the addition of new warnings and contraindications, and also may require the implementation of other risk management measures. Also, new government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or prevent regulatory clearance or approval of our products under development.
We are also subject to other federal, state and local laws and regulations relating to safe working conditions, laboratory and manufacturing practices.
European Union
Our products are regulated in the European Union as medical devices under 2017/745 Medical Device Regulation (MDR), which replaced the Medical Devices Directive (MDD) and as of May 26, 2024, the requirements of the new MDR must be complied with by all medical devices regardless of their risk class. The Medical Devices Regulation envisages, among other items, stricter controls of medical devices, including strengthening of the conformity assessment procedures, increased expectations as regards to clinical data for devices and pre-market regulatory review of high-risk devices. Under transitional provisions, medical devices with notified body certificates issued under the Medical Devices Directive prior to May 26, 2021 may continue to be placed on the market for the remaining validity of the certificate, until May 27, 2024 at the latest. After the expiry of any applicable transitional period, only devices that have been CE marked under the Medical Devices Regulation may be placed on the market in the EEA.
The Medical Devices Regulation requires medical devices meet the essential requirements which are enumerated in the Regulation. Compliance with these requirements is a prerequisite to be able to affix the Conformité Européene, or CE, mark to our products, without which they cannot be sold or marketed in the EEA. To demonstrate compliance with the essential requirements we must perform a conformity assessment procedure, which varies according to the type of medical device and its classification.
The European Union’s Medical Device Regulation (MDR) classifies medical devices into four classes and the classification is based on the device’s intended purpose and the risk it poses to patients. Class I is the lowest risk class, for devices used in basic functions and non-invasive procedures; manufacturers can self-assess conformity to requirements. Class IIa is a moderate risk class, for devices like catheters and short-term devices; manufacturers must receive a declaration of conformity from a notified body. Class IIb is a significant risk class, for devices that administer or remove medicinal products from the body. Class III is the highest risk class, for devices that support vital functions, are implanted for long periods, or are used in critical procedures. Notified bodies consult expert panels before issuing a CE certificate. To determine the class of a medical device, we must follow the 22 classification rules in Annex VIII of the MDR.
Depending on the relevant conformity assessment procedure, the notified body would typically audit and examine the technical file and the quality system for the manufacture, design and final inspection of our devices. The notified body issues a certificate of conformity following successful completion of a conformity assessment procedure conducted in relation to the medical device and its manufacturer and their conformity with the essential requirements. This certificate entitles the manufacturer to affix the CE mark to its medical devices after having prepared and signed a related EC Declaration of Conformity.
As a general rule, demonstration of conformity of medical devices and their manufacturers with the essential requirements must be based, among other things, on the evaluation of clinical data supporting the safety and performance of the products during normal conditions of use. Specifically, a manufacturer must demonstrate that the device achieves its intended performance during normal conditions of use, that the known and foreseeable risks, and any adverse events, are minimized and acceptable when weighed against the benefits of its intended performance, and that any claims made about the performance and safety of the device are supported by suitable evidence. Under the MDR, manufacturers are also required to implement post-market surveillance activities, including post-market clinical follow-up (PMCF), to collect and evaluate clinical data on the performance and safety of devices once they are placed on the market. These activities may include the collection of real-world clinical data to confirm the continued safety and performance of the device and to support the ongoing clinical evaluation required under the MDR. To commercialize medical devices in the EU, we must obtain and maintain a CE marking demonstrating compliance with the MDR.
Other Regions
Most major markets have different levels of regulatory requirements for medical devices. Modifications to the cleared or approved products may require a new regulatory submission in all major markets. The regulatory requirements, and the review time, vary significantly from country to country. Products can also be marketed in other countries that have minimal requirements for medical devices.
Fraud and Abuse and Other Healthcare Regulations
Federal and state governmental agencies and equivalent foreign authorities subject the healthcare industry to intense regulatory scrutiny, including heightened civil and criminal enforcement efforts. These laws constrain the sales, marketing and other promotional activities of medical device manufacturers by limiting the kinds of financial arrangements we may have with hospitals, healthcare practitioners and other potential purchasers of our products. Federal healthcare fraud and abuse laws apply to our business when a customer submits a claim for an item or service that is reimbursed under Medicare, Medicaid or other federally-funded healthcare programs. Patient privacy statutes and regulations by foreign, federal and state governments may also apply in the locations in which we do business. Descriptions of some of the U.S. laws and regulations that may affect our ability to operate follow below.
Federal Healthcare Anti-Kickback Statute
The federal healthcare Anti-Kickback Statute prohibits, among other things, persons or entities from knowingly and willfully soliciting, offering, receiving or paying any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchasing, leasing, ordering, or arranging for or recommending the purchase, lease, or order of any good or service for which payment may be made, in whole or in part, by federal healthcare programs, such as the Medicare and Medicaid programs. The term “remuneration” has been broadly interpreted to include anything of value, and the government can establish a violation of the Anti-Kickback Statute without proving that a person or entity had actual knowledge of the law or a specific intent to violate it. In addition, the government may assert that a claim, including items or services resulting from a violation of the Anti-Kickback Statute, constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act. The Anti-Kickback Statute is subject to evolving interpretations and has been applied by government enforcement officials to a number of common business arrangements in the medical device industry. There are a number of statutory exceptions and regulatory safe harbors protecting certain business arrangements from prosecution under the Anti-Kickback Statute; however, those exceptions and safe harbors are drawn narrowly, and there is no exception or safe harbor for many common business activities, such as reimbursement support programs, educational and research grants or charitable donations. The failure of a transaction or arrangement to fit precisely within one or more applicable statutory exceptions or regulatory safe harbors does not necessarily mean that it is illegal or that prosecution will be pursued. However, conduct and business arrangements that do not fully satisfy all requirements of an applicable safe harbor may result in increased scrutiny by government enforcement authorities and will be evaluated on a case-by-case basis based on a cumulative review of all facts and circumstances.
Federal Civil False Claims Act
The federal civil False Claims Act prohibits, among other things, persons or entities from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment of government funds, or knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim to avoid, decrease or conceal an obligation to pay money to the federal government. A claim including items or services resulting from a violation of the Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the False Claims Act. Actions under the False Claims Act may be brought by the government or as a qui tam action by a private individual in the name of the government. These individuals, sometimes known as “relators” or, more commonly, as “whistleblowers,” may share in any amounts paid by the entity to the government in fines or settlement. The number of filings of qui tam actions has increased significantly in recent years. Qui tam actions are filed under seal and impose a mandatory duty on the U.S. Department of Justice to investigate such allegations. Most private citizen actions are declined by the Department of Justice or dismissed by federal courts. However, the investigation costs for a company can be significant and material even if the allegations are without merit. Various states have adopted laws similar to the False Claims Act, and many of these state laws are broader in scope and apply to all payors, and therefore, are not limited to only those claims submitted to the federal government. Medical device manufacturers and other healthcare companies are also subject to federal fraud and abuse laws, including, among others, federal criminal healthcare fraud and false statement statutes that extend to non-government health benefit programs.
Healthcare Fraud Statute
The Health Insurance Portability and Accountability Act (HIPAA), along with related federal criminal statutes (primarily 18 U.S.C §§ 1347 and 1035), enforce regulations prohibiting fraudulent practices within healthcare benefit programs. Such rules and regulations prohibit, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud healthcare benefit programs. This includes intentional falsification, concealment, or covering up of material facts, knowingly making materially false or fraudulent statements, or using false documentation related to healthcare services, items or benefits. Such offenses are prosecuted by the Department of Justice, often in collaboration with other agencies like the Office of Inspector General (“OIG”) of the U.S. Department of Health and Human Services (“HHS”).
Federal Physician Payments Sunshine Act
The federal Physician Payments Sunshine Act requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program to report annually with certain exceptions to CMS information related to payments or other transfers of value made to a healthcare practitioner or teaching hospital, or to a third party at the request of a healthcare practitioner or teaching hospital, and requires applicable manufacturers and group purchasing organizations to report annually to CMS ownership and investment interests held by healthcare practitioners and their immediate family members. Beginning in 2022, applicable manufacturers are also required to report information regarding payments and transfers of value provided to advanced practice providers, including physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists and certified nurse-midwives.
Patient Data Privacy
HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act (HITECH Act), and their implementing regulations impose obligations on covered entities, such as health plans, healthcare clearinghouses and certain healthcare providers, as well as business associates that provide services involving the use or disclosure of personal health information to or on behalf of covered entities. These obligations, such as mandatory contractual terms, relate to safeguarding the privacy and security of protected health information. Many states also have laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA.
Other State Laws
Certain states also mandate implementation of commercial compliance programs, impose restrictions on device manufacturer marketing practices and/or require tracking and reporting of gifts, compensation and other remuneration to healthcare professionals and entities.
State and federal regulatory and enforcement agencies continue to actively investigate violations of healthcare laws and regulations, and the U.S. Congress continues to strengthen the arsenal of enforcement tools. For example, the Bipartisan Budget Act (“BBA”) of 2018 increased the criminal and civil penalties that can be imposed for violating certain federal healthcare laws, including the Anti-Kickback Statute. Enforcement agencies also continue to pursue novel theories of liability under these laws, such as indirect remuneration, off-label promotion, HIPAA noncompliance and violations of the Sunshine Act.
Enforcement and Penalties for Noncompliance with Fraud and Abuse Laws and Regulations
Compliance with these federal and state laws and regulations requires substantial resources. If our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, disgorgement, exclusion from participation in government healthcare programs such as the Medicare and Medicaid programs, reputational harm, administrative burdens, diminished profits and future earnings, and the curtailment or restructuring of our operations. Companies settling federal civil False Claims Act, Anti-Kickback Statute and other fraud and abuse cases also may be required to enter into a Corporate Integrity Agreement with the U.S. Department of Health and Human Services Office of Inspector General in order to avoid exclusion from participation (i.e., loss of coverage for their products) in federal healthcare programs such as Medicare and Medicaid. Corporate Integrity Agreements typically impose substantial costs on companies to ensure compliance.
For additional information regarding obligations under federal healthcare statutes and regulations, please see the section titled “Risk Factors—If we fail to comply with U.S. federal and state fraud and abuse laws and regulations, including those relating to kickbacks and false claims for reimbursement, we could face substantial penalties and our business operations and financial condition could be adversely affected.”
United States Healthcare Reform
There have been and continue to be proposals by the federal government, state governments, regulators and third-party payors to control or manage the increased costs of healthcare and, more generally, to reform the U.S. healthcare system.
For example, in the United States, in March 2010, the ACA was enacted. The ACA contains a number of significant provisions, including those governing enrollment in federal healthcare programs, reimbursement changes and fraud and abuse measures, all of which will impact existing government healthcare programs and will result in the development of new programs.
Certain provisions of the ACA have faced judicial and Congressional challenges, with Congress previously considering, but not passing, comprehensive legislation to repeal or replace the ACA. While Congress has not passed comprehensive repeal legislation, two legislative actions affecting ACA-related taxes have been signed into law. Notably, the Tax Cuts and Jobs Act of 2017 (“Tax Act”), reduced the tax-based shared responsibility payment – commonly referred to as the “individual mandate” – to zero dollars ($0). On December 14, 2018, a federal district court in Texas ruled the individual mandate is a critical and inseverable feature of the ACA, and therefore, because it was repealed as part of the Tax Act, the remaining provisions of the ACA are invalid as well. On December 18, 2019, the Fifth Circuit U.S. Court of Appeals held that the individual mandate is unconstitutional and remanded the case to the lower court to reconsider its earlier invalidation of the full ACA. On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case and held oral arguments on November 10, 2020. In June 2021, the United States Supreme Court held that the individual plaintiffs and states lacked standing to challenge the constitutionality of the ACA. Consequently, this ruling left the ACA intact, fully operational, and legally valid.
On January 22, 2018, President Trump (in his first administration) signed into law a continuing resolution on appropriations for fiscal year 2018, temporarily delaying certain ACA-mandated fees. These fees included the “Cadillac tax” (a tax on certain high-cost employer-sponsored health insurance plans), the annual fee imposed on certain health insurance providers based on market share, and the excise tax on non-exempt medical devices. Subsequently, on December 20, 2019, President Trump (in his first administration) signed the Further Consolidated Appropriations Act (H.R. 1865) into law, permanently repealing the Cadillac tax, the health insurance provider tax, and the medical device excise tax. Other legislative changes impacting the ACA have also been enacted since its passage. For example, the BBA amended the ACA to close the coverage gap, commonly known as the “donut hole,” in most Medicare prescription drug plans.
In December 2018, CMS published a final rule permitting the continued collections of, and payments to, certain ACA qualified health plan and health insurance issuers under the ACA risk adjustment program – addressing issues raised previously in federal district court litigation over CMS’ method for calculating risk adjustment payments. Since that ruling, CMS has updated the payment parameters for the ACA risk adjustment program annually. In addition, CMS published another final rule, that became effective in 2020, which granted states greater flexibility in establishing benchmark plans for insurers in the individual and small group marketplaces. The increased flexibility has ultimately allowed states to offer marketplace plans with variations in coverage, compared to the original mandates of the ACA.
In addition, other legislative changes have been proposed and adopted since the passage of the ACA. For example, the Budget Control Act of 2011, signed into law on August 2, 2011, introduced automatic across-the-board spending reductions, including a 2% annual reduction in Medicare payments to healthcare providers. These payment cuts, known as Medicare sequestration, began on April 1, 2013 and, due to subsequent legislative amendments to the statute, including the BBA, will remain in effect through 2030, unless further Congressional action modifies or ends them. Another example includes the American Taxpayer Relief Act of 2012, signed into law on January 2, 2013, which, among other things, reduced Medicare reimbursement rates to healthcare providers, including hospitals. The Act also extended the statute of limitations for the government to recover overpayments made to providers from three years to five years. Providers now face a longer period during which the government can audit claims and demand repayment of alleged overpayments.
There has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several U.S. Congressional inquiries and proposed, and enacted federal and state legislation designed to bring transparency to product pricing and reduce the cost of products and services under government healthcare programs. Additionally, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what products to purchase and which suppliers will be included in their healthcare programs.
The results of the 2024 presidential and congressional elections, and potential subsequent developments further increase the uncertainty related to the healthcare regulatory environment, particularly given the Trump administration’s stated commitment to significantly reduce government spending through cuts to federal healthcare programs and reductions in the workforces of key government agencies, such as HHS and FDA. In addition, on June 28, 2024, the U.S. Supreme Court issued an opinion holding that courts reviewing agency action pursuant to the Administrative Procedure Act (APA) “must exercise their independent judgment” and “may not defer to an agency interpretation of the law simply because a statute is ambiguous.” The decision will have a significant impact on how lower courts evaluate challenges to agency interpretations of law, including those by FDA and other agencies with significant oversight of the healthcare industry. The new framework is likely to increase both the frequency of such challenges and their odds of success by eliminating one way in which the government previously prevailed in such cases. As a result, significant regulatory policies may be subject to increased litigation and judicial scrutiny. Any resulting changes in regulation may result in unexpected delays, increased costs, or other negative impacts that are difficult to predict but could have a material adverse effect on our business and financial condition.
Employees and Human Capital Resources
As of December 31, 2025, we employed 51 full-time employees and 3 part-time employees. None of our employees are represented by a collective bargaining agreement and we have never experienced a work stoppage. We believe our employee relations are good.
We recognize that attracting, motivating and retaining talent at all levels is vital to our continued success. Our employees are a significant asset and we aim to create an equitable, inclusive and empowering environment in which our employees can grow and advance their careers, with the overall goal of developing, expanding and retaining our workforce to support our current pipeline and future business goals. By focusing on employee retention and engagement, we also improve our ability to support our clinical trials, our pipeline, our platform technologies, business and operations, and also protect the long-term interests of our stakeholders. Our success also depends on our ability to attract, engage and retain a diverse group of employees. Our efforts to recruit and retain a diverse and passionate workforce include providing competitive compensation and benefits packages and ensuring we listen to our employees.
We value innovation, passion, data-driven decision making, persistence and honesty, and are building a diverse environment where our employees can thrive and be inspired to make exceptional contributions to bring novel and proprietary diagnostic and device solutions to disrupt the approaches to women’s health worldwide.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, motivating and integrating our existing and future employees. The principal purposes of our equity incentive plans are to attract, retain and motivate selected employees, consultants and directors through grants of stock-based compensation awards and payments of cash-based performance bonus awards, in order to increase stockholder value and the success of our company by motivating our employees to perform to the best of their abilities and achieve our objectives. We are committed to providing a competitive and comprehensive benefits package to our employees. Our benefits package provides a balance of protection along with the flexibility to meet the individual health and wellness needs of our employees. We plan to continue to refine our efforts related to optimizing our use of human capital as we grow, including improvements in the way we hire, develop, motivate, and retain employees.
Facilities
We produce all of our products and U.S. product candidate in-house at our facility in Suwanee, Georgia which, together with our research and development, controlled environment room and office space, currently totals approximately 41,000 square feet. We believe that our Georgia facility meets our current needs and that suitable additional alternative spaces will be available in the future on commercially reasonable terms.
Legal Proceedings
We are not currently a party to any legal proceedings the outcome of which we believe, if determined adversely to us, would individually or in the aggregate have a material adverse effect on our business, operating results or financial condition. We have received, and may from time to time receive, letters from third parties alleging patent infringement, violation of employment practices or trademark infringement, and we may in the future participate in litigation to defend ourselves. We cannot predict the results of any such disputes, and despite the potential outcomes, the existence thereof may have an adverse material impact on us due to diversion of management time and attention as well as the financial costs related to resolving such disputes.
About Us and Available Information
Femasys Inc. was incorporated in Delaware on February 19, 2004 and is headquartered in Suwanee, Georgia.
We file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a website that contains annual, quarterly and current reports, proxy statements and other information that issuers (including us) file electronically with the SEC. The SEC’s website is www.sec.gov.
Our website is www.femasys.com. We make available, free of charge, through our website: our annual reports on Form 10-K; quarterly reports on Form 10-Q; current reports on Form 8-K; Forms 3, 4 and 5 filed on behalf of directors and executive officers; and any amendments to those reports filed or furnished pursuant to the Securities Exchange Act of 1934, as amended (the “Exchange Act”), as soon as reasonably practicable after such material is electronically filed with, or furnished to, the SEC. We also make available, through our website, our Corporate Governance Guidelines, the charters of the Audit Committee, Nominating and Corporate Governance Committee and the Compensation Committee of our board of directors, our Code of Business Conduct and Ethics and other information and materials. The information on our website is not incorporated by reference into this Form 10-K.
Investing in our common stock involves a high degree of risk. These risks include, but are not limited to, those described below, each of which may be relevant to an investment decision. You should carefully consider the risks described below, together with all of the other information in this Annual Report on Form 10-K, including our financial statements and related notes and Item 7. “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” before deciding to invest in our common stock. The realization of any of these risks could have a significant adverse effect on our reputation, business, financial condition, results of operations and growth, and our ability to accomplish our strategic objectives. In that event, the market price of our common stock could decline, and you may lose part or all of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also impair our business operations and the market value of our common stock.
Risks Related to Our Financial Position and Need for Additional Capital
We have incurred significant operating losses since inception, we expect to incur operating losses in the future and we may not be able to achieve or sustain profitability. We have limited history operating as a commercial company.
We have incurred
net losses since our inception and expect to continue to incur losses for the
foreseeable future. For the years ended December 31, 2025 and December 31,
2024, we had net losses of $18,627,887 and $18,816,628, respectively. As of
December 31, 2025, we had an accumulated deficit of $145,826,144. Based on our
current operating plan, our current cash and cash equivalents, which includes
approximately $0.4 million we raised subsequent to year end, and revenue are
expected to be sufficient to fund our ongoing operations into the third quarter
of 2026. Our estimate as to how long we expect our existing cash and cash
equivalents and revenue to be able to continue to fund our operations is based
on assumptions that may prove to be wrong, and we could use our available
capital resources sooner than we currently expect. Changing circumstances, some
of which may be beyond our control, could cause us to consume capital
significantly faster than we currently anticipate, and we may need to seek
additional funds sooner than planned.
To date, we have financed our operations primarily through our initial public offering, private placements of our common and convertible preferred stock, sales of common stock under an at-the market-agreement and convertible notes and warrants. We have devoted substantially all of our resources to development activities related to our FemBloc system and FemaSeed product, including research and development and clinical and regulatory initiatives.
We expect that our operating expenses will continue to increase as we continue to build our infrastructure, develop, enhance and commercialize new products and incur additional operational costs associated with being a public company. As a result, we expect to continue to incur operating losses for the foreseeable future and may never achieve profitability. Furthermore, even if we do achieve profitability, we may not be able to sustain or increase profitability on an ongoing basis. If we do not achieve or sustain profitability, it will be more difficult for us to finance our business and accomplish our strategic objectives, either of which would have a material adverse effect on our business, financial condition and results of operations and cause the market price of our common stock to decline. Our current cash position and anticipated revenue are not sufficient to fund our operations through U.S. regulatory approval of FemBloc, our primary product candidate. Completing the FINALE pivotal trial and filing a PMA with the FDA will require us to raise substantial additional capital. The amount and timing of that additional capital requirement will depend on, among other things, the pace of enrolment in the pivotal trial, the cost of our research and development activities, and the timing of any FDA advisory feedback. If we are unable to raise sufficient additional capital when needed, whether through equity offerings, debt financing, or strategic collaboration, we may be required to delay, reduce or cease our clinical development activities, which would materially harm our ability to pursue U.S. commercialization of FemBloc. In addition, failure of our FemBloc solution to be approved to market, or to significantly penetrate existing or new markets with our products would negatively affect our business, financial condition, and results of operations.
We need substantial additional funding and may be unable to raise capital when needed, which could force us to delay or reduce our commercialization efforts or product development programs.
Based on our current operating plan, our current cash, cash equivalents and revenue are expected to be sufficient to fund our ongoing operations into the third quarter of 2026. However, we have based these estimates on assumptions that may prove to be incorrect, and we could spend our available financial resources much faster than we currently expect. Any future funding requirements will depend on many factors, including:
•
The initiation, scope, rate of enrollment, progress, success, and cost of our current or future clinical trials;
•
The cost of our research and development activities;
•
Patient, healthcare practitioner and market acceptance of our intratubal insemination product and permanent birth control system women-specific medical product solutions;
•
The cost of filing and prosecuting patent applications and defending and enforcing our patent or other intellectual property rights;
•
The cost of defending, in litigation or otherwise, any claims that we infringe third-party patents or other intellectual property rights;
•
The cost and timing of additional regulatory clearances, de novo grants or approvals;
•
The cost and timing of establishing additional sales and marketing capabilities;
•
Costs associated with any product recall that may occur;
•
The effect of competing technological and market developments;
•
The extent to which we acquire or invest in products, technologies and businesses, although we currently have no commitments or agreements relating to any of these types of transactions; and
•
The costs of operating as a public company.
Any additional equity or debt financing that we raise may contain terms that are not favorable to us or our stockholders. If we raise additional funds by selling additional shares of our common stock or other securities convertible into or exercisable or exchangeable for shares of our common stock, the issuance of such securities will result in dilution to our stockholders. Furthermore, investors purchasing any securities we may issue in the future may have rights superior to the rights of our common stockholders.
Our ability to raise capital through equity offerings is currently subject to significant structural constraints. In July 2025, we filed a shelf registration statement on Form S-3 pursuant to which we may offer and sell up to $150 million in common stock, debt securities, and warrants. However, because our public float is currently below $75.0 million, we are subject to the limitations imposed by Instruction I.B.6 to Form S-3, commonly referred to as the "baby shelf" rule. For so long as our public float remains below $75.0 million, we may not sell more than the equivalent of one-third of our public float in any 12 consecutive months pursuant to a shelf registration statement, including through our at-the-market offering facility with Piper Sandler & Co. In December 2025, we filed a new prospectus pursuant to which we may sell up to $9.8 million under the at-the-market facility, subject to those baby shelf limitations.
While we also have access to additional potential sources of capital (including the Any Market Purchase Agreement with Alumni Capital LP, pursuant to which we may sell up to an aggregate of $10 million in shares of common stock, and the three series of warrants to purchase up to an aggregate of 49,135,689 shares of common stock issued in connection with our November 2025 financing), we cannot guarantee that those facilities will be utilized or sufficient to meet our funding needs. Sales under the at-the-market facility and the Alumni agreement are subject to market conditions and trading volume limitations. Until our public float exceeds $75.0 million, our available shelf capacity will remain constrained, and we may be unable to raise capital quickly or in sufficient amounts to meet our operating requirements. If we are unable to access additional funding on acceptable terms, we may need to delay, significantly modify or cease our clinical and commercial activities.
In addition, any future debt financing into which we enter may impose upon us covenants that restrict our operations, including limitations on our ability to incur liens or additional debt, pay dividends, repurchase our common stock, make certain investments and engage in certain merger, consolidation or asset sale transactions. If we raise additional funds through collaboration and licensing arrangements with third-parties, it may be necessary to relinquish some rights to our technologies or our products, or grant licenses on terms that are not favorable to us.
Furthermore, we cannot be certain that additional funding will be available on acceptable terms, if at all. If we do not have, or are not able to obtain, sufficient funds, we may have to delay development or commercialization of our products or license to third-parties the rights to commercialize products or technologies that we would otherwise seek to commercialize. We also may have to reduce commercialization efforts, customer support or other resources devoted to our products or cease operations. Any of these factors could harm our business, financial condition, and results of operations.
There is substantial doubt about our ability to continue as a going concern.
As a result of our current limited financial liquidity, we have concluded that substantial doubt exists about our ability to continue as a going concern. If we are unable to raise sufficient capital when needed, our business, financial condition and results of operations will be materially and adversely affected, and we will need to significantly modify our operational plans to continue as a going concern. If we are unable to continue as a going concern, we may have to liquidate our assets, and the values we receive for our assets in liquidation or dissolution could be significantly lower than the values reflected in our financial statements. Our lack of cash resources and our potential inability to continue as a going concern may materially adversely affect our share price and our ability to raise new capital, enter into critical contractual relations with third parties and otherwise execute our development strategy.
There are risks associated with our convertible notes and warrants that could adversely affect our business and financial condition.
In November 2025, we issued $12 million senior secured convertible notes, convertible into shares of common stock at a conversion price of $0.73 per share. The convertible notes accrue interest at a rate of 8.5% per annum, payable annually in kind (“PIK”) by increasing the principal balance outstanding. The 2025 Notes mature in November 2035, unless earlier converted or redeemed. There are no assurances that that we will have sufficient funds available to satisfy the payment due under our convertible notes at maturity, or that the holders will elect to convert the convertible notes into shares of our common stock.
The convertible
note purchase agreement provides for standard and customary events of default,
such as our failing to make timely payments and failing to timely comply with
the reporting requirements of the Exchange Act. The convertible notes also
contain customary affirmative and negative covenants, including limitations on
incurrence of indebtedness, acquisition and investment transactions, the
existence of liens, the repayment of indebtedness, the payment of cash in
respect of dividends, distributions or redemptions, and the transfer of assets.
Our cash flow may not be sufficient to allow us to pay the principal and
accrued interest at maturity in November 2035. Our ability to make these
payments depends on a number of factors, including our operating performance,
competitive developments and financial market conditions, all of which are
significantly affected by financial, business, economic, and other factors,
many of which we are not able to control. The level of our indebtedness under
the convertible notes could have other important consequences, including the
following:
• We
may need to use a substantial portion of our cash flow from operations to pay
interest and principal on the convertible notes, which would reduce funds
available to us for other purposes such as product development and operations;
• If
we do not have sufficient cash to pay the principal and accrued interest on the
convertible notes at maturity, we may be unable to refinance such indebtedness
on terms favorable to us or at all;
• Conversion
of the convertible notes could result in significant dilution of our common
stock and cause the market price of our common stock to decline.
Our capital
structure gives rise to a significant and compounding risk of dilution to
existing stockholders. The outstanding principal amount of the 2025 Notes at
issuance is convertible into approximately 16,378,563 shares of common
stock. As noted above, the 2025 Notes accrue PIK interest at a rate of 8.5% per
annum, by increasing the principal balance outstanding annually. This means the
outstanding principal balance on which conversion shares are calculated will
increase by approximately $1,020,000 in the first year, with further
compounding in each subsequent year through maturity in November 2035. Over the
ten-year term of the 2025 Notes, the cumulative effect of PIK interest
compounding could result in the 2025 Notes being convertible into a materially
greater number of shares than the 16,378,563 currently contemplated.
In connection
with the November 2025 financing, we also issued three series of warrants to
purchase an aggregate of 49,135,689 shares of our common stock at exercise
prices ranging from $0.81 to $1.10 per share. These warrants are immediately
exercisable and, in the case of the Series A-1 Warrants, expire ten years from
the date of issuance.
Taken together,
if the 2025 Notes were fully converted at the current conversion price and all
three series of warrants issued in connection with the November 2025 financing
were exercised in full, the aggregate number of shares issuable would be
approximately 65,694,606,
representing a substantial increase from the 59,602,787 shares of common stock
outstanding as of December 31, 2025. This dilution could be further compounded
by: (i) future issuances under our at-the-market offering facility; (ii)
issuances under the Alumni Capital Any Market Purchase Agreement; and (iii) the
exercise of the remaining 26,280,425 warrants outstanding, including pre-funded
warrants, from our prior financings.
Any such dilution
could cause the market price of our common stock to decline significantly.
Investors should be aware that conversion of the 2025 Notes and exercise of the
warrants are entirely at the election of the holders (or in the case of certain
forced conversion and forced exercise provisions, at our election or
automatically upon specified events), and that we cannot predict when or
whether any such events will occur. We cannot assure you that the holders will
not elect to convert or exercise their instruments at times that are
disadvantageous to existing stockholders, including at times when we may be
seeking to raise additional equity capital.
Our financial results may fluctuate significantly and may not fully reflect the underlying performance of our business.
Our quarterly and annual results of operations may vary significantly in the future, and period-to-period comparisons of our operating results may not be meaningful. Accordingly, the results of any one quarter or period should not be relied upon as an indication of future performance. Our quarterly and annual financial results may fluctuate as a result of a variety of factors, many of which are outside our control and, as a result, may not fully reflect the underlying performance of our business. One such factor includes seasonal variations of sales. We may in the future experience higher sales in the fourth quarter as a result of patients having paid their annual insurance deductibles in full, thereby reducing their out-of-pocket costs.
Other factors that may cause fluctuations in our quarterly and annual results include:
•
Patient and healthcare practitioner adoption of our FemBloc system, if approved to market in the U.S.;
•
Patient and healthcare practitioner adoption of our FemaSeed product in the U.S.;
•
Changes in coverage policies by third-party payors that affect the U.S. reimbursement of procedures using our products;
•
Unanticipated pricing pressure;
•
The hiring, retention and continued productivity of sales representatives for domestic commercial team;
•
Our ability to expand the geographic reach of our sales and marketing efforts;
•
Our ability to obtain regulatory clearance or approval for any products in development or for our current products for additional indications or in additional countries outside the United States;
•
Results of clinical research and trials on our existing products and products in development;
•
Delays in receipt of anticipated purchase orders;
•
Delays in, or failure of, component and raw material deliveries by our suppliers; and
•
Positive or negative coverage in the media or clinical publications of our products or products of our competitors or our industry.
Because our quarterly and annual results may fluctuate, period-to-period comparisons may not be the best indication of the underlying results of our business and should only be relied upon as one factor in determining how our business is performing. These fluctuations may also increase the likelihood that we will not meet our forecasted performance, which could negatively affect the market price for our common stock.
We have received deficiency notices from Nasdaq relating to our non-compliance with Nasdaq's continued listing requirements and our common stock could become subject to delisting from Nasdaq if we fail to regain compliance.
On May 19, 2025, we received a written notice from Nasdaq that for the preceding 30 consecutive business days, the Market Value of Listed Securities ("MVLS") for our common stock was below the minimum $35.0 million requirement for continued listing on The Nasdaq Capital Market pursuant to Nasdaq Listing Rule 5550(b)(2). We also did not meet either of the alternative continued listing standards under Nasdaq Listing Rule 5550(b): (i) stockholders' equity of at least $2.5 million or (ii) net income of $500,000 in the most recently completed fiscal year, or in two of the three most recently completed fiscal years.
Following the completion of our August 2025 financing and subsequent warrant exercises, we determined that our stockholders' equity as of October 15, 2025 would have been approximately $5.2 million on a pro forma basis, meeting the minimum $2.5 million stockholders' equity requirement for continued listing under Nasdaq Listing Rule 5550(b)(1). As reported on our Form 8-K filed October 21, 2025, Nasdaq determined that we comply with Nasdaq Listing Rule 5550(b)(1). However, if we fail to evidence continued compliance upon filing of this Annual Report on Form 10-K or any subsequent periodic report, we may again be subject to a deficiency determination and potential delisting proceedings.
On July 16, 2025, we received a notice from Nasdaq that our common stock had failed to maintain a minimum closing bid price of $1.00 per share for 30 consecutive business days and that we therefore did not comply with Nasdaq Listing Rule 5550(a)(2) (the "Minimum Bid Price Requirement"). In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we had 180 calendar days, or until January 12, 2026, to regain compliance. We did not regain compliance during that initial period.
On January 12, 2026, we submitted a request to Nasdaq for a 180-day extension, advising Nasdaq that we had demonstrated more than $5 million in stockholders' equity as of December 31, 2025 and providing notice of our intention to cure the deficiency during the extended compliance period by effecting a reverse stock split, if necessary. On January 13, 2026, Nasdaq granted us a second 180-day extension through July 13, 2026 to regain compliance with the Minimum Bid Price Requirement. This is the final compliance period available to us under Nasdaq's rules. If we do not regain compliance by July 13, 2026, our common stock will become subject to delisting. In the event we receive notice that our common stock is being delisted, Nasdaq's rules permit us to appeal that determination to a hearings panel, although no assurance can be given that any such appeal would be successful. We intend to continue actively monitoring the closing bid price of our common stock and will consider all available options to regain compliance, including effecting a reverse stock split. However, there can be no assurance that we will be able to regain compliance with the Minimum Bid Price Requirement by July 13, 2026, maintain compliance with the stockholders' equity requirement, or remain in compliance with other Nasdaq listing rules. If our common stock is delisted, it could have a material adverse effect on the price and liquidity of our common stock and our ability to raise additional capital.
Risks Related to Discovery and Development
Enrollment and retention of subjects in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible by multiple factors outside our control.
We may encounter delays or difficulties in enrolling, or be unable to enroll, a sufficient number of subjects to complete our clinical trial on our current timelines, or at all, and even once enrolled, we may be unable to retain a sufficient number of subjects to complete our trial. For example, as a result of the COVID-19 pandemic, we had slower than expected site initiation and subject enrollment for one of our clinical trials due to subject and staff rescheduling, lack of available site staff and turnover and longer timelines to train staff at new sites. Slow site initiation and subject enrollment in this clinical trial led to delays in our development timelines and may cause further delays in the future.
Subject enrollment in clinical trials and completion of subject follow-up depend on many factors, including the size of the subject population, the nature of the trial protocol, the proximity of subjects to clinical sites, the eligibility criteria for the clinical trial, subject compliance, competing clinical trials and clinicians’ and ’subjects’ perceptions as to the potential advantages of the product being studied in relation to other available therapies, including any new treatments that may be approved for the indications we are investigating. For example, subjects may be discouraged from enrolling in our clinical trial if the trial protocol requires them to undergo extensive post-treatment procedures or follow-up to assess the safety and effectiveness of a product candidate, or they may be persuaded to participate in contemporaneous clinical trials of a competitor’s product candidate. In addition, patients participating in our clinical trial may drop out before completion of the trial or experience adverse medical events unrelated to our products. Delays in subject enrollment or failure of subjects to continue to participate in a clinical trial may delay commencement or completion of the clinical trial, cause an increase in the costs of the clinical trial and delays, or result in the failure of the clinical trial.
Delays or failures in planned site initiation and/or subject enrollment or retention may result in increased costs, program delays or both, which could have a harmful effect on our ability to develop our U.S. product candidate or could render further development impossible. In addition, we rely on clinical trial sites to ensure timely conduct of our clinical trials and, while we have entered into agreements governing their services, we are limited in our ability to compel their actual performance.
Our current U.S. product candidate, FemBloc is in late-stage of development. Our U.S. product candidate may fail in development or suffer delays that adversely affect its commercial viability. If we fail to obtain or maintain FDA approval to market and sell our FemBloc our business will be materially harmed.
The process of seeking regulatory approval to market a medical device is expensive and time consuming. There can be no assurance that approval will be granted. If we are not successful in obtaining timely approval of our FemBloc system, we may never be able to generate significant revenue and may be forced to cease operations. The FDA approval process requires an applicant to demonstrate the safety and effectiveness based, in part, on extensive data, including, but not limited to, technical, preclinical, clinical trial, manufacturing and labeling data. The FDA can delay, limit or deny approval of a device for many reasons, including:
•
We may not be able to demonstrate to the FDA’s satisfaction that our product is safe and effective for its intended use;
•
The FDA may disagree that our clinical data supports the label and use that we are seeking;
•
The FDA may disagree that the data from our preclinical studies and clinical trials is sufficient to support marketing authorization; and
•
The manufacturing process and facilities we use may not meet applicable requirements.
Obtaining approval from the FDA or any foreign regulatory authority could result in unexpected and significant costs for us and consume management’s time and other resources. The FDA could ask us to supplement our submissions, collect additional non-clinical data, conduct additional clinical trials, prepare additional manufacturing data or information or engage in other time-consuming actions, or it could simply deny our applications. In addition, if approved, we will be required to obtain additional FDA approvals prior to making certain modification to our devices, and the FDA may revoke the approval or impose other restrictions if post-market data demonstrates safety issues or lack of effectiveness. If we are unable to obtain and maintain the necessary regulatory approvals to market our product, our financial condition may be adversely affected, and our ability to grow domestically and internationally would likely be limited. Additionally, even if approved, FemBloc may not be approved for the indications that are necessary or desirable for successful commercialization or profitability.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In February 2026, we announced MDSAP certification for FemBloc.
As we evolve from a company that is primarily involved in clinical development to a company that is also involved in commercialization, we may encounter difficulties in expanding our operations successfully.
With the FDA clearance and global regulatory approvals of FemaSeed, we needed to expand our development, regulatory, manufacturing, and marketing and sales capabilities and contract with third parties to provide these capabilities, such as collaborators, distributors, marketers and additional suppliers. We currently have limited experience as a company in our infrastructure for sales, marketing and distribution, and our operations have historically been limited primarily to clinical development activities.
We have established a direct sales organization with technical expertise in supporting commercial capabilities to market FemaSeed in the U.S. This is expensive and time-consuming. In addition, we may not be able to hire a sales force that is sufficient in size or has adequate expertise in the medical markets that we target. Any failure or delay in the development of our internal sales, marketing and distribution capabilities would adversely affect the commercialization of FemaSeed and other products and our U.S. product candidate.
Maintaining third-party relationships for these purposes will impose significant added responsibilities on members of our management and other personnel. We must be able to effectively manage our development efforts, recruit and train sales and marketing personnel, effectively manage our participation in the clinical studies in which our U.S. product candidate and any future product candidates are involved and improve our managerial, development, operational and finance systems, all of which may impose a strain on our administrative and operational infrastructure. If we do not establish sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our products.
We are significantly dependent on the FDA’s permission to market our FemBloc system, as well as market acceptance in the United States for it, and our failure to receive FDA authorization to market the FemBloc system or the failure of it to gain such market acceptance would negatively impact our business.
Since our inception, we have devoted significant efforts to the development of our intrauterine delivery technology that is the basis for our FemBloc system. We have not yet received authorization from the FDA to market and sell the FemBloc system in the United States. However, we will incur costs, including costs to build our sales force for commercialization of our other products, in anticipation of potential FDA authorization to market FemBloc. Our FDA-cleared FemaSeed product are different than what we anticipate for our FemBloc system, the sales force we are currently building for our FemaSeed product primarily targets gynecologists to perform infertility evaluation and treatment, followed by infertility specialists. These same gynecologists are expected to represent the primary target providers for the FemBloc system, if approved. As a result, we expect to continue expanding the sales organization currently supporting FemaSeed to address the broader gynecology market and support commercialization of both products. If FemBloc is authorized for marketing, we will need to maintain and support commercialization activities for multiple products simultaneously.
If we are unable to obtain authorization from the FDA to market and sell this system in the United States, or if the product fails to achieve significant market acceptance in the United States, our results of operations could be adversely affected as the United States is expected to represent a principal market for this product. Further, because we are incurring costs prospectively in advance of FDA authorization, we may be unable to recoup these investments if the U.S. product candidate is not authorized for marketing by the FDA. While we currently market other commercial products, their revenues remain limited. Accordingly, if we are unsuccessful in commercializing the FemBloc system or are unable to market the FemBloc system as a result of a quality problem, failure to maintain or obtain regulatory marketing authorizations, unexpected or serious complications or other unforeseen negative effects related to this system or the other factors discussed in these risk factors, we would lose an additional source of revenue, and our business will be materially adversely affected.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In February 2026, we announced MDSAP certification for FemBloc.
The clinical development process required to obtain regulatory approvals is lengthy and expensive with uncertain outcomes, and our data developed in those clinical trials is subject to interpretation by FDA and foreign regulatory authorities. If clinical trials of our FemBloc system and future products do not produce results necessary to support regulatory approval, a granted de novo classification or clearance in the United States or, with respect to our current or future product candidates, elsewhere, we will be unable to commercialize these products and may incur additional costs or experience delays in completing, or ultimately be unable to complete, the commercialization of those products.
We are currently seeking PMA approval for our permanent birth control solution. In order to obtain PMA approval for the FemBloc system, we must conduct a well-controlled clinical trial designed to assess the safety and effectiveness of the U.S. product candidate. Conducting clinical trials is a complex and expensive process, can take many years, and outcomes are inherently uncertain. We incur substantial expense for, and devote significant time to, clinical trials but cannot be certain that the trials will ever result in commercial revenue. We may experience significant setbacks in the clinical trial, even after earlier clinical trials showed promising results, and failure can occur at any time during the clinical development process. Any of our products may malfunction or may produce undesirable adverse effects that could cause us, institutional review boards or IRBs, or regulatory authorities to interrupt, delay or halt clinical trials. We, IRBs, the FDA, or another regulatory authority may suspend or terminate the clinical trial at any time to avoid exposing trial participants to unacceptable health risks.
Successful results of preclinical studies are not necessarily indicative of future clinical trial results, and predecessor clinical trials results may not be replicated in subsequent clinical trials. Moreover, interim results or topline results may be subject to change upon full review of the data from a clinical trial. Additionally, the FDA’s approval of an IDE application permits initiation of the clinical study described in the IDE application but does not mean that FDA agrees that the study design is appropriate or that the results of the study will be sufficient to obtain marketing authorization (i.e., PMA approval, 510(k) clearance, or grant of a de novo request). The FDA may disagree with our interpretation of the data from our preclinical studies and clinical trials, or may find the clinical trial design, conduct or results inadequate to prove safety or effectiveness, and may require us to pursue additional preclinical studies or clinical trials, which could further delay the clearance, de novo classification, or approval of our products. The data we collect from our preclinical studies and clinical trials may not be sufficient to support FDA approval, a request for de novo classification, or clearance, and if we are unable to demonstrate the safety and effectiveness of our future products in our clinical trials, we will be unable to obtain regulatory approval, a granted de novo classification, or clearance to market our products.
In addition, we may estimate and publicly announce the anticipated timing of the accomplishment of various clinical, regulatory and other product development goals, which are often referred to as milestones. These milestones could include the submission to the FDA of an IDE application to commence a clinical trial for a new product candidate; the enrollment of patients in clinical trials; the release of data from clinical trials; regulatory submissions and decisions, compliance with regulatory requirements in jurisdictions where we market our products, including the European Union, and other clinical and regulatory events; and the obtainment of the right to affix the CE mark in the European Union. The actual timing of these milestones could vary dramatically compared to our estimates, in some cases for reasons beyond our control. We cannot assure you that we will meet our projected milestones and if we do not meet these milestones as publicly announced, the commercialization of our products may be delayed and, as a result, our stock price may decline.
Clinical trials are necessary to support PMA applications, certain de novo classification requests, and certain 510(k) pre-market notifications and may be necessary to support PMA supplements or subsequent 510(k) submissions for modified versions of our marketed devices. This would require the enrollment of large numbers of suitable subjects, which may be difficult to identify, recruit and maintain as participants in the clinical trial. The earlier clinical studies for FemBloc involved 228 subjects and supported the IDE for the new pivotal trial, which will be the basis for the PMA application for our FemBloc system. Adverse outcomes in the IDE approved pivotal trial or post-approval studies could also result in restrictions or withdrawal of approval of the PMA. We will likely need to conduct additional clinical trials in the future for the approval of the use of our products in some foreign countries. Clinical testing is difficult to design and implement, can take many years, can be expensive and carries uncertain outcomes. The initiation and completion of any of these studies may be prevented, delayed, or halted for numerous reasons. We may experience a number of events during the conduct of our clinical trials that could adversely affect the costs, timing or successful completion, including:
•
We are required to submit an IDE application to the FDA, which must become effective prior to commencing human clinical trials, and the FDA may reject our IDE application and notify us that we may not begin investigational trials;
•
Regulators and other comparable foreign regulatory authorities may disagree as to the design or implementation of our clinical trials;
•
Regulators and/or IRBs or other reviewing bodies may not authorize us or our investigators to commence a clinical trial, or to conduct or continue a clinical trial at a prospective or specific trial site;
•
We may not reach agreement on acceptable terms with prospective contract research organizations, or CROs, and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites;
•
Clinical trials may produce negative or inconclusive results, or we may not agree with regulatory authorities on the interpretation of our clinical trial results, and we may decide, or regulators may require us, to conduct additional clinical trials or abandon product development programs;
•
The number of subjects or patients required for clinical trials may be larger than we anticipate, enrollment in these clinical trials may be insufficient or slower than we anticipate, and the number of clinical trials being conducted at any given time may be high and result in fewer available patients for any given clinical trial, or patients may drop out of these clinical trials at a higher rate than we anticipate;
•
Our third-party contractors, may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all;
•
We might have to suspend or terminate clinical trials for various reasons, including a finding that the subjects are being exposed to unacceptable health risks;
•
We may have to amend clinical trial protocols or conduct additional studies to reflect changes in regulatory requirements or guidance, which we may be required to submit to an IRB and/or regulatory authorities for reexamination;
•
Regulators, IRBs, or other parties may require or recommend that we or our investigators suspend or terminate clinical research for various reasons, including safety signals or noncompliance with regulatory requirements;
•
The cost of clinical trials may be greater than we anticipate;
•
Clinical sites may not adhere to the clinical protocol or may drop out of a clinical trial;
•
We may be unable to recruit a sufficient number of clinical trial sites or trial subjects;
•
Regulators, IRBs, or other reviewing bodies may fail to approve or subsequently find fault with our manufacturing processes for clinical and commercial supplies, the supply of devices or other materials necessary to conduct clinical trials may be insufficient, inadequate or not available at an acceptable cost, or we may experience interruptions in supply;
•
Approval policies or regulations of FDA or applicable foreign regulatory authorities may change in a manner rendering our clinical data insufficient for approval; and
•
Our current or future products may have undesirable side effects or other unexpected characteristics.
Clinical trials must be conducted in accordance with the laws and regulations of the FDA and other applicable regulatory authorities’ legal requirements, regulations or guidelines, and are subject to oversight by these governmental agencies and IRBs at the medical institutions where the clinical trials are conducted. We have in the past and may in the future have to terminate a clinical trial site which is found through our clinical trial monitoring activities to be noncompliant with our clinical trial protocols or with applicable laws, regulations, requirements and guidelines for the conduct of our clinical trials.
In addition, clinical trials must be conducted with supplies of our devices produced in conformance with design control requirements in 21 CFR § 820.30 and stored and used by clinical trial sites in accordance with our clinical trial protocols. Furthermore, we rely on clinical trial sites to ensure the proper and timely conduct of our clinical trials and while we have agreements governing their committed activities, we have limited influence over their actual performance. We depend on our CROs to support the conduct of our clinical trials in compliance with good clinical practice, or GCP, requirements. To the extent our CROs fail to help oversee the conduct of the study in compliance with GCP standards or are delayed for a significant time in the execution of the trial, including achieving full enrollment, we may be affected by increased costs, program delays or both. In addition, clinical trials that are conducted in countries outside the United States may subject us to further delays and expenses as a result of increased shipment costs, additional regulatory requirements and the engagement of non-U.S. CROs, as well as expose us to risks associated with clinical investigators who are unknown to the FDA, and different standards of diagnosis, screening and medical care.
Failure can occur at any stage of clinical testing. Our clinical trials may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical and non-clinical testing in addition to those we have planned. Our failure to adequately demonstrate the safety and effectiveness of our systems or any product we may develop in the future would prevent receipt of regulatory approval, a granted de novo classification, or 510(k) clearance and, ultimately, the commercialization of that product or indication for use. Even if our future products are approved, de novo classified, or cleared in the United States, commercialization of our products in foreign countries would require approval by regulatory authorities in those countries. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from, and greater than, those in the United States, including additional preclinical studies or clinical trials. Any of these occurrences could have an adverse effect on our business, financial condition and results of operations.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In February 2026, we announced MDSAP certification for FemBloc.
Interim, “topline,” and preliminary data from our clinical trials that we announce or publish from time to time may change as more data become available and are subject to confirmation, audit, and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary or topline data from our preclinical studies and clinical trials, which is based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study or trial. We also make assumptions, estimations, calculations, and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the topline or preliminary results that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data has been received and fully evaluated. Topline data also remains subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline data should be viewed with caution until the final data is available. From time to time, we may also disclose interim data from our clinical trials. Interim or preliminary data from clinical trials are subject to the risk that one or more of the clinical outcomes may materially change as subject enrollment and treatment continues, and more patient data become available or as subjects from our clinical trials continue other treatments for their disease. Adverse differences between preliminary or interim data and final data could significantly harm our business prospects. Further, disclosure of interim data by us or by our competitors could result in volatility in the price of our common stock.
Further, others, including regulatory agencies, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently, which could impact the potential of the particular program, the likelihood of marketing approval, grant, clearance or commercialization of the particular product candidate, any marketed product, and our company in general. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is derived from information that is typically extensive, and you or others may not agree with what we determine is material or otherwise appropriate information to include in our disclosure.
If the interim, topline, or preliminary data that we report differ from actual results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain approval for, and commercialize, our product candidates may be harmed, which could harm our business, operating results, prospects or financial condition.
If patients or healthcare practitioners are not willing to change current practices to adopt our permanent birth control solution or artificial insemination solution, our products may fail to gain increased market acceptance, and our business will be adversely affected.
Our primary strategy to grow our revenue is to drive the adoption of our permanent birth control using the FemBloc system with an ultrasound confirmatory test using FemChec, our artificial insemination solution using the FemaSeed product and companion diagnostic FemVue, and for healthcare practitioners to employ our products to treat or diagnosis their patients with reproductive disorders or cancers. Healthcare practitioners may choose not to adopt our products for women’s healthcare for a number of reasons, including:
•
lack of availability of adequate third-party payor coverage or reimbursement;
•
lack of experience with our products and more familiarity with other widely adopted products, procedures or treatments as alternatives;
•
our inability to convince key opinion leaders to provide recommendations regarding our products, or to convince healthcare practitioners, patients and healthcare payors that our products are an attractive alternative to currently accepted alternatives;
•
perceived inadequacy of evidence supporting clinical benefits, safety or cost-effectiveness of our products’ existing alternatives;
•
liability risks generally associated with the use of new products and procedures; and
•
the training required to use new products.
With respect to FemBloc, we intend to focus our sales, marketing and training efforts primarily on obstetrical and gynecological physicians. However, healthcare practitioners from other disciplines, including primary care physicians, as well as other medical professionals, such as nurse practitioners and physician assistants, are often the initial point of contact for patients with contraceptive needs. We believe that educating healthcare practitioners in these disciplines and other medical professionals about the clinical merits, patient benefits and safety profile of our permanent birth control solution is an element of increasing the adoption of our FemBloc system. If additional healthcare practitioners or other medical professionals do not appreciate and recommend our permanent birth control solution for any reason, including those listed above, our ability to execute our growth strategy will be impaired, and our business may be adversely affected.
In addition, patients may not be able to adopt or may choose not to adopt our permanent birth control solution if, among other potential reasons, their anatomy would not allow for effective treatment with our FemBloc system, they are reluctant to receive a permanent solution to their contraceptive needs, they are worried about potential adverse effects of our permanent birth control solution, such as infection or discomfort, or they are unable to obtain adequate third-party coverage or reimbursement.
With respect to FemaSeed, we intend to focus our sales, marketing and training efforts initially on reproductive endocrinologist physicians with expansion to gynecologists who are often the initial point of contact for patients with infertility needs with the availability of FemSperm. We believe that educating healthcare practitioners in these disciplines and other medical professionals about the clinical merits and patient benefits of our artificial insemination solution is an element of increasing the adoption of our FemaSeed product. If additional healthcare practitioners or other medical professionals do not appreciate and recommend our FemaSeed product for any reason, including those listed above, our ability to execute our growth strategy will be impaired, and our business may be adversely affected.
If we are unable to achieve and maintain adequate levels of coverage or reimbursement for our permanent birth control solution or any other products we seek to commercialize, our commercial success may be severely hindered.
The primary customers for our products are gynecological physicians, related healthcare professionals, and women’s healthcare provider organizations. Our customers typically bill various third-party payors to cover all, or a portion, of the costs and fees associated with the procedures in which our products are used and bill patients for any deductibles or co-payments. Limited third-party payors provide infertility coverage with patient cash pay as often required for treatment and similar services. Under the Patient Protection and Affordable Care Act, as amended by the Health Care and Education and Reconciliation Act, most private health insurance plans are required to cover contraceptive-related procedures. If there are changes to the ACA related to contraceptive coverage, any reduction in the reimbursements our customers receive could make it more difficult for them to choose, or adopt, our FemBloc system and could create additional pricing pressure for us. If we are forced to lower the price we charge for our product, our gross margins will decrease, which could have a material adverse effect on our business, financial condition and results of operations and impair our ability to grow our business.
Third-party payors, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, no uniform policy of coverage and reimbursement for procedures using our other products exists among third-party payors. Therefore, coverage and reimbursement for procedures using our other products can differ significantly from payor to payor. Payors continually review new and existing technologies for possible coverage and can, without notice, deny or reverse coverage for new or existing products and procedures. There can be no assurance that third-party payor policies will provide coverage for procedures in which our products are used. If we are not successful in reversing existing non-coverage policies, or if third-party payors that currently cover or reimburse our products and related procedures reverse or limit their coverage in the future, or if other third-party payors issue similar policies, this could have a material adverse effect on our business.
Further, we believe that future coverage and reimbursement may be subject to increased restrictions, such as additional prior authorization requirements, both in the United States and in international markets. Third-party coverage and reimbursement for procedures using our products or any of our products in development for which we may receive regulatory approval may not be available or adequate in either the United States or international markets, which could have an adverse effect on our business, financial condition and results of operations and impair our ability to grow our business.
Third-party payors and healthcare practitioners who do not cover or use our permanent birth control solution or other women’s healthcare devices may require additional clinical data prior to adopting or maintaining coverage of our products.
Our success depends on healthcare practitioners and where applicable third-party payor acceptance of our permanent birth control solution as effective treatment option and our other healthcare devices for women. If healthcare practitioners or payors do not find our body of published clinical evidence and data compelling or wish to wait for additional studies, they may choose not to use or provide coverage and reimbursement for our products. Currently, infertility treatments often require significant out-of-pocket payments. However, most insurance plans will cover permanent birth control solutions without cost-sharing under the ACA.
Certain healthcare practitioners, hospitals and payors may prefer to see longer-term safety and efficacy data for our permanent birth control solution than we have produced. We cannot provide assurance that any data that we or others may generate in the future will be consistent with that observed in our existing clinical trials.
The training required for healthcare practitioners to use our artificial insemination solution and permanent birth control solution could reduce the market acceptance of our products.
As with any new method or technique, healthcare practitioners must undergo a thorough training program before they perform the procedure. Even after successfully completing the training program, healthcare practitioners could still experience difficulty in successfully providing the solutions and, as a result, limit use of the products significantly in their practice or cease utilizing it altogether.
In addition, we may experience difficulty growing the number of healthcare practitioners who complete our training program if patient demand is low, if the length of time necessary to train each healthcare practitioner is longer than expected, if the capacity of our sales representatives to train healthcare practitioners is less than expected or if we are unable to sufficiently grow our sales organization. All of these events would lead to fewer trained healthcare practitioners to provide our solutions, which could negatively affect our business, financial condition and results of operations and impair our ability to grow our business.
We currently compete and will in the future continue to compete against other companies, some of which have longer operating histories, more established products or greater resources than we do, which may prevent us from achieving increased market penetration and improved operating results.
The biomedical industry is highly competitive, subject to change and significantly affected by new product introductions and other activities of industry participants. Our competitors have historically dedicated, and will continue to dedicate significant resources to promoting their products or developing new products or methods to treat women’s reproductive issues and healthcare. We consider our primary potential competition to be other biomedical companies marketing women-specific medical products. Having received FDA clearance for FemaSeed, we are the only localized directional intratubal insemination approach within the intrauterine insemination category approved for commercialization in North America, but compete with other fertility treatments such as traditional IUI and IVF. For our other FDA-cleared devices, we currently compete with other medical device providers in the United States, Europe and Canada. Once we have received FDA approval, we will be the only non-surgical permanent birth control solution approved for commercialization. We also believe other emerging businesses may be in the early stages of developing women-specific medical products. If one or more manufacturers successfully develops a product for providing localized directional intratubal insemination that is more effective or otherwise more attractive than our artificial insemination solution, sales of our FemaSeed product could be significantly and adversely affected, which could have a material adverse effect on our business, financial condition and results of operations. In addition, if other companies are successful in developing products that are approved for a broader range of indications than our artificial intratubal insemination system, we will be at a further competitive disadvantage, which could also affect our business, financial condition and results of operations. If one or more manufacturers successfully develops a product for providing permanent birth control that is more effective, better tolerated or otherwise results in better compliance by patients, or otherwise more attractive than our permanent birth control solution, sales of our FemBloc system could be significantly and adversely affected, which could have a material adverse effect on our business, financial condition and results of operations. In addition, if other companies are successful in developing devices that are approved for a broader range of indications than our permanent birth control system, we will be at a further competitive disadvantage, which could also affect our business, financial condition and results of operations.
Many of the companies against which we may compete may have competitive advantages with respect to primary competitive factors in the women’s healthcare market, including:
•
greater company, product and brand recognition;
•
superior product safety, reliability and durability;
•
better quality and larger volume of clinical data;
•
more effective marketing to and education of patients and healthcare practitioners;
•
more sales force experience and greater market access;
•
better product support and service;
•
more advanced technological innovation, product enhancements and speed of innovation;
•
more effective pricing and revenue strategies;
•
lower procedure costs to patients;
•
more effective reimbursement teams and strategies;
•
dedicated practice development; and
•
more effective clinical training teams.
We also compete with other biomedical companies to recruit and retain qualified sales, training and other personnel.
In addition, though there are currently no pharmacologic therapies approved to provide permanent birth control, we may in the future face competition from pharmaceutical companies that develop such therapies. We also expect to experience increased competition in the future as other companies develop and commercialize competing women specific devices. Any of these companies may also have the competitive advantages described above.
Our long-term growth depends on our ability to enhance our artificial insemination solution, permanent birth control solution and women-specific medical product solutions, expand our indications and develop and commercialize additional products.
It is important to our business that we continue to enhance our artificial insemination product, permanent birth control system women-specific medical product solutions and develop and introduce new products. Developing products is expensive and time-consuming and could divert management’s attention away from our core business. The success of any new product offering or product enhancements will depend on several factors, including our ability to:
•
properly identify and anticipate healthcare practitioner and patient needs;
•
develop and introduce new products and product enhancements in a timely manner;
•
avoid infringing upon the intellectual property rights of third-parties;
•
demonstrate, if required, the safety and effectiveness of new products with data from preclinical studies and clinical trials;
•
obtain the necessary regulatory clearances, grants or approvals for expanded indications, new products or product modifications;
•
be fully FDA-compliant with marketing of new products or modified products;
•
provide adequate training to potential users of our products;
•
receive adequate coverage and reimbursement for procedures performed with our products; and
•
develop an effective and dedicated sales and marketing team.
If we are not successful in expanding our indications and developing and commercializing new products and product enhancements, our ability to increase our revenue may be impaired, which could have a material adverse effect on our business, financial condition and results of operations.
Our results of operations could be materially harmed if we are unable to accurately forecast customer demand for our artificial insemination solution, permanent birth control solution, and women-specific medical products and manage our inventory.
To ensure adequate inventory supply, we must forecast inventory needs and place orders with our suppliers based on our estimates of future demand for our products. Our ability to accurately forecast demand for our products could be negatively affected by many factors, including our failure to accurately manage our expansion strategy, product introductions by competitors, an increase or decrease in customer demand for our artificial insemination product, permanent birth control system, and women-specific medical products or for products of our competitors, our failure to accurately forecast customer acceptance of new products, unanticipated changes in general market conditions or regulatory matters and weakening of economic conditions or consumer confidence in future economic conditions. For example, tubal ligation procedures sustained an 18% decline in December 2020 compared to December 2019, according to a study published in the publication Contraception in 2021. Although we have no assurance that demand for elective reproductive surgery will return to pre-pandemic levels in the future, or at all, Martinez (2024) reported 1.2million women underwent surgical tubal ligation, an increase from previous estimates of 800,000 annually. Inventory levels in excess of customer demand may result in inventory write-downs or write-offs, which would cause our gross margin to be adversely affected and could impair the strength of our brand. Conversely, if we underestimate customer demand for our products, our third-party suppliers may not be able to deliver components to meet our requirements, and this could result in damage to our reputation and customer relationships. In addition, if we experience a significant increase in demand, additional supplies of raw materials or additional manufacturing capacity may not be available when required on terms that are acceptable to us, or at all, or suppliers may not be able to allocate sufficient capacity in order to meet our increased requirements, which could have an adverse effect on our ability to meet customer demand for our products and our results of operations.
We seek to maintain sufficient levels of inventory and components in order to protect ourselves from supply interruptions. As a result, we are subject to the risk that a portion of our inventory will become obsolete or expire, which could have a material adverse effect on our earnings and cash flows due to the resulting costs associated with the inventory impairment charges and costs required to replace such inventory.
We manufacture and assemble components for our products and U.S. product candidate, and a loss or degradation in the performance of our manufacturing capabilities could have a material adverse effect on our business, financial condition and results of operations.
We manufacture and assemble components used in our artificial insemination product, permanent birth control system, and women-specific medical products. Our ability to maintain sufficient levels of inventory for our products could be negatively affected by many factors, including our failure to accurately manage our staffing requirements or a decrease in production capabilities. Conversely, if we overestimate customer demand for our artificial insemination product, permanent birth control system, and women-specific medical products, our production staff may be in excess of that needed, and this could result in excess cost, which could have a material adverse effect on our business, financial condition and results of operations.
We rely on a limited number of third-party suppliers for components for our products, as well as the sterilization of certain of our products, and a loss or degradation in performance of these suppliers could have a material adverse effect on our business, financial condition and results of operations.
We rely on third-party suppliers for the raw materials and components used in our artificial insemination product, permanent birth control system, and women-specific medical products. These suppliers may be unwilling or unable to supply the necessary materials and components reliably and at the levels we anticipate or that are required by the market. Our ability to supply our products commercially and to develop any future products depends, in part, on our ability to obtain these materials, components and products in accordance with regulatory requirements and in sufficient quantities for commercialization and clinical testing. While our suppliers have generally met our demand for their products and services on a timely basis in the past, we cannot guarantee that they will in the future be able to meet our demand for their products, either because of acts of nature, or our relative importance to them as a customer, and our suppliers may decide in the future to discontinue or reduce the level of business they conduct with us. If we are required to change suppliers due to any change in or termination of our relationships with these third parties, or if our suppliers are unable to obtain the materials they need to produce our components at consistent prices or at all, we may have to make modifications or changes to our products triggering the need for additional regulatory clearances or approvals, lose sales, experience manufacturing or other delays, incur increased costs or otherwise experience impairment to our customer relationships. We cannot guarantee that we will be able to establish alternative relationships on similar terms, without delay or at all.
While we believe replacement suppliers exist for all materials, components and services necessary to manufacture our products, establishing additional or replacement suppliers for any of these materials, components or services, if required, could be time-consuming and expensive, may result in interruptions in our operations and product delivery, may affect the performance specifications of our products or could require that we modify their design. Even if we are able to find replacement suppliers, we will be required to verify that the new supplier maintains facilities, procedures and operations that comply with our quality expectations and applicable regulatory requirements. Furthermore, our suppliers could require us to use alternative materials or components. Any of these events could require that we obtain a new regulatory authority approval before we implement the change, which could result in further delay and which may not be obtained at all. While we seek to maintain sufficient levels of inventory as discussed above, those inventories may not fully protect us from supply interruptions.
We have only limited supply arrangements in place with respect to certain components of our manufacturing process, and these arrangements do not extend to full commercial supply. We acquire certain key materials on a purchase order basis. As a result, we do not have long-term committed arrangements with respect to certain of the materials for our products and U.S. product candidate and other materials. If we obtain marketing approval, grant or clearance for our U.S. product candidate, we will need to establish an agreement for the commercial manufacture of certain key materials with a third party.
In addition, we are dependent on a sole supplier for certain components of our manufacturing process. Our current dependence on a single supplier for these components and the challenges we may face in obtaining adequate replacements involve several risks, including limited control over pricing, availability, quality and delivery schedules. Even if we are able to replace any raw materials or other materials with an alternative, such alternatives may cost more, result in lower yields or not be as suitable for our purposes. In addition, some of the materials that we use to manufacture our U.S. product candidate are complex materials, which may be more difficult to substitute. Therefore, any disruptions arising from our sole suppliers could result in delays and additional regulatory submissions. Our current and anticipated future dependence upon others for the manufacture of certain components of our product candidates or products may adversely affect our business, financial condition and results of operations.
Moreover, we rely on third-party sterilizers to effectively sterilize our products and U.S. product candidate and failure of any third-party sterilizer could result in safety risks associated with our products and U.S. product candidate and could result in patient or study subject injuries which could expose our company to product liability claims and actions. Contract sterilizers are inspected by the FDA and may be inspected by foreign regulatory authorities. Additionally, the closures and potential closures of facilities that use ethylene oxide to sterilize medical devices prior to their use may create delays or interruptions in the supply chain for our products and U.S. product candidate. Any compliance failures at any contract sterilizers we may contract with for sterilization of our products and U.S. product candidate also could create supply chain delays and interruptions and may require that we identify and contract with alternative contract sterilizers which we may not be able to do timely or on terms favorable to us. Any failures in the performance of our contract sterilizers may adversely affect our business, financial condition and results of operations.
Performance issues, service interruptions or price increases by our shipping carriers could adversely affect our business and harm our reputation and ability to provide our services on a timely basis.
Expedited, reliable shipping is essential to our operations. We rely heavily on providers of transport services for reliable and secure point-to-point transport of our products to our customers and for tracking of these shipments. Should a carrier encounter delivery performance issues such as loss, damage or destruction of any systems, it would be costly to replace such systems in a timely manner and such occurrences may damage our reputation and lead to decreased demand for our products and increased cost and expense to our business. In addition, any significant increase in shipping rates could adversely affect our operating margins and results of operations. Similarly, strikes, severe weather, natural disasters or other service interruptions affecting delivery services we use would adversely affect our ability to process orders for our products on a timely basis.
Consolidation in the healthcare industry or group purchasing organizations could lead to demands for price concessions, which may affect our ability to sell our products at prices necessary to support our current business strategies.
Healthcare costs have risen significantly over the past decade, which has resulted in or led to numerous cost reform initiatives by legislators, regulators and third-party payors. Cost reform has triggered a consolidation trend in the healthcare industry to aggregate purchasing power, which may create more requests for pricing concessions in the future. Additionally, group purchasing organizations, independent delivery networks and large single accounts may continue to use their market power to consolidate purchasing decisions for hospitals and healthcare practitioner practices. We expect that market demand, government regulation, third-party coverage and reimbursement policies and societal pressures will continue to change the healthcare industry worldwide, resulting in further business consolidations and alliances among our customers, which may exert further downward pressure on the prices of our products.
We have limited experience marketing and selling our women-specific medical product solutions, and if we are unable to expand, manage and maintain our direct sales and marketing organization we may not be able to generate revenue growth.
We have limited experience marketing and selling our women-specific medical products. We currently sell primarily our FemaSeed. FemVue, and FemSperm products through a direct effort in the U.S., that targets healthcare practitioner gynecologists and reproductive endocrinologists. We offer healthcare practitioners online training and new customer support and utilize various direct-to-patient marketing initiatives, including social media, a healthcare practitioner locator on a patient website, and online videos. As of December 31, 2025, we have 13 employees exclusively involved in our sales and marketing efforts. Our operating results are directly dependent upon the efforts of these employees.
In order to generate future revenue growth, we are in the process of developing geographic scope of a direct sales organization now that the FemaSeed product is available in the U.S. This is expected to represent a significant expansion of our commercialization efforts, costs and attention. Our success depends largely on our ability to hire, train, retain and motivate skilled sales and marketing personnel with significant industry experience and technical knowledge of related products. Because the competition for their services is high, we cannot assure you we will be able to hire and retain additional personnel on favorable or commercially reasonable terms, if at all. Failure to hire or retain qualified sales and marketing personnel would prevent us from expanding our business and generating revenue. If we are unable to expand our sales and marketing capabilities, we may not be able to effectively commercialize our FemaSeed product and other women-specific medical products, which could have an adverse effect on our business, financial condition and results of operations. In order to further expand revenue growth once the FemBloc system is available in the U.S. market, we plan to enlarge the geographic scope of the direct sales organization. Our future success will depend largely on our ability to hire, train, retain and motivate additional skilled sales and marketing personnel with significant industry experience and technical knowledge of related products. If we are unable to expand our sales and marketing capabilities, we may not be able to effectively commercialize our FemBloc system, which could have an adverse effect on our business, financial condition and results of operations.
To successfully market and sell our artificial insemination product, permanent birth control system, and women specific medical product solutions in markets outside of the United States, we must address many international business risks with which we have limited experience.
Our strategy is to increase our international presence in Europe, as well as, other international markets, such as Japan and Israel, which may further increase our revenue from markets outside the United States. International sales are subject to a number of risks, including:
•
difficulties in securing distribution partnerships and managing our international relationships;
•
increased competition as a result of more products and procedures receiving regulatory approval or otherwise free to market in international markets;
•
longer accounts receivable payment cycles and difficulties in collecting accounts receivable;
•
reduced or varied protection for intellectual property rights in some countries;
•
export restrictions, trade regulations, and foreign tax laws;
•
fluctuations in currency exchange rates;
•
foreign certification and regulatory clearance or approval requirements;
•
customs clearance and shipping delays;
•
political, social, and economic instability abroad, terrorist attacks, and security concerns in general;
•
preference for locally produced products;
•
potentially adverse tax consequences, including the complexities of foreign value-added tax systems;
•
the burdens of complying with a wide variety of foreign laws and different legal standards; and
•
increased financial accounting and reporting burdens and complexities.
If one or more of these risks are realized, our business, financial condition and results of operations could be adversely affected.
We plan to rely on our own direct sales force in North America for our women-specific medical products, which may result in higher fixed costs than our competitors and may slow our ability to reduce costs in the face of a sudden decline in demand for our products.
We plan to rely on our own direct sales force in North America and third-party distribution partners in Europe and other international countries, to market and sell our products. Some of our competitors rely predominantly on independent sales agents and third-party distributors. A direct sales force may subject us to higher fixed costs than those of companies that market competing products through independent third parties, due to the costs that we will bear associated with employee benefits, training and managing sales personnel. As a result, we could be at a competitive disadvantage. Additionally, these fixed costs may slow our ability to reduce costs in the face of a sudden decline in demand for our products, which could have a material adverse effect on our business, financial condition and results of operations.
We face the risk of product liability claims that could be expensive, divert management’s attention and harm our reputation and business. We may not be able to maintain adequate product liability insurance.
Our business exposes us to the risk of product liability claims that are inherent in the testing, manufacturing and marketing of medical products, including sterile medical products. This risk exists even if it is approved or cleared for commercial sale by the FDA and manufactured in facilities licensed and regulated by the FDA or an applicable foreign regulatory authority. Our FemBloc system and FemaSeed product are designed to affect, and any future products will be designed to affect, important bodily functions and processes, such as the female reproductive system. Any side effects, manufacturing defects, misuse or abuse associated with our FemBloc system, FemaSeed product and other women specific medical products, including sterilization failures, could result in patient injury or death. The medical device industry has historically been subject to extensive litigation over product liability claims, and we cannot offer any assurance that we will not face product liability suits. For example, Essure, a permanent birth control system previously marketed by Bayer, involved the implant of coils into a woman’s fallopian tubes by way of a hysteroscope, where they were to permanently remain. In 2016, the FDA ordered Bayer to conduct a post-market surveillance study and required a box warning to the product labeling, which included a warning of possible perforation of the uterus and/or fallopian tubes, identification of inserts in the abdominal or pelvic cavity, persistent pain, and suspected allergic or hypersensitivity reactions. In April 2018, the FDA restricted the sale and distribution of Essure. The product was removed by Bayer from all markets, including the U.S. effective December 2018. There can be no assurance that serious adverse safety concerns may not arise with the FemBloc system.
We may be subject to product liability claims if our products cause, or merely appear to have caused, patient injury or death. In addition, an injury that is caused by the activities of our suppliers, such as those who provide us with components and raw materials, or any contract sterilizer, may be the basis for a claim against us. Product liability claims may be brought against us by patients, healthcare providers or others selling or otherwise coming into contact with our products, among others. If we cannot successfully defend ourselves against product liability claims, we will incur substantial liabilities and reputational harm. In addition, regardless of merit or eventual outcome, product liability claims may result in:
•
distraction of management’s attention from our primary business;
•
the inability to commercialize our current and future products;
•
decreased demand for our current and future products;
•
damage to our business reputation;
•
product recalls or withdrawals from the market;
•
withdrawal of clinical trial participants;
•
substantial monetary awards to patients or other claimants; or
While we may attempt to manage our product liability exposure by proactively recalling or withdrawing from the market any defective products, any recall or market withdrawal of our products may delay the supply of those products to our customers and may impact our reputation. We can provide no assurance that we will be successful in initiating appropriate market recall or market withdrawal efforts that may be required in the future or that these efforts will have the intended effect of preventing product malfunctions and the accompanying product liability that may result. Such recalls and withdrawals may also be used by our competitors to harm our reputation for safety or be perceived by patients as a safety risk when considering the use of our products, either of which could have a material adverse effect on our business, financial condition and results of operations.
Although we have product liability and clinical trial liability insurance that we believe is appropriate, this insurance is subject to deductibles and coverage limitations. Our current product liability insurance may not continue to be available to us on acceptable terms, if at all, and, if available, coverage may not be adequate to protect us against any future product liability claims. If we are unable to obtain insurance at an acceptable cost or on acceptable terms or otherwise protect against potential product liability claims, we could be exposed to significant liabilities. A product liability claim, recall or other claim with respect to uninsured liabilities or for amounts in excess of insured liabilities could have a material adverse effect on our business, financial condition and results of operations.
If the quality of our artificial insemination product, permanent birth control system, and women-specific medical product solutions does not meet the expectations of healthcare practitioners or patients, then our brand and reputation or our business could be adversely affected.
In the course of conducting our business, we must adequately address quality issues that may arise with our artificial insemination product, permanent birth control system, and women-specific medical product solutions, including defects in third-party components included in our products. Although we have established internal procedures designed to minimize risks that may arise from quality issues, there can be no assurance that we will be able to eliminate or mitigate occurrences of these issues and associated liabilities. In addition, even in the absence of quality issues, we may be subject to claims and liability if the performance of our products do not live up to the expectations of healthcare practitioners or patients. If the quality of our products do not meet the expectations of healthcare practitioners or patients, then our brand and reputation with those healthcare practitioners or patients, or our business, financial condition and results of operations could be adversely affected.
If we choose to acquire new and complementary businesses, products or technologies, we may be unable to complete these acquisitions or to successfully integrate them in a cost-effective and non-disruptive manner.
Our success depends, in part, on our ability to continually enhance and broaden our product offerings in response to changing customer demands, competitive pressures and advances in technologies. Accordingly, although we have no current commitments with respect to any acquisition or investment, we may in the future pursue the acquisition of, or joint ventures relating to, complementary businesses, products or technologies instead of developing them ourselves. We do not know if we will be able to successfully complete any future acquisitions or joint ventures, or whether we will be able to successfully integrate any acquired business, product or technology or retain any key employees related thereto. Integrating any business, product or technology we acquire could be expensive and time-consuming, disrupt our ongoing business and distract our management. If we are unable to integrate any acquired businesses, products or technologies effectively, our business will be adversely affected. In addition, any amortization or charges resulting from the costs of acquisitions could increase our expenses.
Risks Related to Managing Growth and Employee Matters
We face risks related to health epidemics and outbreaks, which could significantly disrupt our commercialization activities and clinical trials, and could have an adverse impact on our business.
We face risks related to health epidemics or outbreaks of communicable diseases. The extent to which a health epidemic or outbreak may impact our clinical trial operations will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the duration and geographic reach of an outbreak and the effectiveness of actions to contain and treat the outbreak. Epidemics could have a material adverse effect on our business, financial condition, results of operations and prospects.
Failure of a key information technology system, process or site could have an adverse effect on our business.
We rely extensively on information technology systems to conduct our business. These systems affect, among other things, ordering and managing materials from suppliers, shipping products to customers, processing transactions, summarizing and reporting results of operations, complying with regulatory, legal or tax requirements, data security and other processes necessary to manage our business. If our systems are damaged or cease to function properly due to any number of causes, ranging from catastrophic events to power outages to security breaches, and our business continuity plans do not effectively compensate on a timely basis, we may experience interruptions in our operations, which could have an adverse effect on our business. Furthermore, any breach in our IT systems could lead to the unauthorized access, disclosure and use of non-public information, including information from our patient registry or other patient information, which is protected by HIPAA and other laws. Any such access, disclosure, or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, and damage to our reputation.
In addition, we accept payments for some of our sales through credit and debit card transactions, which are handled through a third-party payment processor. As a result, we are subject to a number of risks related to credit and debit card payments. As a result of these transactions, we pay interchange and other fees, which may increase over time and could require us to either increase the prices we charge for our products or experience an increase in our costs and expenses. In addition, as part of the payment processing process, we transmit our customers’ credit and debit card information to our third-party payment processor. We may in the future become subject to lawsuits or other proceedings for purportedly fraudulent transactions arising out of the actual or alleged theft of our customers’ credit or debit card information if the security of our third-party credit card payment processor is breached. We and our third-party credit card payment processor are also subject to payment card association operating rules, certification requirements and rules governing electronic funds transfers, which could change or be reinterpreted to make it difficult or impossible for us to comply. If we or our third-party credit card payment processor fail to comply with these rules or requirements, we may be subject to fines and higher transaction fees and lose our ability to accept credit and debit card payments from our customers, and there may be an adverse effect on our business.
If our facilities are damaged or become inoperable, we will be unable to continue to research, develop, manufacture and supply our products and, as a result, there will be an adverse effect on our business until we are able to secure a new facility and rebuild our inventory.
We do not have redundant facilities. We perform substantially all of our research, development, manufacturing and back-office activity and maintain all our finished goods inventory in a single location in Suwanee, Georgia. Our facility, equipment and inventory would be costly to replace and could require substantial lead time to repair or replace. The facility may be harmed or rendered inoperable by natural or man-made disasters, including, but not limited to, tornadoes, flooding, fire and power outages, which may render it difficult or impossible for us to perform our research, development and commercialization activities for some period of time. The inability to perform those activities, combined with the time it may take to rebuild our inventory of finished product, may result in the loss of customers or harm to our reputation. Although we possess insurance for damage to our property and the disruption of our business, this insurance may not be sufficient to cover all of our potential losses and this insurance may not continue to be available to us on acceptable terms, or at all.
Our ability to maintain our competitive position depends on our ability to attract and retain senior management and other highly qualified personnel.
Our success depends in part on our continued ability to attract, retain and motivate highly qualified management, clinical and other personnel. We are highly dependent upon our management team, particularly our Chief Executive Officer and President and the rest of our senior management, and other key personnel. Although we have entered into employment letter agreements with all of our executive officers, each of them may terminate their employment with us at any time. The replacement of any of our key personnel likely would involve significant time and costs and may significantly delay or prevent the achievement of our business objectives and could therefore have an adverse effect on our business. In addition, we do carry “key person” insurance policy for our Chief Executive Officer and President that could offset potential loss of service under applicable circumstances.
We will need to grow the size of our organization, and we may experience difficulties in managing this growth.
As of December 31, 2025, we had 51 full-time employees, 3 part-time employees and 35 consultants. As our development and commercialization plans and strategies develop, we expect to need additional managerial, operational, sales, marketing, financial and other personnel. Future growth would impose significant added responsibilities on members of management, including:
•
identifying, recruiting, integrating, maintaining and motivating additional employees;
•
managing our internal development efforts effectively, including the clinical and FDA application preparation for our U.S. product candidate, while complying with our contractual obligations to contractors and other third parties; and
•
improving our operational, financial and management controls, reporting systems and procedures.
Our future financial performance and our ability to commercialize our products and any product candidate(s) that are approved for marketing will depend, in part, on our ability to effectively manage any future growth, and our management may also have to divert a disproportionate amount of its attention away from day-to-day activities in order to devote a substantial amount of time to managing these growth activities.
We currently rely, and for the foreseeable future will continue to rely, in substantial part on certain independent organizations, advisors and consultants to provide certain services, including substantially all aspects of legal and compliance, regulatory marketing authorization, clinical trial management and manufacturing. There can be no assurance that the services of independent organizations, advisors and consultants will continue to be available to us on a timely basis when needed, or that we can find qualified replacements. In addition, if we are unable to effectively manage our outsourced activities or if the quality or accuracy of the services provided by consultants is compromised for any reason, our clinical trials may be extended, delayed or terminated, and we may not be able to obtain regulatory approval of our U.S. product candidate or otherwise advance our business. There can be no assurance that we will be able to manage our existing consultants or find other competent outside contractors and consultants on economically reasonable terms, or at all.
If we are not able to effectively expand our organization by hiring new employees and expanding our groups of consultants and contractors, we may not be able to successfully implement the tasks necessary to further develop and commercialize our products and potentially commercialize our U.S. product candidate and, accordingly, may not achieve our research, development and commercialization goals.
The increasing use of social media platforms presents new risks and challenges.
Social media is increasingly being used in connection with our commercialization efforts for our products, our clinical development program and following approval of our U.S. product candidate, if any. Social media practices in the biomedical industry continue to evolve and regulations and regulatory guidance relating to such use are evolving and not always clear. This evolution creates uncertainty and risk of noncompliance with regulations applicable to our business, resulting in potential regulatory actions against us, along with the potential for litigation related to off-label marketing or other prohibited activities and heightened scrutiny by the FDA, the SEC and other regulators. For example, patients may use social media channels to comment on their experience in an ongoing clinical trial or to report an alleged adverse event. If such disclosures occur, there is a risk that trial enrollment may be adversely impacted, that we may fail to monitor and comply with applicable adverse event reporting obligations or that we may not be able to defend our business or the public’s legitimate interests in the face of the political and market pressures generated by social media due to restrictions on what we may say about our U.S. product candidate. There is also a risk of inappropriate disclosure of sensitive information or negative or inaccurate posts or comments about us or the products we are marketing or developing on any social networking website. In addition, we may encounter attacks on social media regarding our company, management, product candidates or products. If any of these events were to occur or we otherwise fail to comply with applicable regulations, we could incur liability, face regulatory actions or incur other harm to our business.
Risks Related to Government Regulation
Our products and operations are subject to extensive government regulation and oversight both in the United States and abroad, and our failure to comply with applicable requirements could harm our business.
We and our products are subject to extensive regulation in the United States and elsewhere, including by the FDA and its foreign counterparts. The FDA and foreign regulatory authorities regulate, among other things, with respect to medical devices: design, development and manufacturing; testing, labeling, clinical trials; product safety; establishment registration and device listing; marketing, sales and distribution; pre-market clearance and approval; complaint handling; record keeping procedures; advertising and promotion; recalls and field safety corrective actions; post-market surveillance, including reporting of deaths or serious injuries and malfunctions that, if they were to recur, would be likely to cause or contribute to death or serious injury; post-market approval studies; and product import and export.
The regulations to which we are subject are complex and have tended to become more stringent over time. Regulatory changes could result in restrictions on our ability to carry on or expand our operations, higher than anticipated costs or lower than anticipated sales. The FDA enforces these regulatory requirements through periodic unannounced inspections. We do not know whether the FDA will identify any areas of noncompliance in any future FDA inspections or those conducted by foreign regulatory authorities. Failure to comply with applicable regulations could jeopardize our ability to sell our products and result in enforcement actions such as: warning letters; fines; injunctions; civil penalties; termination of distribution; recalls or seizures of products; delays in the introduction of products into the market; total or partial suspension of production; refusal to grant future approvals; withdrawals or suspensions of approvals, and in the most serious cases, criminal penalties.
We may not receive the necessary approvals, granted de novo classifications, or clearances for our FemBloc system or future devices and expanded indications, and failure to timely obtain these marketing authorizations would adversely affect our ability to grow our business.
Our strategy is dependent on obtaining regulatory approval of our FemBloc system. In the United States, before we can market a new medical device, or a new use of, certain new claims for, or significant modifications to an existing product, we must first receive either clearance under Section 510(k) of the Federal Food, Drug, and Cosmetic Act, or the FDCA, de novo classification under Section 513(f) (2) of the FDCA, or approval of a PMA from the FDA, unless an exemption applies. In the 510(k) clearance process, before a device may be marketed, the FDA must determine that a proposed device is “substantially equivalent” to a legally-marketed “predicate” device, which includes a device that has been previously cleared through the 510(k) process, a device that was legally marketed prior to May 28, 1976 (pre-amendments device), a device that was originally on the U.S. market pursuant to an approved PMA and later down-classified, a device that was de novo classified under section 513(f)(2) of the FDCA, or a 510(k)-exempt device. To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the predicate device. Clinical data are sometimes required to support substantial equivalence demonstrations. The de novo classification process provides a pathway to classify novel medical devices for which general controls alone, or general and special controls, provide reasonable assurance of safety and effectiveness for the device with the proposed intended use, but for which there is no legally marketed predicate device. A de novo classification is a risk-based classification process through which devices are classified into class I or class II. Devices classified in response to a de novo classification request may be marketed and used as predicates for future pre-market notification 510(k) submissions. In the process of obtaining PMA approval, which is required for our FemBloc system, the FDA must determine that a proposed device is safe and effective for its intended use based, in part, on extensive data, including, but not limited to, technical, preclinical, clinical trial, manufacturing and labeling data. The PMA process is typically required for devices that are deemed to pose the greatest risk, such as life-sustaining, life-supporting or implantable devices. Modifications to products that are approved through a PMA application generally require FDA approval.
Similarly, certain modifications made to products cleared through a 510(k) or granted a de novo classification may require a new 510(k) clearance, or could require a new de novo classification request or even a PMA. The PMA approval, de novo classification, and the 510(k) clearance processes can be expensive, lengthy and uncertain. The FDA’s 510(k) clearance process usually takes from three to seven months, but can last longer, while the de novo classification process is usually longer and often requires a clinical trial. The process of obtaining a PMA is much more costly and uncertain than the de novo or 510(k) clearance processes and generally takes one year, or even longer, from the time the application is filed with the FDA. In addition, a PMA generally requires the performance of one or more clinical trials. Despite the time, effort and cost, a device may not be approved, granted a de novo classification, or cleared by the FDA. Any delay or failure to obtain necessary regulatory authorizations could harm our business. Furthermore, even if we are granted 510(k) clearances, de novo classifications, or approvals, they may include significant limitations on the indications for uses for the device, which may limit the market for the device.
In the United States, we are currently seeking approval of our permanent birth control system through the PMA pathway. Even if the PMA is approved, any future modification to our permanent birth control system may require us to submit a new PMA or PMA supplement and obtain FDA approval prior to implementing the change, although some modifications can be reported in an annual report or through a 30-day Notice. The FDA may not agree with our decisions regarding whether a new PMA or PMA supplement is necessary. If the FDA requires us to go through a lengthier, more rigorous process for future products or modifications to existing products than we had expected, product introductions or modifications could be delayed or canceled, which could adversely affect our ability to grow our business. FemaSeed, FemVue, FemCath, FemChec, FemCerv and FemVue Controlled and Mini have each received 510(k) clearance.
In Europe, before we can market a new medical device, or a new use of, certain new claims for, or significant modifications to an existing product, in member countries of the EEA, our products must comply with the essential requirements of the Medical Devices Regulation (Regulation (EU) 2017/745). Compliance with these requirements is a prerequisite to be able to affix the Conformité Européene, or CE, mark to our products, without which they cannot be sold or marketed in the EEA. To demonstrate compliance with the essential requirements we must perform a conformity assessment procedure, which varies according to the type of medical device and its classification. Except for low-risk medical devices (Class I non-sterile, non-measuring devices), where the manufacturer can issue an EC Declaration of Conformity based on a self-assessment of the conformity of its products with the essential requirements of the EU Medical Devices Regulation, a conformity assessment procedure requires the intervention of an organization accredited by a member state of the EEA to conduct conformity assessments, or a notified body. Depending on the relevant conformity assessment procedure, the notified body would typically audit and examine the technical file and the quality system for the manufacture, design and final inspection of our devices. The notified body issues a certificate of conformity following successful completion of a conformity assessment procedure conducted in relation to the medical device and its manufacturer and their conformity with the essential requirements. This certificate entitles the manufacturer to affix the CE mark to its medical devices after having prepared and signed a related EC Declaration of Conformity. Since 26 May 2021, all manufacturers of medical devices sold in the EEA have to be compliant with the rules set out in the Medical Devices Regulation. The Medical Devices Regulation has the same basic requirements as the repealed EU Medical Devices Directive, but is generally more stringent, especially in terms of risk classes and the oversight provided by notified bodies. There is also more emphasis on vigilance and post-market surveillance.
As a general rule, demonstration of conformity of medical devices and their manufacturers with the essential requirements must be based, among other things, on the evaluation of clinical data supporting the safety and performance of the products during normal conditions of use. Specifically, a manufacturer must demonstrate that the device achieves its intended performance during normal conditions of use, that the known and foreseeable risks, and any adverse events, are minimized and acceptable when weighed against the benefits of its intended performance, and that any claims made about the performance and safety of the device are supported by suitable evidence. If we fail to remain in compliance with applicable European laws, we would be unable to continue to affix the CE mark to our products, which would prevent us from selling them within the EEA.
All medical devices must be registered with the Medicines & Healthcare products Regulatory Agency (MHRA) before being placed on the Great Britain, or GB, market. European CE marks will continue to be recognized in GB until June 30, 2023, following which a UKCA mark will be required for a medical device to be marketed in GB. The EU regulatory framework on medical devices will, however, continue to apply in Northern Ireland under the Northern Irish Protocol and medical devices in Northern Ireland may either carry a European CE mark or a CE UKNI mark (although devices bearing the CE UKNI marking can only be placed on the market in Northern Ireland and will not be accepted on the EU market).
As a general rule, demonstration of conformity of medical devices and their manufacturers with the essential requirements must be based, among other things, on the evaluation of clinical data supporting the safety and performance of the products during normal conditions of use. Specifically, a manufacturer must demonstrate that the device achieves its intended performance during normal conditions of use, that the known and foreseeable risks, and any adverse events, are minimized and acceptable when weighed against the benefits of its intended performance, and that any claims made about the performance and safety of the device are supported by suitable evidence. If we fail to remain in compliance with applicable European laws and directives, we would be unable to continue to affix the CE mark to our products, which would prevent us from selling them within the EEA. FemBloc, FemaSeed, FemVue, FemCath, FemChec, FemCerv and FemVue Mini have each received the CE mark demonstrating compliance with the EU Medical Device Regulation. FemBloc, FemaSeed, FemVue and FemCerv have received approval from the MHRA.
The FDA or foreign regulatory bodies can delay, limit or deny a marketing authorization of a device for many reasons, including:
•
our inability to demonstrate to the satisfaction of the FDA or the applicable regulatory entity or notified body that our products are safe or effective for their intended uses or, in the U.S. for a 510(k) device, that they are substantially equivalent to the predicate;
•
the disagreement of the FDA or the applicable foreign regulatory body or notified body with the design or implementation of our clinical trials or the interpretation of data from preclinical studies or clinical trials;
•
serious and unexpected adverse device effects experienced by participants in our clinical trials;
•
the data from our preclinical studies and clinical trials may be insufficient to support U.S. FDA approval, de novo classification or clearance where required or approval of the applicable regulatory entity or notified body;
•
our inability to demonstrate that the clinical and other benefits of the device outweigh the risks;
•
the manufacturing process or facilities we use may not meet applicable requirements; and
•
the potential for approval policies or regulations of the FDA or applicable foreign regulatory bodies or notified body to change significantly in a manner rendering our clinical data or regulatory filings insufficient to support a marketing authorization.
In addition, the FDA may change its policies, adopt additional regulations or revise existing regulations, or take other actions, which may prevent or delay approval, de novo classification or clearance of our future products under development or impact our ability to modify our currently cleared products on a timely basis. Such policy or regulatory changes could impose additional requirements upon us that could delay our ability to obtain new approvals, granted de novo classifications, or 510(k) clearances, or increase the costs of compliance or restrict our ability to maintain our current 510(k) clearances. For example, as part of the FDA Reauthorization Act, or FDARA, in 2017, Congress reauthorized the Medical Device User Fee Amendments with various FDA performance goal commitments and enacted several regulatory improvements related to devices and miscellaneous reforms, which are further intended to clarify and improve medical device regulation both pre- and post- clearance and approval. Some of these proposals and reforms could impose additional regulatory requirements upon us that could delay our ability to obtain new approvals, granted de novo classifications, or 510(k) clearances, or increase the costs of compliance.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. In February 2026, we announced MDSAP certification for FemBloc.
Modifications to our products may require us to obtain new PMA approvals or approvals of a PMA supplement, and if we market modified products without obtaining necessary approvals, we may be required to cease marketing or recall the modified products until required approvals are obtained for the United States.
For the U.S., certain modifications to a PMA-approved device may require approval of a new PMA or a PMA supplement, while other modifications can be reported in an annual report or through a 30-day Notice. The FDA may not agree with our decisions regarding whether a new PMA or PMA supplement is necessary. We may make modifications to our approved devices in the future that we believe do not require approval of a new PMA or PMA supplement. If the FDA disagrees with our determination and requires us to submit a new PMA or PMA supplement for modifications to our previously approved products, we may be required to cease marketing or to recall the modified product until we obtain approval, and we may be subject to significant regulatory fines or penalties. For de novo classified or 510(k) cleared devices we will need to submit a new 510(k) pre-market notification for any change or modification in the device that could significantly affect the safety or effectiveness of the device, or for a major change or modification in the intended use of the device. The FDA may not agree with our determination whether a new 510(k) is required for a modification, in which case we may be required to cease marketing or recall the modified product until we receive 510(k) clearance.
In addition, the FDA may not approve, de novo classify or clear our products for the indications that are necessary or desirable for successful commercialization or could require clinical trials to support any modifications. Any delay or failure in obtaining required authorizations would adversely affect our ability to introduce new or enhanced products in a timely manner, which in turn would harm our future growth.
Failure to comply with post-marketing regulatory requirements could subject us to enforcement actions, including substantial penalties, and might require us to recall or withdraw a product from the market.
After approval for our permanent birth control system, we will be subject to ongoing and pervasive regulatory requirements governing, among other things, the manufacture, marketing, labeling, packaging, advertising, medical device reporting, sale, promotion, registration, storage, distribution and listing of devices. For example, we must submit periodic reports to the FDA as a condition of PMA approval. These reports include safety and effectiveness information about the device after its approval. Failure to submit such reports, or failure to submit the reports in a timely manner, could result in enforcement action by the FDA. Following its review of the periodic reports, the FDA might ask for additional information or initiate further investigation.
In addition, the PMA approval for our FemBloc system in the U.S. may be subject to several conditions of approval, including a post-market extended follow-up of the pre-market study cohort. Any failure to comply with the conditions of approval could result in the withdrawal of PMA approval and the inability to continue to market the device. Adverse outcomes in these studies could also be grounds for withdrawal of approval of the PMA.
The regulations to which we are subject are complex and have become more stringent over time. Regulatory changes could result in restrictions on our ability to continue or expand our operations, higher than anticipated costs, or lower than anticipated sales. Even after we have obtained the proper regulatory authorization to market a device, we have ongoing responsibilities under FDA regulations and applicable foreign laws and regulations. The FDA, state and foreign regulatory authorities have broad enforcement powers. Our failure to comply with applicable regulatory requirements could result in enforcement action by the FDA, state or foreign regulatory authorities, which may include any of the following sanctions:
•
untitled letters or warning letters;
•
fines, injunctions, consent decrees and civil penalties;
•
recalls, termination of distribution, administrative detention, or seizure of our products;
•
customer notifications or repair, replacement or refunds;
•
operating restrictions or partial suspension or total shutdown of production;
•
delays in or refusal to grant our requests for future PMA approvals or foreign regulatory approvals of new products, new intended uses, or modifications to existing products;
•
withdrawals or suspensions of our current PMA or foreign regulatory approvals, resulting in prohibitions on sales of our products;
•
FDA refusal to issue certificates to foreign governments needed to export products for sale in other countries; and
Any of these sanctions could result in higher than anticipated costs or lower than anticipated sales and have a material adverse effect on our reputation, business, financial condition and results of operations.
Our products must be manufactured in accordance with federal and state regulations, and we or any of our suppliers could be forced to recall our products or terminate production if we fail to comply with these regulations.
The methods used in, and the facilities used for, the manufacture of our products must comply with the FDA’s Quality System Regulation, or QSR, which is a complex regulatory scheme that covers good manufacturing practices for the procedures and documentation of the design, testing, production, process controls, quality assurance, labeling, packaging, handling, storage, distribution, installation, and servicing of medical devices. Furthermore, we are required to verify that our suppliers and service providers maintain facilities, procedures and operations that comply with our quality standards and applicable regulatory requirements. The FDA enforces the QSR through periodic announced or unannounced inspections of medical device manufacturing facilities, which may include the facilities of subcontractors. Our products are also subject to similar state regulations, including state wholesale distribution requirements, and various laws and regulations of foreign countries governing manufacturing.
We may not take the necessary steps to comply with applicable regulations, which could cause delays in the delivery of our products. In addition, failure to comply with applicable FDA requirements or later discovery of previously unknown problems with our products or manufacturing processes could result in, among other things:
•
warning letters or untitled letters;
•
fines, injunctions or civil penalties;
•
suspension or withdrawal of approvals;
•
seizures or recalls of our products;
•
total or partial suspension of production or distribution;
•
administrative or judicially imposed sanctions;
•
the FDA’s refusal to grant pending or future approvals for our products;
•
clinical holds; refusal to permit the import or export of our products; and
•
criminal prosecution of us or our employees.
Any of these actions could significantly and negatively affect supply of our products. If any of these events occur, our reputation could be harmed, we could be exposed to product liability claims and we could lose customers and experience reduced sales and increased costs.
If treatment guidelines for permanent birth control or other women healthcare treatments change or the standard of care evolves, we may need to redesign and seek new marketing authorization from the FDA for one or more of our products.
If treatment guidelines for permanent birth control or other women healthcare treatments changes or the standard of care for any of these conditions evolve, we may need to redesign the applicable product and seek new approvals, grants or clearances from the FDA. If treatment guidelines change so that different treatments become desirable, the clinical utility of one or more of our products could be diminished and our business could be adversely affected.
The misuse or off-label use of our products may harm our reputation in the marketplace, result in injuries that lead to product liability suits or result in costly investigations, fines or sanctions by regulatory bodies if we are deemed to have engaged in the promotion of these uses, any of which could be costly to our business.
Although our products are marketed for the specific treatments for which the devices were designed and our personnel are trained not to promote our products for uses outside of the FDA-approved or cleared indications for use, known as “off-label uses”, we cannot, however, prevent a healthcare practitioner from using our products, when in the healthcare practitioner’s independent professional medical judgment, he or she deems it appropriate. There may be increased risk of injury to patients if healthcare practitioners attempt to use our products off-label. Furthermore, the use of our products for indications other than those approved, granted or cleared by the FDA or authorized by any foreign regulatory body may not effectively treat such conditions, which could harm our reputation in the marketplace among healthcare practitioners and patients.
If the FDA or any foreign regulatory body determines that our promotional materials or training constitute promotion of an off-label use, it could request that we modify our training or promotional materials or subject us to regulatory or enforcement actions, including the issuance or imposition of a warning letter or an untitled letter, injunction, seizure, civil fine or criminal penalties. It is also possible that other federal, state or foreign enforcement authorities might take action under other regulatory authority, such as false claims laws, if they consider our business activities to constitute promotion of an off-label use, which could result in significant penalties, including, but not limited to, criminal, civil and administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs and the curtailment of our operations.
In addition, healthcare practitioners may misuse our products or use improper techniques if they are not adequately trained, potentially leading to injury and an increased risk of product liability. If our products are misused or used with improper technique, we may become subject to costly litigation by our customers or their patients. Similarly, in an effort to decrease costs, healthcare practitioners may also reuse our products despite them being intended for a single use or may purchase reprocessed products from third-party reprocessors in lieu of purchasing a new product from us, which could result in product failure and liability. As described above, product liability claims could divert management’s attention from our core business, be expensive to defend and result in sizeable damage awards against us that may not be covered by insurance.
Our products may cause or contribute to adverse medical events or be subject to failures or malfunctions that we are required to report to the FDA, and if we fail to do so, we would be subject to sanctions that could harm our reputation, business, financial condition and results of operations. The discovery of serious safety issues with our products, or a recall of our products either voluntarily or at the direction of the FDA or another governmental authority, could have a negative impact on us.
We are subject to the FDA’s medical device reporting regulations and similar foreign regulations, which require us to report to the FDA when we receive or become aware of information that reasonably suggests that one or more of our products may have caused or contributed to a death or serious injury or malfunctioned in a way that, if the malfunction were to recur, would be likely to cause or contribute to a death or serious injury. The timing of our obligation to report is triggered by the date we become aware of the adverse event as well as the nature of the event. We may fail to report adverse events of which we become aware within the prescribed timeframe. We may also fail to recognize that we have become aware of a reportable adverse event, especially if it is not reported to us as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of the product. If we fail to comply with our reporting obligations, the FDA could take action, including warning letters, untitled letters, administrative actions, criminal prosecution, imposition of civil monetary penalties, seizure of our products or delay in clearance or approval of future products.
The FDA and foreign regulatory bodies have the authority to require the recall of commercialized products in the event of material deficiencies or defects in design or manufacture of a product or in the event that a product poses an unacceptable risk to health. The FDA’s authority to require a recall must be based on a finding that there is reasonable probability that the device could cause serious, adverse health consequences or death. We may also choose to voluntarily recall a product if any material deficiency is found. A government-mandated or voluntary recall by us could occur as a result of an unacceptable risk to health, component failures, malfunctions, manufacturing defects, labeling or design deficiencies, packaging defects or other deficiencies or failures to comply with applicable regulations. Product defects or other errors may occur in the future.
Depending on the corrective action we take to redress a product’s deficiencies or defects, the FDA may require, or we may decide, that we will need to obtain new clearances, grants or approvals for the device before we may market or distribute the corrected device. Seeking such clearances, grants or approvals may delay our ability to replace the recalled devices in a timely manner. Moreover, if we do not adequately address problems associated with our devices, we may face additional regulatory enforcement action, including FDA warning letters, product seizure, injunctions, administrative penalties or civil or criminal fines.
Certain voluntary field actions are required to be reported to the FDA and other regulatory authorities. Companies are required to maintain certain records of recalls and corrections, even if they are not reportable to the FDA. We may initiate voluntary withdrawals or corrections for our products in the future that we determine do not require notification of the FDA. If the FDA disagrees with our determinations, it could require us to report those actions as recalls and we may be subject to enforcement action. A future recall announcement could harm our reputation with customers, potentially lead to product liability claims against us and negatively affect our sales. Any corrective action, whether voluntary or involuntary, as well as defending ourselves in a lawsuit, will require the dedication of our time and capital, distract management from operating our business and may harm our reputation and financial results.
If we do not obtain and maintain international regulatory registrations or approvals for our products, we will be unable to market and sell our products outside of United States.
Sales of our products outside of United States are subject to foreign regulatory requirements that vary widely from country to country. In addition, the FDA regulates exports of medical devices from the United States. While the regulations of some countries may not impose barriers to marketing and selling our products or only require notification, others require that we obtain the approval of a specified regulatory body. Complying with foreign regulatory requirements, including obtaining registrations or approvals, can be expensive and time-consuming, and we may not receive regulatory approvals in each country in which we plan to market our products or we may be unable to do so on a timely basis. The time required to obtain registrations or marketing authorizations, if required by other countries, may be longer than that required for FDA approval, grant or clearance, and requirements for such registrations and marketing authorizations may significantly differ from FDA requirements. If we modify our products, we may need to apply for additional regulatory approvals before we are permitted to sell the modified product. In addition, we may not continue to meet the quality and safety standards required to maintain the authorizations that we have received. If we are unable to maintain our authorizations in a particular country, we will no longer be able to sell the applicable product in that country.
Regulatory approval, grant or clearance by the FDA does not ensure registration or marketing authorization by regulatory authorities in other countries, and registration or approval by one or more foreign regulatory authorities does not ensure registration or marketing authorization by regulatory authorities in other foreign countries or by the FDA. However, a failure or delay in obtaining registration or marketing authorization in one country may have a negative effect on the regulatory process in others.
In March 2025, we announced CE mark certification under EU MDR as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively.
Legislative or regulatory reforms in the United States or the European Union may make it more difficult and costly for us to obtain regulatory approvals, grants or clearances for our products or to manufacture, market or distribute our products after clearance or approval is obtained.
From time to time, legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the regulation of medical devices. In addition, FDA regulations and guidance are often revised or reinterpreted by the FDA in ways that may significantly affect our business and our products. Any new statutes, regulations or revisions or reinterpretations of existing regulations, requirements, and regulatory processes may impose additional costs or lengthen review times of any future products or make it more difficult to obtain approval for, manufacture, market or distribute our products. We cannot determine what effect changes in regulations, statutes, legal interpretation or policies, when and if promulgated, enacted or adopted may have on our business in the future. Such changes could, among other things, require: additional testing prior to obtaining clearance, grant or approval; changes to manufacturing methods; recall, replacement or discontinuance of our products; or additional record keeping.
On April 5, 2017, the European Parliament passed the Medical Devices Regulation (Regulation 2017/745), which repeals and replaces the EU Medical Devices Directive and the EU Active Implantable Medical Devices Directive (Directive 90/385/EEC) with effect from May 26, 2021. Unlike directives, which must be implemented into the national laws of the EEA member states, the Medical Devices Regulation is directly applicable, i.e., without the need for adoption of EEA member state laws implementing them, in all EEA member states and is intended to eliminate current differences in the regulation of medical devices among EEA member States.
The Medical Devices Regulation will among other things:
•
strengthen the rules on placing devices on the market and reinforce surveillance once they are available;
•
establish explicit provisions on manufacturers’ responsibilities for the follow-up of the quality, performance and safety of devices placed on the market;
•
improve the traceability of medical devices throughout the supply chain to the end-user or patient through a unique identification number;
•
set up a central database to provide patients, healthcare professionals and the public with comprehensive information on products available in the EU;
•
strengthen rules for the assessment of certain high-risk devices, such as implants, which may have to undergo an additional check by experts before they are placed on the market.
Under transitional provisions, medical devices with notified body certificates issued under the Medical Devices Directive prior to May 26, 2021 may continue to be placed on the market for the remaining validity of the certificate, until May 27, 2024 at the latest. After the expiry of any applicable transitional period, only devices that have been CE marked under the Medical Device Regulation may be placed on the market in the EEA. The new requirements introduced by the Medical Devices Regulation may make it harder for us to CE mark our products and may have an effect on the way we conduct our business in the EEA.
We are subject to certain federal, state and foreign fraud and abuse laws, health information privacy and security laws and transparency laws, which, if violated, could subject us to substantial penalties. Additionally, any challenge to or investigation into our practices under these laws could cause adverse publicity and be costly to respond to, and thus could harm our business.
There are numerous U.S. federal and state, as well as foreign, laws pertaining to healthcare fraud and abuse, including anti-kickback, false claims and physician transparency laws. Our business practices and relationships with providers are subject to scrutiny under these laws. We may also be subject to privacy and security regulation related to patient, customer, employee and other third-party information by both the federal government and the states and foreign jurisdictions in which we conduct our business. The healthcare laws and regulations that may affect our ability to operate include, but are not limited to:
•
the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce either the referral of an individual or furnishing or arranging for a good or service, for which payment may be made, in whole or in part, under federal healthcare programs, such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation. The U.S. government has interpreted this law broadly to apply to the marketing and sales activities of manufacturers. Moreover, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act. Violations of the federal Anti-Kickback Statute may result in civil monetary penalties. Civil penalties for such conduct can further be assessed under the federal False Claims Act. Violations can also result in criminal penalties. Similarly, violations can result in exclusion from participation in government healthcare programs, including Medicare and Medicaid. On November 20, 2020, the Department of Health and Human Services’ Office of the Inspector General, or OIG, finalized further modifications to the federal Anti-Kickback Statute. Under the final rules, the OIG added safe harbor protections under the Anti-Kickback Statute for certain coordinated care and value-based arrangements among clinicians, providers, and others. These rules (with exceptions) became effective January 19, 2021. We continue to evaluate these regulatory nuances in an attempt to maintain compliance with these evolving enforcement trends;
•
the federal civil and criminal false claims laws and civil monetary penalties laws, including the federal civil False Claims Act, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid or other federal healthcare programs that are false or fraudulent. These laws can apply to manufacturers who provide information on coverage, coding, and reimbursement of their products to persons who bill third-party payers. Private individuals can bring False Claims Act “qui tam” actions, on behalf of the government and such individuals, commonly known as “whistleblowers,” may share in amounts paid by the entity to the government in fines or settlement. When an entity is determined to have violated the federal civil False Claims Act, the government may impose civil fines and penalties, and exclude the entity from participation in Medicare, Medicaid and other federal healthcare programs;
•
the federal Civil Monetary Penalties Law, which prohibits, among other things, offering or transferring remuneration to a federal healthcare beneficiary that a person knows or should know is likely to influence the beneficiary’s decision to order or receive items or services reimbursable by the government from a particular provider or supplier;
•
the Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created additional federal criminal statutes that prohibit, among other things, executing a scheme to defraud any healthcare benefit program and making false statements relating to healthcare matters. Similar to the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation;
•
the federal Physician Sunshine Act under the ACA, which require certain applicable manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, or CHIP, to report annually to the DHHS Centers for Medicare and Medicaid Services (“CMS”), information related to payments and other transfers of value to physicians, which is defined broadly to include other healthcare providers and teaching hospitals, and applicable manufacturers and group purchasing organizations, to report annually ownership and investment interests held by physicians and their immediate family members. Applicable manufacturers are required to submit annual reports to CMS. Failure to submit required information may result in civil monetary penalties for all payments, transfers of value or ownership or investment interests that are not timely, accurately, and completely reported in an annual submission, and may result in liability under other federal laws or regulations. We have not, to date, submitted reports under the Physician Sunshine Act under the ACA;
•
HIPAA, as amended by the HITECH Act, and their respective implementing regulations, which impose requirements on certain covered healthcare providers, health plans and healthcare clearinghouses as well as their business associates that perform services for them that involve individually identifiable health information, relating to the privacy, security and transmission of individually identifiable health information without appropriate authorization, including mandatory contractual terms as well as directly applicable privacy and security standards and requirements. Failure to comply with the HIPAA privacy and security standards can result in civil monetary penalties, and, in certain circumstances, criminal penalties. State attorneys general can also bring a civil action to enjoin a HIPAA violation or to obtain statutory damages on behalf of residents of his or her state;
•
analogous state and foreign law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers or patients; state laws that require device companies to comply with the industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state laws that require device manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm customers, foreign and state laws, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts; privacy and data protection laws, including the EU General Data Protection Regulation, governing the privacy and security of personal data, including health information; federal government price reporting laws, which may require calculations and reporting of complex pricing metrics in an accurate and timely manner to government programs; and state laws related to insurance fraud in the case of claims involving private insurers; and
•
In addition, certain states have adopted new or modified privacy and security laws and regulations that may apply to our business. The California Consumer Privacy Act (“CCPA”) imposes obligations on businesses that process personal information of California residents. Among other things, the CCPA requires disclosures to such residents about the data collection, use and sharing practices of covered businesses; provides such individuals expanded rights to access and delete their personal information, and opt-out of certain sales or transfers of personal information; and provides such individuals with a private right of action and statutory damages for certain data breaches the California Privacy Rights Act (“CPRA”) significantly amends and expands existing CCPA requirements, includes, among other things, additional limitations on the sharing of personal information for cross-context behavioral advertising and on the use of “sensitive” personal information, the creation of a new correction right for California residents, and the establishment of a new agency to enforce California privacy laws. The enactment of the CCPA is prompting a wave of similar legislative developments in other states in the United States, which creates the potential for a patchwork of overlapping but different state laws. Many other states are currently reviewing or proposing the need for greater regulation of the collection, sharing, use and other processing of information related to individuals for marketing purposes or otherwise, and there remains increased interest at the federal level as well. Further, in order to comply with the varying state laws around data breaches, we must maintain adequate security measures, which require significant investments in resources and ongoing attention.
These laws and regulations, among other things, constrain our business, marketing and other promotional activities by limiting the kinds of financial arrangements, including sales programs, we may have with hospitals, healthcare practitioners or other potential purchasers of our products. Due to the breadth of these laws, the narrowness of statutory exceptions and regulatory safe harbors available, and the range of interpretations to which they are subject, it is possible that some of our current or future practices might be challenged under one or more of these laws.
To enforce compliance with the healthcare regulatory laws, certain enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry. Responding to investigations can be time and resource-consuming and can divert management’s attention from the business. Additionally, as a result of these investigations, healthcare providers and entities may have to agree to additional compliance and reporting requirements as part of a consent decree or corporate integrity agreement. Any such investigation or settlement could increase our costs or otherwise have an adverse effect on our business. Even an unsuccessful challenge or investigation into our practices could cause adverse publicity and be costly to respond to. If our operations are found to be in violation of any of the healthcare laws or regulations described above or any other healthcare regulations that apply to us, we may be subject to penalties, including administrative, civil and criminal penalties, damages, fines, exclusion from participation in government healthcare programs, such as Medicare and Medicaid, imprisonment, contractual damages, reputational harm, disgorgement and the curtailment or restructuring of our operations.
Inadequate funding for the FDA, the SEC and other government agencies, including from government shut downs, or other disruptions to these agencies’ operations, could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities, is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new product candidates to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years the U.S. government has shut down several times and certain regulatory agencies, such as the FDA and the SEC, have had to furlough critical FDA, SEC and other government employees and stop critical activities. If a prolonged government shutdown occurs, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, future government shutdowns could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Compliance with ever evolving federal, state, and foreign laws relating to handling of information about individuals involves significant expenditure and resources, and any failure by us or our vendors to comply may result in significant liability, negative publicity, and/or an erosion of trust, and could materially adversely affect our business, results of operations, and financial condition.
In the conduct of our business, we may at times process personal data, including health-related personal data. We also depend on a number of third-party vendors in relation to the operation of our business, a number of which process data on our behalf. We and our vendors are subject to a variety of federal, state and foreign data privacy laws, rules, regulations, industry standards and other requirements, including those that apply generally to the handling of information about individuals, and those that are specific to certain industries, sectors, contexts, or locations. These requirements, and their application, interpretation and amendment are constantly evolving and developing.
In the United States, numerous federal and state laws, including state data breach notification laws and state health information privacy laws, govern the collection, use, and disclosure and protection of health-related and other personal information. The Federal Trade Commission and state regulators enforce a variety of data privacy issues, such as promises made in privacy policies or failures to appropriately protect information about individuals, as unfair or deceptive acts or practices in or affecting commerce in violation of the Federal Trade Commission Act or similar state laws.
We are subject to the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, and regulations implemented thereunder (collectively, “HIPAA”). HIPAA imposes privacy, security and breach notification obligations on certain healthcare providers, health plans, and healthcare clearinghouses, known as covered entities, as well as their business associates that perform certain services that involve creating, receiving, maintaining or transmitting individually identifiable health information (“protected health information,” or “PHP”) for or on behalf of such covered entities, and their covered subcontractors. HIPAA requires covered entities and business associates to develop and maintain policies with respect to the protection of, use and disclosure of PHI, including the adoption of administrative, physical and technical safeguards to protect such information, and certain notification requirements in the event of a breach of unsecured PHI.
Additionally, under HIPAA, covered entities must report breaches of unsecured PHI to affected individuals without unreasonable delay, not to exceed 60 days following discovery of the breach by a covered entity or its agents. Notification also must be made to the U.S. Department of Health and Human Services Office for Civil Rights, or OCR, and, in certain circumstances involving large breaches, to the media. Business associates must report breaches of unsecured PHI to covered entities within 60 days of discovery of the breach by the business associate or its agents. A non-permitted use or disclosure of PHI is presumed to be a breach under HIPAA unless the covered entity or business associate establishes that there is a low probability the information has been compromised consistent with requirements enumerated in HIPAA.
Entities that are found to be in violation of HIPAA as the result of a breach of unsecured PHI, a complaint about privacy practices or an audit by the U.S. Department of Health and Human Services (“HHS”) may be subject to significant civil, criminal and administrative fines and penalties and/or additional reporting and oversight obligations if required to enter into a resolution agreement and corrective action plan with HHS to settle allegations of HIPAA non-compliance. HIPAA also authorizes state Attorneys General to file suit on behalf of their residents. Courts may award damages, costs and attorneys’ fees related to violations of HIPAA in such cases. While HIPAA does not create a private right of action allowing individuals to sue us in civil court for violations of HIPAA, its standards have been used as the basis for duty of care in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI.
Moreover, we may also be subject to U.S. federal rules, regulations and guidance concerning data security for medical devices, including guidance from the FDA. In addition, state privacy and security laws vary from state to state and, in some cases, can impose more restrictive requirements than U.S. federal law. Where state laws are more protective, we must comply with the stricter provisions. In addition to fines and penalties that may be imposed for failure to comply with state law, some states also provide for private rights of action to individuals for misuse of personal information.
Other jurisdictions have also adopted similar laws, regulations, guidelines and rules. For example, the EU also has laws and regulations dealing with the collection, use and processing of personal data originating from the EU, which are often more restrictive than those in the United States and which restrict transfers of personal data to the United States unless certain requirements are met. These obligations may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another and may conflict with other requirements or our practices. In addition, these rules have been subject to frequent scrutiny. For example, following a decision of the Court of Justice of the European Union in October 2015, the U.S. Safe Harbor Scheme (which had allowed the transfer of personal data to U.S. companies that had certified as members of the U.S. Safe Harbor Scheme) was declared invalid. In July 2016, the European Commission adopted the U.S.-EU Privacy Shield Framework which replaced the Safe Harbor Scheme. However, this Framework was itself invalidated in July 2020, before being replaced by the EU-U.S. Data Privacy Framework (“DPF”) remains in effect as of March 18, 2025, certain actions by the U.S. government in relation to aspects of the DPF mean that there are concerns that the DPF may be similarly invalidated. We rely on a mixture of mechanisms to transfer personal data from our EU business to the U.S. and could be impacted by changes in law as a result of a future review of these transfer mechanisms by regulators under the EU General Data Protection Regulation (GDPR) as well as current challenges to these mechanisms in the EU courts.
As well as imposing transfer restrictions, such EU laws impose significant compliance obligations. On May 25, 2018, the EU General Data Protection Regulation (“GDPR”) became applicable throughout the EU and, as a regulation, has direct effect in all member states. The GDPR was designed to harmonize data privacy laws across the EU and change the way organizations approach data privacy. The GDPR introduced new obligations and expanded the extraterritorial reach of the EU data protection regime. It applies to (i) organizations that process personal data in the context of an establishment in the EU (regardless of whether the processing takes place in the EU) and (ii) organizations outside the EU that offer goods or services to data subjects in the EU, or that monitor the behavior of EU data subjects. Compliance with the GDPR involves significant obligations, including requirements around accountability and transparency, contracting with service providers that process personal data, responding to data subjects’ rights requests within prescribed timelines, reporting of data breaches to data subjects and/or data protection or supervisory authorities, taking account of data protection as any new services are developed, and limiting the amount of personal data collected, stored or otherwise processed. The GDPR also significantly increased fines for non-compliance, up to €20,000,000 or up to 4% of the total worldwide annual turnover of the preceding financial year, whichever is higher. It also includes private rights of action. These obligations and restrictions have a significant impact on the ability to collect, analyze and transfer personal data, including in the context of clinical trials.
If privacy and data protection laws are interpreted or applied in a manner inconsistent with our policies and procedures, we may be fined or ordered to change our business practices in a manner that adversely impacts our operations. Compliance with these laws may also divert time and effort away from the management of our business and entail substantial expense. Any actual or perceived failure by us or the third parties with whom we work to comply with privacy or security laws, policies, legal obligations or industry standards, or any security incident that results in an effect on personal information, may result in governmental enforcement actions and investigations including by European Data Protection Authorities and U.S. federal and state regulatory authorities, fines and penalties, litigation and/or adverse publicity, including by consumer advocacy groups, and could cause our customers, their patients and other healthcare professionals to lose trust in us, which could harm our reputation and have a material adverse effect on our business, financial condition and results of operations.
Healthcare policy changes, including recently enacted legislation reforming the U.S. healthcare system, could harm our business, financial condition and results of operations.
In the United States, there have been and continue to be a number of legislative initiatives to contain healthcare costs. In March 2010, the ACA was enacted in the United States, which made a number of substantial changes in the way healthcare is financed by both governmental and private insurers. Among other ways in which it may affect our business, the ACA:
•
imposed an annual excise tax of 2.3% on any entity that manufactures or imports medical devices offered for sale in the United States, with limited exceptions (described in more detail below), although the effective rate paid may be lower. Through a series of legislative amendments, the tax was suspended for 2016 through 2019. Absent further legislative action, the device excise tax was to be reinstated on medical device sales starting January 1, 2020. The Further Consolidated Appropriations Act, 2020 H.R. 1865 (Pub.L.116-94), signed into law on December 20, 2019, repealed the medical device excise tax previously imposed by Internal Revenue Code section 4191. Prior to the repeal, the tax was on a 4-year moratorium. As a result of the repeal and the prior moratorium, sales of taxable medical devices after December 31, 2015, are not subject to the tax. We are unsure whether similar taxes could be reinstated in the future;
•
established the Patient-Centered Outcomes Research Institute to oversee and identify priorities in comparative clinical efficacy research in an effort to coordinate and develop such research;
•
implemented payment system reforms including the Bundled Payments for Care Improvement (“BPCI”) initiative to encourage hospitals, physicians and other providers to improve the coordination, quality and efficiency of certain healthcare services through bundled payment models; and
•
expanded the eligibility criteria for Medicaid programs (adoption varies by state).
We cannot pinpoint the full impact that the ACA has on our business. The taxes imposed by the ACA and the expansion in the government’s role in the U.S. healthcare industry may have, and may continue to, result in decreased profits, lower reimbursement by payors for our permanent birth control systems and women-specific medical devices, and/or reduce medical procedure volumes – all of which has a material adverse effect on our business, financial condition and results of operations. Several provisions of the ACA have faced implementation delays and challenges through judicial and legislative actions. While comprehensive repeal efforts have not succeeded, specific aspects of the aca have been modified. For instance, the Tax Cuts and Jobs Act of 2017 reduced the individual mandate penalty – the tax imposed on individuals without qualifying health coverage – to zero dollars ($0), effective January 1, 2019. Subsequently, in December 2018, a federal district court in Texas ruled that, without the penalty, the individual mandate was unconstitutional and deemed the entire ACA invalid. However, this decision was appealed, and the U.S. Supreme Court held that plaintiffs lacked standing to challenge the ACA’s minimum essential coverage provision, thereby leaving the ACA intact.
Further, on January 20, 2017, former President Trump signed an Executive Order directing federal agencies with authorities and responsibilities under the ACA to waive, defer, grant exemptions from, or delay the implementation of any provision of the ACA that would impose a fiscal burden on states or a cost, fee, tax, penalty or regulatory burden on individuals, healthcare providers, health insurers, or manufacturers of pharmaceuticals or medical devices. Subsequently, on October 13, 2017, former President Trump signed an Executive Order terminating the cost-sharing subsidies that reimburse insurers under the ACA. The Trump administration ceased cost-sharing reduction (“CSR”) payments to insurance companies, as required under the ACA, citing the absence of specific appropriations from Congress. Several state Attorneys General filed suit to stop the administration from terminating the subsidies, but their request for a restraining order was denied by a federal judge in California on October 25, 2017. On August 14, 2020, the U.S. Court of Appeals for the Federal Circuit ruled in two separate cases that the federal government is liable for the full amount of unpaid CSR for 2017 and prior years, while indicating that claims for 2018 and beyond would require further litigation. Additionally, on June 14, 2018, the U.S. Court of Appeals for the Federal Circuit ruled that the federal government was not obligated to pay over $12 billion in ACA risk corridor payments; however, on April 27, 2020, the United States Supreme Court reversed this decision, remanding the case to the U.S. Court of Federal Claims and concluding that the government has an obligation to pay these risk corridor payments. The implications of these regulatory developments on our business remain unclear, yet we continue to evaluate these evolving enforcement trends and nuances stemming therefrom.
In addition, since its enactment, the ACA has undergone numerous legislative changes and has faced significant legal challenges. By way of limited example, on August 2, 2011, the Budget Control Act of 2011 was signed into law, which introduced a 2% annual reduction in Medicare payments to providers starting April 1, 2013, with sequestration extended through fiscal year 2030. By way of further example, on January 2, 2013, the American Taxpayer Relief Act of 2012 was signed into law, which, among other things, reduced Medicare payments to various providers and extended the statute of limitations period for the government to recover overpayments to providers from three to five years. By way of further example, the Medicare Access and CHIP Reauthorization Act of 2015 (“MACRA”) enacted on April 16, 2015, repealed the formula by which Medicare made annual payment adjustments to physicians and replaced the formula with fixed annual payment updates and a new incentive payment system based on performance measures and participation in alternative payment models; the MACRA began in 2019 and continues to this day. of incentive payments scheduled to begin in 2019 that are based on various performance measures and healthcare practitioners’ participation in alternative payment models such as accountable care organizations. While it is difficult to determine the exact effects newer payment programs, such as MACRA, have on our business, we continue to monitor these legislative changes in an attempt to maintain compliance.
We expect additional state and federal healthcare policies and reform measures to be adopted in the future, any of which could limit reimbursement for healthcare products and services or otherwise result in reduced demand for our FemBloc system or additional pricing pressure and have a material adverse effect on our industry generally and on our customers. Any changes of, or uncertainty with respect to, future coverage or reimbursement rates could affect demand for our FemBloc system, which in turn could impact our ability to successfully commercialize our FemBloc system and could have a material adverse effect on our business, financial condition and results of operations.
Our business involves the use of hazardous materials, and we must comply with environmental laws and regulations, which may be expensive and restrict how we do business.
Our manufacturing activities involve the controlled storage, use and disposal of hazardous materials and are subject to federal, state, local and foreign laws and regulations governing the use, generation, manufacture, storage, handling and disposal of these hazardous materials. We currently carry no insurance specifically covering environmental claims relating to the use of hazardous materials. Although we believe the safety procedures for handling and disposing of these materials and waste products comply with the standards prescribed by these laws and regulations, we cannot eliminate the risk of accidental injury or contamination from the use, storage, handling or disposal of hazardous materials. In the event of an accident, state or federal or other applicable authorities may curtail our use of these materials and interrupt our business operations, which could adversely affect our business.
We are subject to anti-bribery, anti-corruption, and anti-money laundering laws, including the U.S. Foreign Corrupt Practices Act, as well as export control laws, customs laws, sanctions laws and other laws governing our operations. If we fail to comply with these laws, we could be subject to civil or criminal penalties, other remedial measures and legal expenses, which could adversely affect our business, results of operations and financial condition.
As we grow our international presence and global operations, we will be increasingly exposed to trade and economic sanctions and other restrictions imposed by the United States, the European Union and other governments and organizations. The U.S. Departments of Justice, Commerce, State and Treasury and other federal agencies and authorities have a broad range of civil and criminal penalties they may seek to impose against corporations and individuals for violations of economic sanctions laws, export control laws, the U.S. Foreign Corrupt Practices Act, (FCPA) and other federal statutes and regulations, including those established by the Office of Foreign Assets Control (OFAC). In addition, the U.K. Bribery Act of 2010, or the Bribery Act, prohibits both domestic and international bribery, as well as bribery across both private and public sectors. An organization that “fails to prevent bribery” by anyone associated with the organization can be charged under the Bribery Act unless the organization can establish the defense of having implemented “adequate procedures” to prevent bribery. Under these laws and regulations, as well as other anti-corruption laws, anti-money laundering laws, export control laws, customs laws, sanctions laws and other laws governing our operations, various government agencies may require export licenses, may seek to impose modifications to business practices, including cessation of business activities in sanctioned countries or with sanctioned persons or entities and modifications to compliance programs, which may increase compliance costs, and may subject us to fines, penalties and other sanctions. A violation of these laws or regulations would negatively affect our business, financial condition and results of operations.
We have implemented policies and procedures that are designed to ensure compliance by us and our directors, officers, employees, representatives, consultants and agents with the FCPA, OFAC restrictions, the Bribery Act and other export control, anti-corruption, anti-money-laundering and anti-terrorism laws and regulations. We cannot assure you, however, that our policies and procedures are sufficient or that directors, officers, employees, representatives, consultants and agents have not engaged and will not engage in conduct for which we may be held responsible, nor can we assure you that our business partners have not engaged and will not engage in conduct that could materially affect their ability to perform their contractual obligations to us or even result in our being held liable for such conduct. Violations of the FCPA, OFAC restrictions, the Bribery Act or other export control, anti-corruption, anti-money laundering and anti-terrorism laws or regulations may result in severe criminal or civil sanctions, and we may be subject to other liabilities, which could have a material adverse effect on our business, financial condition and results of operations.
We bear the risk of warranty claims on our products.
We bear the risk of warranty claims on our products. We may not be successful in claiming recovery under any warranty or indemnity provided to us by our suppliers or vendors in the event of a successful warranty claim against us by a customer or that any recovery from such vendor or supplier would be adequate. In addition, warranty claims brought by our customers related to third-party components may arise after our ability to bring corresponding warranty claims against such suppliers expires, which could result in costs to us.
Risks Related to Intellectual Property Matters
If we are unable to adequately protect our intellectual property rights, or if we are accused of infringing on the intellectual property rights of others, our competitive position could be harmed or we could be required to incur significant expenses to enforce or defend our rights.
Our commercial success will depend in part on our success in obtaining and maintaining issued patents, trademarks and other intellectual property rights in the United States and elsewhere and protecting our proprietary technology. If we do not adequately protect our intellectual property and proprietary technology, competitors may be able to use our technologies or the goodwill we have acquired in the marketplace and erode or negate any competitive advantage we may have, which could harm our business and ability to achieve profitability.
We own numerous issued patents and pending patent applications that relate to our intrauterine artificial insemination product, permanent birth control system and women-specific medical product solutions. As of December 31, 2025, we owned 41 issued U.S. patents and 187 issued foreign patents, 15 pending U.S. patent applications and 41 pending foreign patent applications. These issued patents, and any patents granted from such applications, are expected to expire between 2026 and 2046, without taking potential patent term extensions or adjustments into account. We believe that the patents expiring in 2026 are not material to our business.
We cannot provide any assurances that any of our patents have, or that any of our pending patent applications that mature into issued patents will include, claims with a scope sufficient to protect our intrauterine artificial insemination product, permanent birth control system, and women-specific medical product solutions, and any additional features we develop for our products. Other parties may have developed technologies that may be related or competitive to our intrauterine artificial insemination product, permanent birth control system, and women-specific medical product solutions, may have filed or may file patent applications and may have received or may receive patents that overlap or conflict with our patent applications, either by claiming the same methods or devices or by claiming subject matter that could dominate our patent position. The patent positions of medical device companies, including our patent position, may involve complex legal and factual questions, and, therefore, the scope, validity and enforceability of any patent claims that we may obtain cannot be predicted with certainty. Patents, if issued, may be challenged, deemed unenforceable, invalidated or circumvented. Proceedings challenging our patents could result in either loss of the patent or denial of the patent application or loss or reduction in the scope of one or more of the claims of the patent or patent application. In addition, such proceedings may be costly. Thus, any patents that we may own may not provide any protection against competitors. Furthermore, an adverse decision in an interference proceeding can result in a third party receiving the patent right sought by us, which in turn could affect our ability to commercialize our products.
Though an issued patent is presumed valid and enforceable, its issuance is not conclusive as to its validity or its enforceability and it may not provide us with adequate proprietary protection or competitive advantages against competitors with similar products. Competitors could purchase our intrauterine artificial insemination product, permanent birth control system, and women-specific medical product solutions and attempt to replicate some or all of the competitive advantages we derive from our development efforts, willfully infringe our intellectual property rights, design around our patents, or develop and obtain patent protection for more effective technologies, designs or methods. We may be unable to prevent the unauthorized disclosure or use of our technical knowledge or trade secrets by consultants, suppliers, vendors, former employees and current employees. The laws of some foreign countries do not protect our proprietary rights to the same extent as the laws of the United States, and we may encounter significant problems in protecting our proprietary rights in these countries.
Our ability to enforce our patent rights depends on our ability to detect infringement. It may be difficult to detect infringers who do not advertise the components that are used in their products. Moreover, it may be difficult or impossible to obtain evidence of infringement in a competitor’s or potential competitor’s product. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded if we were to prevail may not be commercially meaningful.
In addition, proceedings to enforce or defend our patents could put our patents at risk of being invalidated, held unenforceable or interpreted narrowly. Such proceedings could also provoke third parties to assert claims against us, including that some or all of the claims in one or more of our patents are invalid or otherwise unenforceable. If any of our patents covering our FemBloc system or FemaSeed product are invalidated or found unenforceable, or if a court found that valid, enforceable patents held by third parties covered one or more of our products, our competitive position could be harmed or we could be required to incur significant expenses to enforce or defend our rights.
The degree of future protection for our proprietary rights is uncertain, and we cannot ensure that:
•
any of our patents, or any of our pending patent applications, if issued, will include claims having a scope sufficient to protect our FemBloc system and FemaSeed product;
•
any of our pending patent applications will issue as patents;
•
we will be able to successfully commercialize our products on a substantial scale, if approved, before our relevant patents we may have expire;
•
we were the first to make the inventions covered by each of our patents and pending patent applications;
•
we were the first to file patent applications for these inventions;
•
others will not develop similar or alternative technologies that do not infringe our patents; any of our patents will be found to ultimately be valid and enforceable;
•
any patents issued to us will provide a basis for an exclusive market for our commercially viable products, will provide us with any competitive advantages or will not be challenged by third parties;
•
we will develop additional proprietary technologies or products that are separately patentable; or
•
our commercial activities or products will not infringe upon the patents of others.
We rely, in part, upon unpatented trade secrets, unpatented know-how and continuing technological innovation to develop and maintain our competitive position, which we seek to protect, in part, by confidentiality agreements with our employees and consultants. We also have agreements with our employees and consultants that obligate them to assign their inventions to us and have non-compete agreements with some, but not all, of our consultants. It is possible that technology relevant to our business will be independently developed by a person who is not a party to such an agreement. Furthermore, if the employees and consultants who are parties to these agreements breach or violate the terms of these agreements, we may not have adequate remedies for any such breach or violation, and we could lose our trade secrets through such breaches or violations. Further, our trade secrets could otherwise become known or be independently discovered by our competitors.
Obtaining and maintaining patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and applications are required to be paid to the United States Patent and Trademark Office, or USPTO, and various governmental patent agencies outside of the United States in several stages over the lifetimes of the patents and applications. The USPTO and various non-U.S. governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process and after a patent has issued. There are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Under the terms of some of our licenses, we do not have the ability to maintain or prosecute patents in the portfolio and must therefore rely on third parties to comply with these requirements.
Litigation or other proceedings or third-party claims of intellectual property infringement could require us to spend significant time and money and could prevent us from developing or selling our products or affect our stock price.
Our commercial success will depend in part on not infringing the patents or violating the other proprietary rights of others. Significant litigation regarding patent rights occurs in our industry. Our competitors in both the United States and abroad, many of which have substantially greater resources and have made substantial investments in patent portfolios and competing technologies, may have applied for or obtained or may in the future apply for and obtain, patents that will prevent, limit or otherwise interfere with our ability to make, use and sell our products. We do not always conduct independent reviews of patents issued to third parties. In addition, patent applications in the United States and elsewhere can be pending for many years before issuance, or unintentionally abandoned patents or applications can be revived, so there may be applications of others now pending or recently revived patents of which we are unaware. These applications may later result in issued patents, or the revival of previously abandoned patents, that will prevent, limit or otherwise interfere with our ability to make, use or sell our products. Third parties may, in the future, assert claims that we are employing their proprietary technology without authorization, including claims from competitors or from non-practicing entities that have no relevant product revenue and against whom our own patent portfolio may have no deterrent effect. As we continue to commercialize our products in their current or updated forms, launch new products and enter new markets, we expect competitors may claim that one or more of our products infringe their intellectual property rights as part of business strategies designed to impede our successful commercialization and entry into new markets. The large number of patents, the rapid rate of new patent applications and issuances, the complexities of the technology involved, and the uncertainty of litigation may increase the risk of business resources and management’s attention being diverted to patent litigation. We have, and we may in the future, receive letters or other threats or claims from third parties inviting us to take licenses under, or alleging that we infringe, their patents.
Moreover, we may become party to future adversarial proceedings regarding our patent portfolio or the patents of third parties. Such proceedings could include supplemental examination or contested post-grant proceedings such as review, reexamination, inter parties review, interference or derivation proceedings before the U.S. Patent and Trademark Office and challenges in U.S. District Court. Patents may be subjected to opposition, post-grant review or comparable proceedings lodged in various foreign, both national and regional, patent offices. The legal threshold for initiating litigation or contested proceedings may be low, so that even lawsuits or proceedings with a low probability of success might be initiated. Litigation and contested proceedings can also be expensive and time-consuming, and our adversaries in these proceedings may have the ability to dedicate substantially greater resources to prosecuting these legal actions than we can. We may also occasionally use these proceedings to challenge the patent rights of others. We cannot be certain that any particular challenge will be successful in limiting or eliminating the challenged patent rights of the third party.
Any lawsuits resulting from such allegations could subject us to significant liability for damages and invalidate our proprietary rights. Any potential intellectual property litigation also could force us to do one or more of the following:
•
stop making, selling or using products or technologies that allegedly infringe the asserted intellectual property;
•
lose the opportunity to license our technology to others or to collect royalty payments based upon successful protection and assertion of our intellectual property rights against others; incur significant legal expenses;
•
pay substantial damages or royalties to the party whose intellectual property rights we may be found to be infringing;
•
pay the attorney’s fees and costs of litigation to the party whose intellectual property rights we may be found to be infringing;
•
redesign those products that contain the allegedly infringing intellectual property, which could be costly, disruptive and infeasible; and
•
attempt to obtain a license to the relevant intellectual property from third parties, which may not be available on reasonable terms or at all, or from third parties who may attempt to license rights that they do not have.
Any litigation or claim against us, even those without merit, may cause us to incur substantial costs, and could place a significant strain on our financial resources, divert the attention of management from our core business and harm our reputation. If we are found to infringe the intellectual property rights of third parties, we could be required to pay substantial damages (which may be increased up to three times awarded damages) and/or substantial royalties and could be prevented from selling our products unless we obtain a license or are able to redesign our products to avoid infringement. Any such license may not be available on reasonable terms, if at all, and there can be no assurance that we would be able to redesign our products in a way that would not infringe the intellectual property rights of others. We could encounter delays in product introductions while we attempt to develop alternative methods or products. If we fail to obtain any required licenses or make any necessary changes to our products or technologies, we may have to withdraw existing products from the market or may be unable to commercialize one or more of our products.
In addition, we generally indemnify our customers with respect to infringement by our products of the proprietary rights of third parties. Third parties may assert infringement claims against our customers. These claims may require us to initiate or defend protracted and costly litigation on behalf of our customers, regardless of the merits of these claims. If any of these claims succeed or settle, we may be forced to pay damages or settlement payments on behalf of our customers or may be required to obtain licenses for the products they use. If we cannot obtain all necessary licenses on commercially reasonable terms, our customers may be forced to stop using our products.
Third parties may assert ownership or commercial rights to inventions we develop.
Third parties may in the future make claims challenging the inventorship or ownership of our intellectual property. We may face claims by third parties that our agreements with employees, contractors or consultants obligating them to assign intellectual property to us are ineffective or in conflict with prior or competing contractual obligations of assignment, which could result in ownership disputes regarding intellectual property we have developed or will develop and interfere with our ability to capture the commercial value of such intellectual property. Litigation may be necessary to resolve an ownership dispute, and if we are not successful, we may be precluded from using certain intellectual property or may lose our exclusive rights in that intellectual property. Either outcome could harm our business and competitive position. If we are unable to protect the confidentiality of our trade secrets, our business and competitive position could be harmed.
In addition to patent protection, we also rely upon copyright and trade secret protection, as well as non-disclosure agreements and invention assignment agreements with our employees, consultants and third parties, to protect our confidential and proprietary information. In addition to contractual measures, we try to protect the confidential nature of our proprietary information using commonly accepted physical and technological security measures. Such measures may not, for example, in the case of misappropriation of a trade secret by an employee or third party with authorized access, provide adequate protection for our proprietary information. Our security measures may not prevent an employee or consultant from misappropriating our trade secrets and providing them to a competitor, and recourse we take against such misconduct may not provide an adequate remedy to protect our interests fully. Unauthorized parties may also attempt to copy or reverse engineer certain aspects of our products that we consider proprietary. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret can be difficult, expensive and time-consuming, and the outcome is unpredictable. Even though we use commonly accepted security measures, trade secret violations are often a matter of state law, and the criteria for protection of trade secrets can vary among different jurisdictions. In addition, trade secrets may be independently developed by others in a manner that could prevent legal recourse by us. If any of our confidential or proprietary information, such as our trade secrets, were to be disclosed or misappropriated, or if any such information was independently developed by a competitor, our business and competitive position could be harmed.
We may be unable to enforce our intellectual property rights throughout the world.
The laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. This could make it difficult for us to stop infringement of our foreign patents, if obtained, or the misappropriation of our other intellectual property rights. For example, some foreign countries have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, some countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries.
Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business. Accordingly, our efforts to protect our intellectual property rights in such countries may be inadequate. In addition, changes in the law and legal decisions by courts in the United States and foreign countries may affect our ability to obtain adequate protection for our technology and the enforcement of our intellectual property.
Third parties may assert that our employees or consultants have wrongfully used or disclosed confidential information or misappropriated trade secrets.
We employ individuals who previously worked with other companies, including our competitors or potential competitors. Although we try to ensure that our employees and consultants do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise used or disclosed intellectual property or personal data, including trade secrets or other proprietary information, of a former employer or other third party. Litigation may be necessary to defend against these claims. If we fail in defending any such claims or settling those claims, in addition to paying monetary damages or a settlement payment, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
U.S. patent laws could diminish the value of patents in general and may limit our ability to obtain, defend and/or enforce our patents.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. The Leahy-Smith America Invents Act, or the Leahy-Smith Act, includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted and also affect patent litigation. The U.S. Patent and Trademark Office recently developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first to file provisions, which became effective on March 16, 2013. The first to file provisions limit the rights of an inventor to patent an invention if not the first to file an application for patenting that invention, even if such invention was the first invention. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business.
However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the enforcement and defense of our issued patents. For example, the Leahy-Smith Act provides that an administrative tribunal known as the Patent Trial and Appeals Board, or PTAB, provides a venue for challenging the validity of patents at a cost that is much lower than district court litigation and on timelines that are much faster. Although it is not clear what, if any, long-term impact the PTAB proceedings will have on the operation of our business, the initial results of patent challenge proceedings before the PTAB since its inception in 2013 have resulted in the invalidation of many U.S. patent claims. The availability of the PTAB as a lower-cost, faster and potentially more potent tribunal for challenging patents could increase the likelihood that our own patents will be challenged, thereby increasing the uncertainties and costs of maintaining and enforcing them.
Patent terms may be inadequate to protect our competitive position on our products for an adequate amount of time.
Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire shortly after such candidates are commercialized. We expect to seek extensions of patent terms in the United States and, if available, in other countries where we are prosecuting patents. In the United States, the Drug Price Competition and Patent Term Restoration Act of 1984 permits a patent term extension of up to five years beyond the normal expiration of the patent, which is limited to the approved indication (or any additional indications approved during the period of extension). However, the applicable authorities, including the FDA and the USPTO in the United States, and any equivalent regulatory authority in other countries, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request.
If our trademarks or trade names are denied by regulatory authorities or are not adequately protected, we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
We rely on our trademarks and trade names to distinguish our products from the products of our competitors and have registered or applied to register many of these trademarks. We cannot assure you that our trademark applications will be approved in a timely manner or at all. During the trademark registration process, we may receive office actions from the USPTO objecting to the registration of our trademark. Although we would be given an opportunity to respond to those objections, we may be unable to overcome them. Our registered or unregistered trademarks or trade names may be denied by other regulatory authorities or challenged, infringed, circumvented or declared generic or determined to be infringing on other marks. We may be unable to use these trademarks and trade names or protect our rights to these trademarks and trade names, which we need to build name recognition by potential partners or customers in our markets of interest. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected. If other entities use trademarks similar to ours in different jurisdictions, or have senior rights to ours, it could interfere with our use of our current trademarks throughout the world. If we are required to use an alternative trademark, any goodwill and recognition that we have built for these trademarks would be lost. If any party infringes on any of the trademarks on which we rely, enforcing those trademarks may be difficult, costly, time-consuming and ultimately unsuccessful.
Risks Related to Our Common Stock
We are a “smaller reporting company” and an “emerging growth company” and the reduced disclosure requirements applicable to “smaller reporting companies” may make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. We will remain an emerging growth company until the earlier of (i) the last day of the fiscal year (a) following the fifth anniversary of the completion of our IPO, (b) in which we have total annual gross revenue of at least $1.07 billion or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeded $700.0 million as of our most recently completed second fiscal quarter and (ii) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.
An emerging growth company may take advantage of specified reduced reporting requirements and other burdens that are otherwise applicable generally to public companies. These provisions include:
•
being permitted to present only two years of audited financial statements and only two years of related management’s discussion and analysis of financial condition and results of operations;
•
not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act;
•
an exemption from compliance with any new requirements adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotations;
•
reduced disclosure obligations regarding executive compensation in our periodic reports, proxy statements and registration statements; and
•
exemptions from the requirement to hold a nonbinding advisory vote on executive compensation and to obtain stockholder approval of any golden parachute payments not previously approved.
We have elected to take advantage of certain of the reduced disclosure obligations and may elect to take advantage of other reduced reporting requirements in the future. As a result, the information that we provide to our investors may be different from the information you might receive from other public reporting companies that are not emerging growth companies in which you hold equity interests. The JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards applicable to public companies until those standards would otherwise apply to private companies. We have elected not to take advantage of such extended transition period, which means that we will adopt a new standard when it is issued or revised.
We are also a “smaller reporting company,” meaning that the market value of our shares held by non-affiliates is less than $700.0 million and our annual revenue was less than $100.0 million during the most recently completed fiscal year, or if the market value of our shares held by non-affiliates is less than $250.0 million. If we are a smaller reporting company at the time we cease to be an emerging growth company, we may continue to rely on exemptions from certain disclosure requirements that are available to smaller reporting companies. Specifically, as a smaller reporting company, we may choose to present only the two most recent fiscal years of audited financial statements in our Annual Report on Form 10-K and have reduced disclosure obligations regarding executive compensation, and, similar to emerging growth companies, if we are a smaller reporting company with less than $100 million in annual revenue, we would not be required to obtain an attestation report.
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in the amended and restated certificate of incorporation and our amended and restated bylaws may delay or prevent an acquisition of us or a change in our management. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors. Because our board of directors is responsible for appointing the members of our management team, these provisions could in turn affect any attempt by our stockholders to replace current members of our management team. These provisions include:
•
a prohibition on actions by our stockholders by written consent;
•
advance notice requirements for election to our board of directors and for proposing matters that can be acted upon at stockholder meetings;
•
a requirement that directors may only be removed “for cause”;
•
a requirement that only the board of directors may change the number of directors and fill vacancies on the board;
•
division of our board of directors into three classes, serving staggered terms of three years each; and
•
the authority of the board of directors to issue preferred stock with such terms as the board of directors may determine.
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, as amended, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner. These provisions would apply even if the proposed merger or acquisition could be considered beneficial by some stockholders.
We incur significant costs as a result of being a public company, which may adversely affect our business, financial condition and results of operations.
We incur costs associated with corporate governance requirements that are applicable to us as a public company, including rules and regulations of the SEC, under the Sarbanes-Oxley Act, the Dodd-Frank Wall Street Reform and Consumer Protection Act of 2010, and the Exchange Act, as well as the rules of Nasdaq. These rules and regulations have significantly increased our accounting, legal and financial compliance costs and have made some activities more time-consuming. These rules and regulations have made it more expensive for us to maintain directors’ and officers’ liability insurance. As a result, it may be more difficult for us to attract and retain qualified persons to serve on our board of directors or as executive officers. Accordingly, the increases in costs incurred as a result of being a publicly traded company may adversely affect our business, financial condition and results of operations.
We are obligated to develop and maintain proper and effective internal controls over financial reporting and any failure to maintain the adequacy of these internal controls may adversely affect investor confidence in our Company and, as a result, the value of our common stock.
To comply with the requirements of being a public company, we have undertaken various actions, including implementing new internal controls and procedures and hiring new accounting or internal audit staff. The Sarbanes-Oxley Act requires that we maintain effective disclosure controls and procedures and internal control over financial reporting. We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure that information required to be disclosed by us in the reports that we file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, and that information required to be disclosed in reports under the Exchange Act is accumulated and communicated to our principal executive and financial officers. Our current controls and any new controls that we develop may become inadequate and weaknesses in our internal control over financial reporting may be discovered in the future. Any failure to develop or maintain effective controls when we become subject to this requirement could negatively affect the results of periodic management evaluations and annual independent registered public accounting firm attestation reports regarding the effectiveness of our internal control over financial reporting that we may be required to include in our periodic reports we will file with the SEC under Section 404 of the Sarbanes-Oxley Act, harm our operating results, cause us to fail to meet our reporting obligations or result in a restatement of our prior period financial statements. In the event that we are not able to demonstrate compliance with the Sarbanes-Oxley Act, that our internal control over financial reporting is perceived as inadequate or that we are unable to produce timely or accurate financial statements, investors may lose confidence in our operating results and the price of our common stock could decline. In addition, if we are unable to continue to meet these requirements, we may be unable to remain listed on Nasdaq.
Our disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
We are subject to the periodic reporting requirements of the Exchange Act. We designed our disclosure controls and procedures to provide reasonable assurance that information we must disclose in reports we file or submit under the Exchange Act is accumulated and communicated to management, and recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls and procedures, no matter how well-conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.
These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements due to error or fraud may occur and not be detected.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware will be the exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the exclusive forum for state law claims for (i) any derivative action or proceeding brought on our behalf, (ii) any action asserting a claim of breach of a fiduciary duty or other wrongdoing by any of our directors, officers, employees or agents to us or our stockholders, (iii) any action asserting a claim arising pursuant to any provision of the DGCL or our amended and restated certificate of incorporation or amended and restated bylaws or (iv) any action asserting a claim governed by the internal affairs doctrine , or the Delaware Forum Provision. The Delaware Forum Provision does not apply to any causes of action arising under the Securities Act or the Exchange Act. Our amended and restated bylaws further provides that unless we consent in writing to the selection of an alternative forum, the United States District Court for the District of Delaware will be the sole and exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act, or the Federal Forum Provision, as the Company is incorporated in the State of Delaware. In addition, our amended and restated bylaws provides that any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock will be deemed to have notice of and consented to the Delaware Forum Provision and the Federal Forum Provision; provided, however, that stockholders cannot and will not be deemed to have waived our compliance with the U.S. federal securities laws and the rules and regulations thereunder.
We believe the Delaware Forum Provision and the Federal Forum Provision benefits us by providing increased consistency in the application of Delaware law by chancellors particularly experienced in resolving corporate disputes, efficient administration of cases on a more expedited schedule relative to other forums and protection against the burdens of multi-forum litigation. However, this provision may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and other employees and also may impose additional litigation costs on stockholders in pursuing any such claims. In addition, while the Delaware Supreme Court ruled in March 2020 that federal forum selection provisions purporting to require claims under the Securities Act be brought in federal court are “facially valid” under Delaware law, there is uncertainty as to whether other courts will enforce our Federal Forum Provision. If the Federal Forum Provision is found to be unenforceable, we may incur additional costs associated with resolving such matters. The Federal Forum Provision may also impose additional litigation costs on stockholders who assert that the provision is not enforceable or invalid. The Court of Chancery of the State of Delaware and the United States District Court for the District of Delaware may also reach different judgments or results than would other courts, including courts where a stockholder considering an action may be located or would otherwise choose to bring the action, and such judgments may be more or less favorable to us than our stockholders.
Because we do not anticipate paying any cash dividends on our capital stock in the foreseeable future, capital appreciation, if any, will be your sole source of gain.
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. As a result, capital appreciation, if any, of our common stock will be your sole source of gain for the foreseeable future.
General Risk Factors
Adverse developments affecting the financial services industry could adversely affect our current and projected business operations and our financial condition and results of operations.
Adverse developments that affect financial institutions, such as events involving liquidity that are rumored or actual, have in the past and may in the future lead to bank failures and market-wide liquidity problems. The U.S. Department of Treasury, FDIC and Federal Reserve Board have announced a program to provide up to $25 billion of loans to financial institutions secured by certain of such government securities held by financial institutions to mitigate the risk of potential losses on the sale of such instruments, widespread demands for customer withdrawals or other liquidity needs of financial institutions for immediately liquidity may exceed the capacity of such program. There is no guarantee, however, that the U.S. Department of Treasury, FDIC and Federal Reserve Board will provide access to uninsured funds in the future in the event of the closure of other banks or financial institutions, or that they would do so in a timely fashion.
Uncertainty remains over liquidity concerns in the broader financial services industry, and our business, our business partners, or industry as a whole may be adversely impacted in ways that we cannot predict at this time.
Although we assess our banking relationships as we believe necessary or appropriate, our access to cash in amounts adequate to finance or capitalize our current and projected future business operations could be significantly impaired by factors that affect the financial institutions with which we have banking relationships, and in turn, us. These factors could include, among others, events such as liquidity constraints or failures, the ability to perform obligations under various types of financial, credit or liquidity agreements or arrangements, disruptions or instability in the financial services industry or financial markets, or concerns or negative expectations about the prospects for companies in the financial services industry. These factors could also include factors involving financial markets or the financial services industry generally. The results of events or concerns that involve one or more of these factors could include a variety of material and adverse impacts on our current and projected business operations and our financial condition and results of operations. These could include, but may not be limited to, delayed access to deposits or other financial assets or the uninsured loss of deposits or other financial assets; or termination of cash management arrangements and/or delays in accessing or actual loss of funds subject to cash management arrangements.
In addition, widespread investor concerns regarding the U.S. or international financial systems could result in less favorable commercial financing terms, including higher interest rates or costs and tighter financial and operating covenants, or systemic limitations on access to credit and liquidity sources, thereby making it more difficult for us to acquire financing on acceptable terms or at all. Any decline in available funding or access to our cash and liquidity resources could, among other risks, adversely impact our ability to meet our operating expenses, financial obligations or fulfill our other obligations, result in breaches of our financial and/or contractual obligations or result in violations of federal or state wage and hour laws. Any of these impacts, or any other impacts resulting from the factors described above or other related or similar factors not described above, could have material adverse impacts on our liquidity and our current and/or projected business operations and financial condition and results of operations.
In addition, a critical vendor or business partner could be adversely affected by any of the liquidity or other risks that are described above as factors, which in turn, could have a material adverse effect on our current and/or projected business operations and results of operations and financial condition. Any business partner or supplier bankruptcy or insolvency, or any breach or default by a business partner or supplier, or the loss of any significant business partner or supplier relationships, could result in material adverse impacts on our current and/or projected business operations and financial condition.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. Global economic and business activities continue to face widespread uncertainties, and global credit and financial markets have experienced extreme volatility and disruptions in the past several years, including severely diminished liquidity and credit availability, rising inflation and monetary supply shifts, rising interest rates, labor shortages, declines in consumer confidence, declines in economic growth, increases in unemployment rates, recession risks, and uncertainty about economic and geopolitical stability. A severe or prolonged economic downturn, such as the global financial crisis, could result in a variety of risks to our business, including weakened demand for our products, and our ability to raise additional capital when needed on acceptable terms, if at all. Supply chain disruptions have lengthened our suppliers’ timelines and increased costs. The occurrence of, or acceleration or exacerbation of, any of the foregoing could harm our business and we cannot anticipate all of the ways in which the economic climate and financial market conditions could adversely affect our business.
Our internal computer systems, or those of any of our Contract Research Organizations (“CROs”), manufacturers, other contractors, consultants, existing or future collaborators, may fail or suffer security or data privacy breaches or other unauthorized or improper access to, use of or destruction of our proprietary and confidential data, employee data or personal data, which could result in additional costs, significant liabilities, harm to our reputation and material disruption of our operations.
Despite the implementation of security measures, our internal computer systems and those of our current and any future CROs, other contractors, consultants, potential future collaborators and other third-party service providers are vulnerable to damage from various methods, including cybersecurity attacks, breaches, intentional or accidental mistakes or errors, attacks using artificial intelligence, or other technological failures, which can include, among other things, computer viruses, unauthorized access attempts, including third parties gaining access to systems using stolen or inferred credentials, denial-of-service attacks, phishing attempts, service disruptions, natural disasters, fire, terrorism, war and telecommunication and electrical failures. As the cyber-threat landscape evolves, these attacks are growing in frequency, sophistication and intensity, and are becoming increasingly difficult to detect. If such an event were to occur and cause interruptions in our operations or result in the unauthorized acquisition of or access to personally identifiable information or individually identifiable health information (violating certain privacy laws such as HIPAA, or HITECH Act, the CCPA and GDPR), it could result in a material disruption of our U.S. product candidate development programs and our business operations, and we could incur significant liabilities. Some of the federal, state and foreign government requirements include obligations of companies to notify individuals of security breaches involving particular personally identifiable information, which could result from breaches experienced by us or by our vendors or contractors. Notifications and follow-up actions related to a security breach could impact our reputation and cause us to incur significant costs, including legal expenses and remediation costs. For example, the loss of clinical trial data from completed, ongoing or future clinical trials involving our U.S. product candidate could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the lost data.
To the extent that any disruption or security breach were to result in a loss of, or damage to, our data, or inappropriate disclosure of confidential or proprietary information, we could be exposed to litigation and governmental investigations, the further development and commercialization of our U.S. product candidate could be delayed, and we could be subject to significant fines or penalties for any noncompliance with certain state, federal or international privacy and security laws.
Our insurance policies may not be adequate to compensate us for the potential losses arising from any such disruption, failure or security breach. In addition, such insurance may not be available to us in the future on economically reasonable terms, or at all. Further, our insurance may not cover all claims made against us and could have high deductibles in any event, and defending a suit, regardless of its merit, could be costly and divert management attention.
The estimates of market opportunity and forecasts of market growth that we provide may prove to be inaccurate, and even if the markets in which we compete achieve the forecasted growth, our business may not grow at similar rates, or at all.
The market opportunity estimates and growth forecasts we provide are subject to significant uncertainty and are based on assumptions and estimates which may not prove to be accurate. The estimates and forecasts relating to size and expected growth of our target market may prove to be inaccurate. Even if the markets in which we compete meet the size estimates and growth forecasts, our business may not grow at similar rates, or at all. Our growth is subject to many factors, including our success in implementing our business strategy, which is subject to many risks and uncertainties.
Our employees, independent contractors, consultants, commercial partners, collaborators and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
We are exposed to the risk of employee fraud or other illegal activity by our employees, independent contractors, consultants, commercial partners, collaborators, service providers and vendors. Misconduct by these parties could include intentional, reckless and/or negligent conduct that fails to comply with the laws of the FDA and other similar foreign regulatory bodies, provide true, complete and accurate information to the FDA and other similar foreign regulatory bodies, comply with manufacturing standards we have established, comply with healthcare fraud and abuse laws in the United States and similar foreign fraudulent misconduct laws, or report financial information or data accurately or to disclose unauthorized activities to us. As we begin commercializing our products and if we obtain FDA approval of our U.S. product candidate in the United States, our potential exposure under such laws will increase significantly, and our costs associated with compliance with such laws will also increase. These laws may impact, among other things, our current activities with principal investigators and research patients, as well as proposed and future sales, marketing and education programs. We have a code of business conduct and ethics and maintain a training program, but it is not always possible to identify and deter misconduct by our employees, independent contractors, consultants, commercial partners and vendors, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any actions are instituted against us and we are not successful in defending ourselves or asserting our rights, those actions could result in the imposition of civil, criminal and administrative penalties, damages, monetary fines, imprisonment, disgorgement, possible exclusion from participation in government healthcare programs, additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of noncompliance with these laws, contractual damages, reputational harm, diminished profits and future earnings and the curtailment of our operations.
Our ability to use our net operating losses and research and development credit carryforwards to offset future taxable income may be subject to certain limitations.
In general, under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, or the Code, a corporation that undergoes an “ownership change,” generally defined as a more than 50 percentage points increase in ownership by value in its equity ownership by certain shareholders over their lowest ownership percentage within a rolling three-year period, is subject to limitations on its ability to utilize its pre-change net operating losses, or NOLs, and its research and development credit carryforwards to offset future taxable income. Certain substantial changes in our ownership between February 2004 to date will more likely than not limit our ability to utilize the amount of our existing NOLs and research and development credit carryforwards, and if we undergo any further ownership change, our ability to utilize NOLs and research and development credit carryforwards could be further limited by Sections 382 and 383 of the Code. In addition, our ability to deduct net interest expense may be limited if we have insufficient taxable income for the year during which the interest is incurred, and any carryovers of such disallowed interest would be subject to the limitation rules similar to those applicable to NOLs and other attributes. Future changes in our stock ownership, some of which might be beyond our control, could result in an ownership change under Section 382 of the Code. For these reasons, in the event we experience a change of control, we may not be able to utilize a material portion of the NOLs, research and development credit carryforwards or disallowed interest expense carryovers, even if we attain profitability.
An active trading market for our common stock may not be sustained.
We cannot assure you that an active trading market for our common stock will be sustained. The lack of an active trading market may impair the value of your shares and your ability to sell your shares at the time you wish to sell them. An inactive trading market may also impair our ability to raise capital by selling shares of our common stock and enter into strategic partnerships or acquire other complementary products, technologies or businesses by using shares of our common stock as consideration. Furthermore, there can be no guarantee that we will continue to satisfy the continued listing standards of Nasdaq. If we fail to satisfy the continued listing standards, we could be de-listed, which would have a negative effect on the price of our common stock.
We expect that the price of our common stock will fluctuate substantially.
The market price of our common stock has been highly volatile and may fluctuate substantially due to many factors, some of which are beyond our control, including:
•
announcements of U.S. regulatory approval or disapproval of our FemBloc system or the FDA’s decision to grant or decline any future approvals or clearances for enhancements to our products;
•
announcements of international regulatory approval or disapproval of our FemBloc system or the foreign regulatory body or notified body’s decision to grant or decline any future approvals for enhancements to our products;
•
adverse results from or delays in clinical pivotal trial of our FemBloc system;
•
unanticipated safety concerns related to the use of our FemBloc system;
•
unanticipated safety concerns related to the use of our FemaSeed product or other products;
•
FDA or other U.S. or foreign regulatory or legal actions or changes affecting us or our industry;
•
our ability to develop, obtain regulatory clearance or approval for, and market new and enhanced medical products on a timely basis;
•
any voluntary or mandated product recalls;
•
adverse developments concerning our suppliers or any future strategic partnerships;
•
the volume and timing of sales of our products;
•
the introduction of new products or product enhancements by us or others in our industry;
•
disputes or other developments with respect to our or others’ intellectual property rights;
•
product liability claims or other litigation;
•
quarterly variations in our results of operations or those of others in our industry;
•
media exposure of our products or of those of others in our industry;
•
changes in governmental regulations or in reimbursement;
•
changes in earnings estimates or recommendations by securities analysts;
•
changes in financial estimates or guidance, including our ability to meet our future revenue and operating profit or loss estimates or guidance;
•
the public’s reaction to our earnings releases, other public announcements and filings with the SEC;
•
sales of substantial amounts of our stock by directors, officers or significant stockholders, or the expectation that such sales might occur;
•
operating and stock performance of other companies that investors deem comparable to us and overall performance of the equity markets;
•
additions or departures of key personnel;
•
changes in our capital structure, such as future issuances of securities and the incurrence of debt;
•
general market conditions and other factors, including factors unrelated to our operating performance or the operating performance of our competitors; and
•
other factors described in this “Risk Factors” section.
In recent years, the stock markets generally have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies. Broad market and industry factors may significantly affect the market price of our common stock, regardless of our actual operating performance.
In addition, in the past, class action litigation has often been instituted against companies whose securities have experienced periods of volatility in market price. Securities litigation brought against us following volatility in our stock price, regardless of the merit or ultimate results of such litigation, could result in substantial costs, which would hurt our financial condition and operating results and divert management’s attention and resources from our business.
Securities analysts may not continue to publish favorable research or reports about our business or may publish no information at all, which could cause our stock price or trading volume to decline.
The trading market will be influenced to some extent by the research and reports that industry or financial analysts publish about us and our business. We do not control these analysts. Our research coverage may be inconsistent and not as robust as larger and more established public companies and, as we begin to establish a commercialization operation, analysts may be unable to accurately forecast our results and could make it more likely that we fail to meet their estimates. If any of the analysts who cover us provide inaccurate or unfavorable research or issue an adverse opinion regarding our stock price, our stock price could decline. If one or more of these analysts cease coverage of us or fail to publish reports covering us regularly, we could lose visibility in the market, which in turn could cause our stock price or trading volume to decline and result in the loss of all or a part of your investment in us.
Item 1B.
Unresolved Staff Comments.
None.
Risk Management and Strategy
We employ a cybersecurity program which we believe is appropriate for the potential of cybersecurity threats faced by us and is built upon a set of policies, procedures and standards supported by training and education. Our information systems employ local and cloud storage and recovery services from various third parties. We have established processes to assess, identify, and manage risks from cybersecurity threats as part of our broader enterprise-wide risk management system and processes and engaged third-party consultants to assess and gather threat intelligence and cyber risk trends and conduct assessments and testing of our cybersecurity procedures and protections, which is reported to and overseen by our Board of Directors through our Audit Committee, along with our executive management.
Our cybersecurity program focuses on all areas of our business, including cloud-based environments, devices used by employees and contractors, facilities, networks, applications, vendors, disaster recovery, business continuity and controls and safeguards enabled through business processes and tools. We continuously monitor for unauthorized access to our information technology systems and identify potential security threats through various automated detection solutions. To protect the security of our information infrastructure and protect our systems and information from unauthorized access, we draw on the knowledge and insights of an external information technology consultant who acts as our primary IT administrator and employ an array of third-party tools and technologies. We also maintain cybersecurity insurance to cover us for costs and expenses we may incur due to a cybersecurity incident.
As of the date of this Annual Report, we have not encountered any risks from cybersecurity threats that have materially affected or are reasonably likely to materially affect the Company, including its business strategy, results of operations, or financial condition. For more information on our cybersecurity related risks, see “General Risks Factors” included elsewhere in this Annual Report on Form 10-K.
Governance and Role of Management and the Board
Our Chief Financial Officer leads our cybersecurity program and reports directly, with respect to cybersecurity incidents, to the Board of Directors and the Audit Committee, which is the principal committee charged with the Board’s risk management oversight, including cybersecurity risks. The Chief Financial Officer briefs the Board and Audit Committee on the effectiveness of our cybersecurity program quarterly. The Chief Financial Officer oversees the operation of our cybersecurity program and has over 30 years of executive experience overseeing risk management and internal controls. The Chief Financial Officer is informed about and monitors the prevention, detection, mitigation, and remediation of cybersecurity incidents through the Financial Officer’s oversight of the Company’s information technology function. We have not experienced any cybersecurity threats or incidents that have had a material impact on our business to date.
We lease our facility in Suwanee, Georgia which, together with our research and development, controlled environment room and office space, currently totals approximately 41,000 square feet. As of December 31, 2025, our monthly rent payment was $52,614 and is subject to increases on an annual basis. Our lease expires in April 2029, and we have the option to extend the term for one consecutive term of five years, subject to certain terms and conditions.
We are not currently a party to any legal proceedings the outcome of which we believe, if determined adversely to us, would individually or in the aggregate have a material adverse effect on our business, operating results or financial condition. We have received, and may from time to time receive, letters from third parties alleging patent infringement, violation of employment practices or trademark infringement, and we may in the future participate in litigation to defend ourselves. We cannot predict the results of any such disputes, and despite the potential outcomes, the existence thereof may have an adverse material impact on us due to diversion of management time and attention as well as the financial costs related to resolving such disputes.
Not applicable
Item 5.
Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
MARKET FOR COMMON EQUITY
Market Information
Our common stock is listed on the NASDAQ Capital Market under the ticker symbol FEMY.
On March 20, 2026
there were 147 holders of record of our common stock. This number does not
include stockholders for whom shares were held in a “nominee” or “street” name.
We have not declared or paid any cash dividends on our common stock and do not anticipate declaring or paying any cash dividends in the foreseeable future. We currently expect to retain future earnings, if any, for the continued development of our business.
Our transfer agent and registrar is Broadridge Investor Communication Solutions, Inc.
Recent Sales of Unregistered Securities
None.
Equity Compensation Plan Information
The information required by Item 5 of Form 10-K regarding equity compensation plans is incorporated herein by reference to Item 11. of Part III of this Annual Report on Form 10-K.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
Other Information
Not applicable
Item 7.
Management’s Discussion and Analysis of Financial Condition and Results of Operations.
We are a leading biomedical innovator, addressing significant unmet needs in women’s health worldwide, with a broad patent-protected portfolio of disruptive, accessible, in-office therapeutic and diagnostic products. The Company is a U.S. manufacturer that has received global regulatory approvals for its product portfolio, which is currently being commercialized in the U.S. and key international markets. FemaSeed® Intratubal Insemination, a groundbreaking first-step infertility treatment delivering sperm directly to the site of conception, is U.S. FDA-cleared and approved in Europe, United Kingdom (UK), Canada, Israel, Australia and New Zealand. A peer-reviewed publication of positive data from its pivotal clinical trial of FemaSeed demonstrated effectiveness and safety with high satisfaction from both patients and practitioners. FemSperm®, which includes setup, preparation and analysis kits designed to expand our infertility portfolio and, for the first time, enables gynecologists to perform in-office sperm preparation and analysis for use with FemaSeed. FemVue®, a companion diagnostic for fallopian tube assessment via ultrasound, is U.S. FDA-cleared and approved in Europe, UK, Canada, Japan, Israel, Australia and New Zealand. FemVue Controlled is a US FDA-cleared diagnostic device and is the next-generation design integrating features of FemVue and FemChec® technologies into a single platform, enabling multiple clinical uses within one solution, including confirmation of tubal patency prior to use with FemaSeed. FemCerv®, an endocervical tissue sampler for cervical cancer diagnosis, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel and New Zealand.
FemBloc® permanent birth control is a revolutionary first-in-class non-surgical solution which involves minimally-invasive placement of a patented delivery system for precise delivery of our proprietary synthetic tissue adhesive (blended polymer) into both fallopian tubes simultaneously. Over time, the blended polymer fully degrades and produces nonfunctional scar tissue to permanently block the fallopian tubes in a safe and natural approach. This is in stark contrast to centuries-old surgical sterilization with reported risks that include infection, minor or major bleeding, injury to nearby organs, anesthesia-related events, and even death. Along with the various surgical risks, some patients may not qualify as good surgical candidates due to obesity or medical comorbidities. The FemBloc non-surgical approach has the potential to offer a safe and effective, more accessible in-office alternative with fewer risks, contraindications, and substantially lower cost than the surgical alternative. A peer-reviewed publication of positive data from its initial clinical trials of FemBloc has demonstrated compelling effectiveness and five-year safety with high satisfaction from both patients and practitioners. In March 2025, we announced Conformité Européenne (CE) mark certification under European Union (“EU”) Medical Device Regulation (“MDR”) as the first regulatory approval in the world for the FemBloc delivery system for non-surgical female permanent birth control and in June 2025, we announced CE mark certification under EU MDR for the class III blended polymer component, achieving approval for the entire FemBloc system in the EU. In August and September 2025, we announced UK and New Zealand regulatory approvals, respectively, for FemBloc. In March and September 2025, we announced strategic distribution partnerships for FemBloc in Spain and France/Benelux region, respectively. The Company received FDA approval in November 2025 of its investigational device exemption (“IDE”) supplement to move forward to the final phase of the pivotal clinical trial (clinicaltrials.gov: NCT05977751) for U.S. approval. In March 2026, we announced
the initiation of enrollment in this final phase. FemChec, a companion diagnostic product for FemBloc’s ultrasound-based confirmation test, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel, Australia and New Zealand. FemCath® for selective fallopian tube evaluation is U.S. FDA-cleared and approved in Europe, Canada and Israel for selective fallopian tube evaluation. We are a woman-founded and led company with an expansive, internally created intellectual property portfolio with approximately 228 issued patents globally, in-house chemistry, manufacturing, and controls (CMC) and device manufacturing capabilities and proven ability to develop products with commercialization efforts underway. Our suite of products and U.S. product candidate address what we believe are multi-billion dollar global market segments in which there has been little advancement for many years, helping women avoid pharmaceutical solutions, implants and surgery that can be expensive and expose women to harm.
Factors Affecting Our Business
There are a number of factors that have impacted, and we believe will continue to impact, our results of operations and growth. These factors include:
•
Commencement and conduct of clinical trial for our U.S. product candidate. We must successfully recruit and enroll clinical trial participants in our clinical trial for FemBloc, in order to have the requisite data for regulatory submissions to the FDA for marketing authorization.
•
Regulatory approval of our U.S. product candidate. We must successfully obtain timely approval for our U.S. product candidate. For our sales to grow, we will need to receive FDA approval for the FemBloc system for permanent birth control, and will need to obtain additional regulatory approvals or marketing authorizations of our U.S. product candidate in international markets.
•
Clinical results. Publications of clinical results by us, our competitors and other third parties can have a significant influence on whether, and the degree to which, our products are used by healthcare practitioners and the procedures and treatments those healthcare practitioners choose to provide.
•
Commercialization and market acceptance. The success of our business will ultimately depend on our ability to commercialize our approved products and gain broad market acceptance of our products, which will require an extensive education process for both healthcare practitioners and patients of the benefits of our products, development of a robust sales force infrastructure and increased manufacturing capacity.
•
Competition. Our industry has a number of large, well-capitalized companies. We must continue to successfully compete in light of our competitors’ existing and future products and related pricing and their resources to successfully market to the healthcare practitioners who use our products.
•
Our financial condition. We need substantial additional funding to continue our development and commercialization plan and may be unable to raise capital when needed. We must comply with covenants and payment obligations under our convertible notes.
While these factors may present significant opportunities for us, they also pose significant risks and challenges that we must address. See the section titled “Risk Factors” for more information.
Components of Our Comprehensive Loss
Sales
Sales are primarily from the sale of our FemaSeed, FemBloc and FemVue products.
We sell our product to medical centers, including healthcare practitioner offices, primarily through our direct commercial team in the U.S., as well as through distribution partners in select international markets. Kebomed Europe AG, a distributor in France, accounted for approximately 21% of our revenue for the year ended December 31, 2025. Comercial Medico Quirugica, SA and Durgalab, distributors in Spain, accounted for approximately 8% and 8%, respectively, of our total revenue for the year ended December 31, 2025, and 15% and 12%, respectively, for the year ended December 31, 2024. For the years ended December 31, 2025 and 2024, Bayer Yakuhin, Ltd. accounted for approximately 3% and 7%, respectively, of our total revenue. For products sold in the U.S. through direct customer service, control is transferred upon shipment to customers. For products sold to distributors internationally, control is transferred upon shipment or delivery to the customer’s named location, based on the contractual shipping terms.
Cost of sales
Cost of sales consists primarily of costs of components for use in our products, the materials and labor that are used to produce our products, and the manufacturing overhead that directly supports production. We expect cost of sales to increase in absolute terms as our revenue grows.
Research and development
Research and development, or R&D, expenses consist of engineering, product development, clinical, quality assurance and regulatory expenses. R&D expenses include:
•
cost of clinical trials to support our U.S. product candidate FemBloc and product enhancements, including expenses for activities conducted by third-party services providers, primarily clinical research organizations, or CROs, and site payments;
•
certain personnel-related expenses, including salaries, benefits and stock-based compensation;
•
materials and supplies used for internal R&D and clinical activities;
•
allocated overhead information technology expenses; and
•
cost of outside consultants, who assist with technology development, regulatory affairs, clinical affairs and quality assurance, and testing fees.
We track outsourced development costs and other external research and development costs to specific product candidates on a program-by-program basis, fees paid to CROs, manufacturing and clinical development activities. However, we do not track our internal research and development expenses on a program-by-program basis as they primarily relate to compensation, overhead and early research and other costs which are deployed across multiple projects under development.
R&D costs are expensed as incurred. In the future, we expect R&D expenses to increase in absolute dollars as we continue to develop our U.S. product candidate FemBloc, expand our product candidate pipeline, enhance our existing products and technologies and perform activities related to obtaining additional regulatory approval.
Sales and marketing
Sales and marketing expenses consist of personnel-related expenses, including compensation, benefits, and stock-based compensation. Other sales and marketing expenses include marketing and promotional activities, including travel, trade shows and market research, and cost of outside consultants. We expect to expand our sales force and increase our marketing efforts as we commercialize our products based on our platform technologies. As a result, we expect sales and marketing expenses to increase in absolute dollars in future periods.
General and administrative
General and administrative expenses consist of personnel-related expenses, including compensation, benefits, travel and stock-based compensation. Other general and administrative expenses include professional services fees, including legal, audit and tax fees, insurance costs, cost of outside consultants and employee recruitment and training costs.
Depreciation and amortization
Depreciation and amortization expenses consist of depreciation expense associated with our fixed assets and amortization associated with our lease right-of-use assets and patents. We expect to invest in capital equipment to support our ongoing and planned commercialization efforts and continue to invest in our intellectual property. As a result, we expect our depreciation and amortization expenses to increase in absolute dollars in the future.
Other expense, net
Other expense, net consists
of debt financing costs, interest expense and other expenses, partially offset
by changes in fair value of derivatives and interest earned on our cash
equivalents.
Income tax expense
Income tax expense consists of the minimum state income taxes we are required to pay. We have a full valuation allowance for deferred tax assets, including net operating loss carryforwards and tax credits related primarily to R&D.
Results of Operations
Comparison of the Years Ended December 31, 2025 and 2024
The following table shows our results of operations for the years ended December 31, 2025 and 2024:
| |
| |
Year Ended December 31,
|
|
| | | |
| | | |
| |
| |
2025
| |
| |
2024
| |
| |
Change
| |
| |
% Change
| |
|
Sales
|
| $ | 2,293,313 | |
| | 1,629,108 | |
| | 664,205 | |
| | 40.8 | % |
|
Cost of sales (excluding depreciation expense)
|
| | 872,400 | |
| | 544,903 | |
| | 327,497 | |
| | 60.1 | % |
| |
| | | |
| | | |
| | | |
| | | |
|
Operating expenses:
|
| | | |
| | | |
| | | |
| | | |
|
Research and development
|
| | 7,577,704 | |
| | 8,216,543 | |
| | (638,839 | ) |
| | -7.8 | % |
|
Sales and marketing
|
| | 4,443,807 | |
| | 4,030,150 | |
| | 413,657 | |
| | 10.3 | % |
|
General and administrative
|
| | 6,646,037 | |
| | 6,325,999 | |
| | 320,038 | |
| | 5.1 | % |
|
Depreciation and amortization
|
| | 342,034 | |
| | 297,318 | |
| | 44,716 | |
| | 15.0 | % |
|
Total operating expenses
|
| | 19,009,582 | |
| | 18,870,010 | |
| | 139,572 | |
| | 0.7 | % |
|
Loss from operations
|
| | (17,588,669 | ) |
| | (17,785,805 | ) |
| | 197,136 | |
| | -1.1 | % |
|
Other (expense) income:
|
| | | |
| | | |
| | | |
| | | |
|
Interest income
|
| | 101,760 | |
| | 582,354 | |
| | (480,594 | ) |
| | -82.5 | % |
|
Change in fair
value of conversion option liability
|
| | 503,000 | |
| | — | |
| | 731,000 | |
| | 100 | % |
|
Change in fair
value of warrants liabilities
|
| |
731,000
| |
| | — | |
| | 503,000 | |
| | 100 | % |
|
Interest expense
|
| | (1,891,119 | ) |
| | (1,603,575 | ) |
| | (287,544 | ) |
| | 17.9 | % |
|
Other expense
|
| | (482,562 | ) |
| | — | |
| | (482,562 | ) |
| | -100 | % |
|
Total other expense, net
|
| | (1,037,921 | ) |
| | (1,021,221 | ) |
| | (16,700 | ) |
| | 1.6 | % |
|
Loss before income taxes
|
| | (18,626,590 | ) |
| | (18,807,026 | ) |
| | 180,436 | |
| | -1.0 | % |
|
Income tax expense
|
| | 1,297 | |
| | 9,602 | |
| | (8,305 | ) |
| | -86.5 | % |
|
Net loss
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
| | 188,741 | |
| | -1.0 | % |
Sales increased by $664,205 or 40.8%, to $2,293,313 in 2025 from $1,629,108 in 2024. The increase is attributable primarily to FemBloc product sales of $810,000, which were commercialized in 2025.
Cost of sales
Cost of sales increased by $327,497 or 60.1%, to $872,400 in 2025 from $544,903 in 2024. The increase in cost of sales is primarily attributed to increased sales and newly commercialized products.
Research and development
The following table summarizes our R&D expenses incurred during the periods presented:
| |
| |
| |
| | | |
| |
| |
Year Ended December 31, |
|
| |
| |
2025
| |
| |
2024
| |
|
Compensation and related personnel costs
|
| $ |
4,506,695 | |
| | 4,652,802 | |
|
Clinical-related costs
|
| |
1,023,995 | |
| | 1,665,736 | |
|
Material and development costs
|
| |
886,064 | |
| | 1,113,129 | |
|
Professional and outside consultant costs
|
| |
508,262 | |
| | 673,733 | |
|
Other costs
|
| |
652,688 | |
| | 111,143 | |
|
Total research and development expenses
|
| $ |
7,577,704 | |
| | 8,216,543 | |
R&D expenses decreased by $638,839 or 7.8%, to $7,577,704 in 2025 from $8,216,543 in 2024. The decrease is primarily due to reduced clinical costs, commercialization of development products into inventory, reduced professional fees, and reduced compensation and related personnel costs, partially offset by increased regulatory costs.
Sales and marketing
Sales and marketing expenses increased by $413,657 or 10.3%, to $4,443,807 in 2025 from $4,030,150 in 2024. The increase relates primarily to compensation, marketing and travel costs as we began recruitment of a commercial team in 2024 to promote our available products, partially offset by reduced professional fees.
General and administrative
General and administrative expenses increased by $320,038, or 5.1%, to $6,646,037 in 2025 from $6,325,999 in 2024. The increase relates primarily to increased compensation expense, partially offset by reduced facility and other overhead costs.
Depreciation and amortization
Depreciation and amortization expenses increased by $44,716, or 15.0%, to $342,034 in 2025 from $297,318 in 2024. The increase relates additional fixed assets and intangible assets in service.
Other expense, net
Total other expense, net
increased by $16,700, or 1.6%, to $1,037,921 in 2025 from $1,021,221 in 2024. Other
expense, net, primarily consists of interest income earned on our cash and cash
equivalents, interest expense for our 2025 and 2023 convertible notes, non-cash
interest expense related to the amortization of debt issuance costs for our
2025 and 2023 convertible notes, and non-cash charges recognized for the change
in fair value of our 2025 Conversion Option liability and Warrants
liabilities, issuance costs related to the AMPA derivative liability, and
deferred financing costs expensed in relation to an expired shelf. Refer below
for further details on the separate components of other expense, net.
| |
| | | |
| | | |
| |
| Year Ended December 31, | |
| |
|
2025
| |
|
2024
| |
|
Interest income
|
| $ | 101,760 | |
| | 582,354 | |
Amortization of debt issuance costs and discount for 2025 and 2023 Notes
|
| | (1,387,050 | ) |
| | (1,192,574 | ) |
|
Interest expense
|
| | (504,069 | ) |
| | (411,001 | ) |
|
Debt issuance costs for AMPA
|
| | (286,295 | ) |
| | — | |
|
Change in fair value of Conversion Option liability
|
| | 503,000 | |
| | — | |
|
Change in fair value of Warrants liabilities
|
| | 731,000 | |
| | — | |
|
Other expenses
|
| | (196,267 | ) |
| | — | |
| Total other expense, net |
| $ | (1,037,921 | ) |
| | (1,021,221 | ) |
Income tax expense
Income tax expense decreased by $8,305 or 86.5%, to $1,297 as compared to $9,602 in 2024 due to a decrease in the state minimum taxes we were required to pay.
Liquidity and Capital Resources
Sources of liquidity
Since our inception through December 31, 2025, our operations have been financed primarily by net proceeds from the sale of our common stock and convertible indebtedness and, to a lesser extent, product revenue. As of December 31, 2025, we had $9,266,353 of cash and cash equivalents and an accumulated deficit of $145,826,144.
In July 2022, we entered into an Equity Distribution Agreement (the “Equity Distribution Agreement”) with Piper Sandler & Co. (“Piper Sandler” or the “Sales Agent”) and filed a related prospectus establishing an “at-the-market” facility, pursuant to which we may offer and sell shares of our common stock from time to time through the Sales Agent. As of December 31, 2025, approximately 10.2 million shares of common stock have been sold to date for aggregate proceeds of $16.7 million under the Equity Distribution Agreement pursuant to the prospectus. In December 2025, we filed a new prospectus pursuant to which we may sell up to $9.8 million under the Equity Distribution Agreement. Subsequent to December 31, 2025, we raised approximately $0.4 million under the Equity Distribution Agreement. Based on our public float, as of the date of the filing of this Annual Report on Form 10-K, we are only permitted to utilize a shelf registration statement, including the registration statement under which our at the market offering is operated, subject to Instruction I.B.6 to Form S-3, which is referred to as the “baby shelf” rule. For so long as our public float is less than $75.0 million, we may not sell more than the equivalent of one-third of our public float during any 12 consecutive months pursuant to the baby shelf rules.
In June 2025, we sold 3,600,000 shares of common stock in an underwritten public offering at $0.85 per share. Separately and concurrently, we sold 1,686,275 shares of common stock in a private placement at a price of $0.85 per share to certain existing institutional stockholders and a price of $1.02 per share to certain directors and officers. Net proceeds from the transaction were $3,705,061.
On June 30, 2025, we entered into an Any Market Purchase Agreement (“Purchase Agreement” or “AMPA”) with Alumni Capital LP (“Alumni”) whereby we have the right, but not obligation, to sell to Alumni up to an aggregate of $10 million in shares of common stock in a series of purchases until December 31, 2026. We may elect that Alumni purchase up to $1 million in shares of common stock (or up to $5 million if mutually agreed) at either (i) the lowest traded price for 4 previous business days, multiplied by 90%, or (ii) up to the lesser of (a) $1 million in shares of common stock, or (b) 100% of the average daily trading volume of common stock for previous two business days at lowest daily dollar volume-weighted-average price, multiplied by 97%. We are limited to issuances to Alumni or 19.99% of the shares of common stock outstanding immediately prior to the execution of the Purchase Agreement.
In August 2025, we sold an aggregate of (i) 10,434,586 shares of common stock in a public offering and to certain Company officers (ii) pre-funded warrants to purchase up to 11,750,000 shares of common stock and (iii) common warrants to purchase up to 22,184,586 shares of common stock. Additionally, common warrants to purchase 443,692 shares of common stock were issued to the underwriter as compensation for services performed. The purchase price per share for the common stock was $0.36, except for shares sold to certain company officers, which was $0.5151 per share. The purchase price per share for the pre-funded warrants was $0.3599. The net proceeds from the August 2025 Financing at closing were approximately $7.1 million. As of December 31, 2025, 5,000,000 pre-funded warrants and 9,219,110 common warrants were exercised for approximately $3.3 million.
In November 2025, we entered
into a definitive agreement for the issuance of (i) senior secured convertible
notes in an aggregate principal amount of $12,000,000, convertible into shares
of 16,378,563 common stock at a conversion price of $0.73 per share, (ii)
Series A-1 Warrants to purchase up to an aggregate of 16,378,563 shares of
common stock at an exercise price of $0.81 per share, subject to adjustments
(iii) Series B-1 Warrants to purchase an aggregate of 16,378,563 shares of
common stock at an exercise price of $0.92 per share, subject to adjustments,
and (iv) Series C-1 Warrants to purchase an aggregate of 16,378,563 shares of
common stock at an exercise price of $1.10 per share, subject to adjustments.
The financing resulted in aggregate gross proceeds of $12,000,000, with total
potential funding of approximately $58 million, if all warrants are exercised
for cash.
Funding requirements
Based on our current operating plan, our current cash and cash equivalents and anticipated revenues from product sales are expected to be sufficient to fund our ongoing operations into the third quarter of 2026. Our estimate as to how long we expect our existing cash and cash equivalents to be able to continue to fund our operations is based on assumptions that may prove to be wrong, and we could use our available capital resources sooner than we currently expect. Changing circumstances, some of which may be beyond our control, could cause us to consume capital significantly faster than we currently anticipate.
Based on our current operating plan, our cash and cash equivalents as of December 31, 2025, anticipated revenues from product sales, proceeds from our 2025 financings, and approximately $0.4 million we raised subsequent to year end, will not be sufficient to sustain our operations, including funding our U.S. product candidate, FemBloc, through regulatory approval, and we will need to raise additional capital to complete the U.S. development and EU commercialization of our product candidate. However, we can give no assurances that we will be able to secure additional sources of funds to support our operations, or if such funds will be available to us, that such additional financing will be sufficient to meet our needs or be on terms acceptable to us. This risk may increase if economic and market conditions deteriorate. If we are unable to obtain additional financing when needed, we may need to terminate, significantly modify, or delay the development of our U.S. product candidate, or we may need to obtain funds through collaborations or otherwise on terms that may require us to relinquish rights to our technologies or U.S. product candidate that we might otherwise seek to develop or commercialize independently. If we are unable to raise adequate additional capital as and when required in the future, we could be forced to cease development activities and terminate our operations, and you could experience a complete loss of your investment.
We expect to continue to make substantial investments in our ongoing pivotal trial that is designed to provide clinical evidence of the safety and effectiveness of our U.S. product candidate, FemBloc. We also expect to continue to make investments in research and development to develop future products, manufacturing, regulatory affairs and post-market clinical trials. We will additionally need to make investments in our sales and marketing organization for FemaSeed and FemBloc. Because of these and other factors, we expect to continue to incur substantial net losses and negative cash flows from operations for the foreseeable future.
Our future capital requirements will depend on many factors, including:
•
the cost, timing and results of our clinical trial and U.S. regulatory reviews;
•
the cost and timing of establishing sales, marketing and distribution capabilities;
•
the timing, receipt and amount of sales from our current and potential products;
•
our ability to continue manufacturing our products and U.S. product candidate and to secure the components, services and supplies needed in their production;
•
the degree of success we experience in commercializing our products;
•
the emergence of competing or complementary technologies;
•
the cost of preparing, filing, prosecuting, maintaining, defending and enforcing any patent claims and other intellectual property rights; and
•
the extent to which we acquire or invest in businesses, products or technologies, although we currently have no commitments or agreements relating to any of these types of transactions.
Cash Flows
Comparison of the Years Ended December 31, 2025 and 2024
The following table summarizes our cash flows for the years ended December 31:
| |
| |
| |
| | | |
| |
| |
Year Ended December 31, |
|
| |
| |
2025
| |
| |
2024
| |
|
Net cash used in operating activities
|
| $ |
(18,690,565 | ) |
| | (19,444,009 | ) |
|
Net cash used in investing activities
|
| |
(630,098 | ) |
| | (847,764 | ) |
|
Net cash provided by financing activities
|
| |
25,135,255 | |
| | 2,027,457 | |
|
Net change in cash and cash equivalents
|
| $ |
5,814,592 | |
| | (18,264,316 | ) |
Operating activities
In 2025, cash used in
operating activities was $18,690,565, attributable to a net loss of $18,627,887
and a net change in our net operating assets and liabilities of $2,056,843,
partially offset by non-cash charges of $1,994,165. Non-cash charges primarily
consisted of $1,387,050 in amortization of the discount on the 2023 and 2025
convertible notes, $850,456 in depreciation and amortization, $790,145 in
stock-based compensation, $132,140 in accretion of paid-in-kind interest and
$62,650 in loss on property and equipment dispositions, partially offset by a
change in fair value of $503,000 and $731,000 of the Conversion Option
liability and Warrants liabilities, respectively. The change in our net operating
assets and liabilities was primarily due to increases in inventory of
$2,693,926 and accounts receivable of $128,227, decreases in lease liabilities
and other liabilities of $517,967 and $23,178, respectively, partially offset
by a decrease in prepaid and other assets of $496,902 and increases in accrued
expenses and accounts payable of $474,724 and $334,829, respectively, We intend
to meet future operating cash requirements through increased sales of
commercial products and fundraising, as discussed in Funding requirements.
In 2024, cash used in operating activities was $19,444,009, attributable to a net loss of $18,816,628, a net change in our net operating assets and liabilities of $3,136,778, partially offset by non-cash charges of $2,509,397. Non-cash charges primarily consisted of $1,192,574 in amortization of the discount on the convertible notes, $872,000 in depreciation and amortization expenses and $444,147 in stock-based compensation. The change in our net operating assets and liabilities was primarily due to increases of $2,379,205 in inventory, $389,467 in accounts receivable, $139,136 in prepaid and other assets, a decrease of $406,636 in lease liabilities, partially offset by an increase of $207,481 in accounts payable. The Company intends to meet future operating cash requirements through increased sales of commercial products and fundraising, as discussed in Funding requirements.
Investing activities
In 2025, cash used in investing activities for the purchases of fixed assets and intangible assets was $525,007 and $105,091, respectively.
In 2024, cash used in investing activities for the purchases of fixed assets and intangible assets was $761,706 and $86,058, respectively.
Financing activities
In 2025, cash provided by financing activities was $25,135,255,
attributable to proceeds of $11,281,472 from the November 2025 financing, net
of issuance costs, proceeds of $10,388,073 from the August 2025 financing, net
of issuance costs and inclusive of exercised warrants, sales under the
at-the-market facility of $6,802,767, net of issuance costs, proceeds of
$3,705,061 from the June 2025 financing, net of issuance costs, and $90,332 in
proceeds from the issuance of shares under the ESPP Plan. The proceeds were
partially offset by repayments of the 2023 Convertible Notes for $6,765,000 and
note payable for $367,450.
In 2024, cash provided by financing activities was $2,027,457, attributable to sales under the at-the-market facility of $1,964,351, net of issuance costs and proceeds from common stock issued through the employee stock purchase plan of $63,106.
Off-Balance Sheet Arrangements
During the periods presented, we did not have, nor do we currently have, any off-balance sheet arrangements as defined in the rules and regulations of the SEC.
Critical Accounting Estimates
Management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles, or GAAP. The preparation of these financial statements requires us to make estimates and assumptions for the reported amounts of assets, liabilities, revenue, expenses and related disclosures. Our estimates are based on our historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions and any such differences may be material.
While our significant accounting policies are more fully described in Note 2 to our financial statements appearing elsewhere in this Annual Report on Form 10-K, we believe the following discussion addresses our most critical accounting policy, which are those that are most important to our financial condition and results of operations and require our most difficult, subjective and complex judgments.
Revenue recognition
Our policy is to recognize revenue when a customer obtains control of the promised goods under Accounting Standards Codification 606—Revenue from Contracts with Customers (Topic 606), which we adopted effective January 1, 2018. The amount of revenue recognized reflects the consideration to which we expect to be entitled to receive in exchange for these goods, and we have elected to exclude amounts collected from customers for all sales (and other similar) taxes from the transaction price. Revenue is recognized upon shipment of the Company’s goods based upon contractually stated pricing at standard payment terms typically ranging from 30 to 60 days. All revenue is recognized at a point in time.
The majority of products sold directly to U.S. customers are shipped via common carrier, and the customer pays for shipping and handling and assumes control Free on Board (FOB) shipping point. Products shipped to our international distributors are in accordance with their respective agreements; however, the shipping terms are generally EX-Works, reflecting that control is assumed by the distributor at the shipping point. Returns are only accepted with prior authorization from the Company. Items to be returned must be in original unopened cartons and are subject to a 30% restocking fee. As of December 31, 2025, we have not had a history of significant returns.
Recent Accounting Pronouncements
See Notes 2(aa) and 2(ab) to our financial statements in Part II, Item 8 for information related to recently issued accounting pronouncements.
Item 7A.
Quantitative and Qualitative Disclosures About Market Risk.
Emerging Growth Company Status
We are an emerging growth company, as defined in the JOBS Act. Under the JOBS Act, emerging growth companies can delay adopting new or revised accounting standards issued subsequent to the enactment of the JOBS Act until such time as those standards apply to private companies. We have elected not to take advantage of such extended transition period, which means that we will adopt a new standard when a standard is issued or revised.
Financial Statements
Table of Contents
| | |
| | Page |
| | 91 |
| | |
| | 92-93 |
| | |
| | 94 |
| | |
| | 95 |
| | |
| | 96 |
| | |
| | 97-125 |
 | |
| |
KPMG LLP
Suite 2000
303 Peachtree Street, N.E.
Atlanta, GA 30308-3210 |
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
Femasys Inc.:
Opinion on the Financial Statements
We have audited the accompanying balance sheets of Femasys Inc. (the Company) as of December 31, 2025 and 2024, the related statements of comprehensive loss, stockholders’ equity, and cash flows for each of the years in the two-year period ended December 31, 2025, and the related notes (collectively, the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the years in the two-year period ended December 31, 2025, in conformity with U.S. generally accepted accounting principles.
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has suffered recurring losses and negative cash flows from operations, has a net accumulated deficit and expects to incur additional losses and negative operating cash flows. These factors raise substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
We have served as the Company’s auditor since 2016.
Atlanta, Georgia
March 31, 2026
| | |
|
|
KPMG LLP, a Delaware limited liability partnership, and its subsidiaries are part of the KPMG global organization of independent member firms affiliated with KPMG International Limited, a private English company limited by guarantee.
|
|
FEMASYS INC.
| | | | | | | | | |
|
Assets
|
| |
December 31,
2025
| |
| |
December 31,
2024
| |
|
Current assets:
|
| | | |
| | | |
|
Cash and cash equivalents
|
| $ | 9,266,353 | |
| | 3,451,761 | |
|
Accounts receivable, net
|
| | 616,600 | |
| | 488,373 | |
|
Inventory
|
| | 5,740,249 | |
| | 3,046,323 | |
|
Prepaid and other current assets
|
| | 833,133 | |
| | 1,035,993 | |
|
Total current assets
|
| | 16,456,335 | |
| | 8,022,450 | |
|
Property and equipment, at cost:
|
| | | |
| | | |
|
Leasehold improvements
|
| | 1,238,886 | |
| | 1,238,886 | |
|
Office equipment
|
| | 78,155 | |
| | 60,921 | |
|
Furniture and fixtures
|
| | 417,876 | |
| | 417,876 | |
|
Machinery and equipment
|
| | 3,065,713 | |
| | 2,856,740 | |
|
Construction in progress
|
| | 897,885 | |
| | 762,445 | |
| |
| | 5,698,515 | |
| | 5,336,868 | |
|
Less accumulated depreciation
|
| | (3,802,940 | ) |
| | (3,740,769 | ) |
|
Net property and equipment
|
| | 1,895,575 | |
| | 1,596,099 | |
|
Long-term assets:
|
| | | |
| | | |
|
Lease right-of-use assets, net
|
| | 1,297,121 | |
| | 1,805,543 | |
|
Intangible assets, net of accumulated amortization
|
| | 134,914 | |
| | 65,918 | |
|
Other long-term assets
|
| | 940,232 | |
| | 954,992 | |
|
Total long-term assets
|
| | 2,372,267 | |
| | 2,826,453 | |
| |
| | | |
| | | |
|
Total assets
|
| $ | 20,724,177 | |
| | 12,445,002 | |
(continued)
FEMASYS INC.
Balance Sheets
| | | | | | | | | |
|
Liabilities and Stockholders’ Equity
|
| |
December 31,
2025
| |
| |
December 31,
2024
| |
|
Current liabilities:
|
| | | |
| | | |
|
Accounts payable
|
| $ | 1,830,124 | |
| | 1,419,044 | |
|
Accrued expenses
|
| | 1,265,773 | |
| | 1,151,049 | |
|
Convertible notes payable, net (including related parties)
|
| | — | |
| | 5,406,228 | |
|
Clinical holdback – current portion
|
| | 52,644 | |
| | 88,581 | |
|
Operating lease liabilities – current portion
|
| | 487,624 | |
| | 517,967 | |
|
Total current liabilities
|
| | 3,636,165 | |
| | 8,582,869 | |
|
Long-term liabilities:
|
| | | |
| | | |
|
Clinical holdback – long-term portion
|
| | 52,370 | |
| | 39,611 | |
|
Convertible notes payable, net
|
| | 3,178,864 | |
| | — | |
|
Conversion option liability
|
| | 2,014,000 | |
| | — | |
|
Warrants liabilities
|
| | 4,943,000 | |
| | — | |
|
Operating lease liabilities – long-term portion
|
| | 1,030,476 | |
| | 1,518,100 | |
|
Total long-term liabilities
|
| | 11,218,710 | |
| | 1,557,711 | |
|
Total liabilities
|
| | 14,854,875 | |
| | 10,140,580 | |
|
Commitments and contingencies
|
| | | |
| | | |
|
Stockholders' equity:
|
| | | |
| | | |
| Common stock, $0.001 par, 200,000,000 authorized, 59,720,010 shares issued and 59,602,787 outstanding as of December 31, 2025; and 23,473,149 shares issued and 23,355,926 outstanding as of December 31, 2024 |
| | 59,720 | |
| | 23,473 | |
| Treasury stock, 117,223 common shares |
| | (60,000 | ) |
| | (60,000 | ) |
|
Warrants
|
| | 5,246,150 | |
| | 1,860,008 | |
|
Additional paid-in capital
|
| | 146,449,576 | |
| | 127,679,198 | |
|
Accumulated deficit
|
| | (145,826,144 | ) |
| | (127,198,257 | ) |
|
Total stockholders’ equity
|
| | 5,869,302 | |
| | 2,304,422 | |
| |
| | | |
| | | |
|
Total liabilities and stockholders' equity
|
| $ | 20,724,177 | |
| | 12,445,002 | |
See accompanying notes to financial statements.
FEMASYS INC.
Statements of Comprehensive Loss
Years ended December 31, 2025 and 2024
| | | | | | | | | |
| |
| |
December 31,
2025
| |
| |
December 31,
2024
| |
|
Sales
|
| $ | 2,293,313 | |
| | 1,629,108 | |
|
Cost of sales (excluding depreciation expense)
|
| | 872,400 | |
| | 544,903 | |
| |
| | | |
| | | |
|
Operating expenses:
|
| | | |
| | | |
|
Research and development
|
| | 7,577,704 | |
| | 8,216,543 | |
|
Sales and marketing
|
| | 4,443,807 | |
| | 4,030,150 | |
|
General and administrative
|
| | 6,646,037 | |
| | 6,325,999 | |
|
Depreciation and amortization
|
| | 342,034 | |
| | 297,318 | |
|
Total operating expenses
|
| | 19,009,582 | |
| | 18,870,010 | |
|
Loss from operations
|
| | (17,588,669 | ) |
| | (17,785,805 | ) |
|
Other (expense) income:
|
| | | |
| | | |
|
Interest income
|
| | 101,760 | |
| | 582,354 | |
|
Change in fair value of conversion option liability
|
| | 503,000 | |
| | — | |
|
Change in fair value of warrants liabilities
|
| | 731,000 | |
| | — | |
|
Interest expense
|
| | (1,891,119 | ) |
| | (1,603,575 | ) |
|
Other expense
|
| | (482,562 | ) |
| | — | |
|
Total other expense, net
|
| | (1,037,921 | ) |
| | (1,021,221 | ) |
|
Loss before income taxes
|
| | (18,626,590 | ) |
| | (18,807,026 | ) |
|
Income tax expense
|
| | 1,297 | |
| | 9,602 | |
| |
| | | |
| | | |
|
Net loss
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
| |
| | | |
| | | |
|
Net loss attributable to common stockholders, basic and diluted
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
| |
| | | |
| | | |
|
Net loss per share attributable to common stockholders, basic and diluted
|
| $ | (0.47 | ) |
| | (0.85 | ) |
|
Weighted-average common shares and pre-funded warrants outstanding- basic and diluted
|
| | 39,549,218 | |
| | 22,267,695 | |
See accompanying notes to financial statements.
FEMASYS INC.
Statements of Stockholders’ Equity
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| |
| | Common stock | |
| | Treasury common stock | |
| | | |
| |
Additional
paid-in capital
| |
| |
Accumulated
deficit
| |
| |
Total
stockholders’
equity
| |
| |
| |
Shares
| |
| |
Amount
| |
| |
Shares
| |
| |
Amount
| |
| |
Warrants
| |
|
Balance at December 31, 2023
|
| | 21,657,381 | |
| $ | 21,775 | |
| | 117,223 | |
| $ | (60,000 | ) |
| $ | 2,787,137 | |
| $ | 123,985,306 | |
| $ | (108,381,629 | ) |
| $ | 18,352,589 | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
|
Issuance of common stock in connection with at-the-market offering, net of issuance costs
|
| | 1,314,593 | |
| | 1,314 | |
| | — | |
| | — | |
| | — | |
| | 1,959,894 | |
| | — | |
| | 1,961,208 | |
|
Issuance of common shares in payment of convertible note interest, including related parties
|
| | 315,790 | |
| | 316 | |
| | — | |
| | — | |
| | —
| |
| | 299,684 | |
| | — | |
| | 300,000 | |
|
Issuance of common stock in connection with Employee Stock Purchase Plan
|
| | 68,162 | |
| | 68 | |
| | — | |
| | — | |
| | — | |
| | 63,038 | |
| | — | |
| | 63,106 | |
|
Share-based compensation expense
|
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | 444,147 | |
| | — | |
| | 444,147 | |
|
Expiration of warrant
|
| | — | |
| | — | |
| | — | |
| | — | |
| | (927,129 | ) |
| | 927,129 | |
| | — | |
| | — | |
|
Net loss
|
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | (18,816,628 | ) |
| | (18,816,628 | ) |
|
Balance at December 31, 2024
|
| | 23,355,926 | |
| $ | 23,473 | |
| | 117,223 | |
| $ | (60,000 | ) |
| $ | 1,860,008 | |
| $ | 127,679,198 | |
| $ | (127,198,257 | ) |
| $ | 2,304,422 | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
|
Issuance of common stock in connection with at-the-market offering, net of issuance costs
|
| | 5,628,980 | |
| | 5,629 | |
| | — | |
| | — | |
| | — | |
| | 6,786,252 | |
| | — | |
| | 6,791,881 | |
|
Issuance of common stock in connection with June 2025 financing, net of issuance costs
|
| | 5,286,275 | |
| | 5,286 | |
| | — | |
| | — | |
| | — | |
| | 3,695,024 | |
| | — | |
| | 3,700,310 | |
|
Issuance of warrants in connection with June 2025 financing
|
| | — | |
| | — | |
| | — | |
| | — | |
| | 56,035 | |
| | (56,035 | ) |
| | — | |
| | — | |
|
Issuance of common stock and warrants in connection with August 2025 financing, net of issuance costs
|
| | 10,434,586 | |
| | 10,435 | |
| | — | |
| | — | |
| | 5,833,237 | |
| | 1,225,022 | |
| | — | |
| | 7,068,694 | |
|
Exercise of pre-funded warrants
|
| | 5,000,000 | |
| | 5,000 | |
| | — | |
| | — | |
| | (1,080,574 | ) |
| | 1,076,074 | |
| | — | |
| | 500 | |
|
Exercise of common warrants
|
| | 9,219,110 | |
| | 9,219 | |
| | — | |
| | — | |
| | (1,328,257 | ) |
| | 4,637,917 | |
| | — | |
| | 3,318,879 | |
|
Issuance of common shares in payment of convertible note interest, including related parties
|
| | 455,697 | |
| | 456 | |
| | — | |
| | — | |
| | — | |
| | 359,544 | |
| | — | |
| | 360,000 | |
|
Issuance of common stock in connection with ESPP
|
| | 150,180 | |
| | 150 | |
| | — | |
| | — | |
| | — | |
| | 90,182 | |
| | — | |
| | 90,332 | |
|
Conversion of 2023 convertible notes into common stock
|
| | 72,033 | |
| | 72 | |
| | — | |
| | — | |
| | — | |
| | 71,954 | |
| | — | |
| | 72,026 | |
|
Share-based compensation expense
|
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | 790,145 | |
| | — | |
| | 790,145 | |
|
Expiration of warrant
|
| | — | |
| | — | |
| | — | |
| | — | |
| | (94,299 | ) |
| | 94,299 | |
| | — | |
| | — | |
|
Net loss
|
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | — | |
| | (18,627,887 | ) |
| | (18,627,887 | ) |
|
Balance at December 31, 2025
|
| | 59,602,787 | |
| $ | 59,720 | |
| | 117,223 | |
| $ | (60,000 | ) |
| $ | 5,246,150 | |
| $ | 146,449,576 | |
| $ | (145,826,144 | ) |
| $ | 5,869,302 | |
See accompanying notes to financial statements.
FEMASYS INC.
Years ended December 31, 2025 and 2024
| | | | | | | | | |
| |
| | | |
| | | |
| |
| | Years ended December 31 | |
| |
| |
2025
| |
| |
2024
| |
|
Cash flows from operating activities:
|
| | | |
| | | |
|
Net loss
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
| | | |
| | | |
|
Depreciation
|
| | 295,859 | |
| | 277,178 | |
|
Amortization
|
| | 46,175 | |
| | 20,140 | |
|
Other noncash items
|
| | 5,724 | |
| | — | |
|
Change in fair value of conversion option liability
|
| | (503,000 | ) |
| | — | |
|
Change in fair value of warrants liabilities
|
| | (731,000 | ) |
| | — | |
|
Amortization of right-of-use assets
|
| | 508,422 | |
| | 574,682 | |
|
Loss on fixed asset dispositions
|
| | 62,650 | |
| | 676 | |
|
Share-based compensation expense
|
| | 790,145 | |
| | 444,147 | |
|
Amortization of debt issuance costs and discount
|
| | 1,387,050 | |
| | 1,192,574 | |
|
Accretion of paid-in-kind interest
|
| | 132,140 | |
| | — | |
|
Changes in operating assets and liabilities:
|
| | | |
| | | |
|
Accounts receivable
|
| | (128,227 | ) |
| | (389,467 | ) |
|
Inventory
|
| | (2,693,926 | ) |
| | (2,379,205 | ) |
|
Prepaid and other assets
|
| | 496,902 | |
| | (139,136 | ) |
|
Accounts payable
|
| | 334,829 | |
| | 207,481 | |
|
Accrued expenses
|
| | 474,724 | |
| | 6,753 | |
|
Lease liabilities
|
| | (517,967 | ) |
| | (406,636 | ) |
|
Other liabilities
|
| | (23,178 | ) |
| | (36,568 | ) |
|
Net cash used in operating activities
|
| | (18,690,565 | ) |
| | (19,444,009 | ) |
|
Cash flows from investing activities:
|
| | | |
| | | |
|
Acquisition of patents
|
| | (105,091 | ) |
| | (86,058 | ) |
|
Purchases of property and equipment
|
| | (525,007 | ) |
| | (761,706 | ) |
|
Net cash used in investing activities
|
| | (630,098 | ) |
| | (847,764 | ) |
|
Cash flows from financing activities:
|
| | | |
| | | |
|
Proceeds from the issuance of common stock in June 2025 financing
|
| | 4,510,001 | |
| | — | |
|
Issuance costs for June 2025 financing
|
| | (804,940 | ) |
| | — | |
|
Proceeds from the issuance of common stock, accompanying warrants and pre-funded warrants in August 2025 financing
|
| | 7,998,826 | |
| | — | |
|
Issuance costs for August 2025 financing
|
| | (930,132 | ) |
| | — | |
|
Proceeds from exercise of pre-funded warrants
|
| | 500 | |
| | — | |
|
Proceeds from exercise of common warrants
|
| | 3,318,879 | |
| | — | |
|
Proceeds from November 2025 convertible notes
|
| | 12,000,000 | |
| | — | |
|
Issuance costs for November 2025 convertible notes
|
| | (718,528 | ) |
| | — | |
|
Proceeds from common stock issued through ESPP
|
| | 90,332 | |
| | 63,106 | |
|
Proceeds from at-the-market sales of common stock
|
| | 7,013,162 | |
| | 2,025,104 | |
|
Issuance costs for at-the-market sales of common stock
|
| | (210,395 | ) |
| | (60,753 | ) |
|
Repayment of November 2023 convertible notes, including related parties
|
| | (6,765,000 | ) |
| | — | |
|
Repayment of note payable
|
| | (367,450 | ) |
| | — | |
|
Net cash provided by financing activities
|
| | 25,135,255 | |
| | 2,027,457 | |
|
Net change in cash and cash equivalents
|
| | 5,814,592 | |
| | (18,264,316 | ) |
|
Cash and cash equivalents:
|
| | | |
| | | |
|
Beginning of period
|
| | 3,451,761 | |
| | 21,716,077 | |
|
End of period
|
| $ | 9,266,353 | |
| | 3,451,761 | |
| |
| | | |
| | | |
|
Supplemental cash flow information
|
| | | |
| | | |
|
Cash paid for:
|
| | | |
| | | |
|
Interest
|
| $ | 8,615 | |
| | 111,000 | |
| Income taxes |
| | 6,806 | |
| | 5,708 | |
|
Non-cash investing and financing activities:
|
| | | |
| | | |
|
Property and equipment costs included in accounts payable and accrued expense
|
| | 60,447 | |
| | 73,740 | |
|
Acquisition of patents included in accounts payable
|
| | 15,804 | |
| | — | |
|
Issuance of warrants for underwriter commission in June 2025 financing
|
| | 56,035 | |
| | — | |
|
Issuance of warrants for underwriter commission in August 2025 financing
|
| | 97,612 | |
| | — | |
|
Conversion of November 2023 convertible notes into common stock
|
| | 85,000 | |
| | — | |
|
Payment of November 2023 convertible note interest in common stock
|
| | 360,000 | |
| | 300,000 | |
|
Deferred offering costs reclassified to additional paid-in capital
|
| | 15,637 | |
| | 3,143 | |
|
Fair value of conversion option liability at November 2025 issuance
|
| | 2,517,000 | |
| | — | |
|
Fair value of warrants liabilities at November 2025 issuance
|
| | 5,674,000 | |
| | — | |
See accompanying notes to financial statements.
(1)
Organization, Nature of Business, and Liquidity
Organization and Nature of Business
Femasys Inc. (the Company or Femasys) was incorporated in Delaware on February 19, 2004 and is headquartered in Suwanee, Georgia. The Company is a leading biomedical innovator, addressing significant unmet needs in women’s health worldwide, with a broad patent-protected portfolio of disruptive, accessible, in-office therapeutic and diagnostic products. The Company is a U.S. manufacturer that has received global regulatory approvals for its product portfolio, which are currently being commercialized in the U.S. and key international markets. The Company’s suite of products and U.S. product candidate address what the Company believes are multi-billion dollar global market segments in which there has been little advancement for many years, helping women avoid pharmaceutical solutions, implants and surgery that can be expensive and expose women to harm.
Basis of Presentation
The Company has prepared the accompanying financial statements in accordance with accounting principles generally accepted in the United States of America (GAAP) and pursuant to the rules and regulations of the U.S. Securities and Exchange Commission (SEC).
Liquidity
As of December 31, 2025, the Company has cash and cash equivalents of $9,266,353. The Company plans to finance its operations and development needs with its existing cash and cash equivalents, additional equity and/or debt financing arrangements, and revenue primarily anticipated from domestic sales of FemaSeed and FemVue and international sales of FemBloc, FemaSeed and FemVue to support the Company’s commercial efforts and research and development activities, primarily focused on FemBloc. There can be no assurance that the Company will be able to obtain additional financing on terms acceptable to the Company, on a timely basis, or at all. If the Company is not able to obtain sufficient funds on acceptable terms when needed, the Company’s business, results of operations, and financial condition could be materially adversely impacted.
For the year ended December
31, 2025, the Company generated a net loss of $18,627,887. The Company expects
such losses to increase over the next few years as the Company commercializes
FemaSeed and its other products and advances FemBloc through clinical
development if and until FDA approval is received and is available to be
marketed in the U.S.
The financial statements have
been prepared on a going-concern basis, which contemplates the realization of
assets and the satisfaction of liabilities in the normal course of business.
The Company has incurred net operating losses in every year since inception and
has an accumulated deficit as of December 31, 2025 of $145,826,144 and expects
to incur additional losses and negative operating cash flows for at least the
next twelve months. The Company’s ability to meet its obligations is dependent
upon its ability to generate sufficient cash flows from operations and future
financing transactions. Although management expects the Company will continue
as a going concern, there is no assurance that management’s plans will be
successful since the availability and amount of such funding is not certain.
Accordingly, substantial doubt exists about the Company’s ability to continue
as a going concern for at least one year from the issuance of these financial
statements. The accompanying financial statements do not include any
adjustments to reflect the possible future effects on the recoverability of
assets or the amounts and classifications of liabilities that may result from
the possible inability of the Company to continue as a going concern.
(2)
Summary of Significant Accounting Policies
(a)
Use of Estimates in Preparation of Financial Statements
The preparation of financial
statements in conformity with U.S. generally accepted accounting principles
(GAAP) requires the Company to make estimates and assumptions that affect the
reported amounts of assets and liabilities and disclosure of contingent assets
and liabilities at the date of the financial statements, and the reported
amounts of revenue and expense during the reporting periods. The most
significant estimates used in these financial statements include stock options,
warrants, conversion option and warrants liabilities, the valuation of useful
lives of property and equipment, and clinical trial cost accruals. Actual
results could differ from those estimates.
(b)
Certain Risks and Uncertainties
Products being developed by the Company, such as FemBloc, will require approval from the FDA or corresponding foreign regulatory agencies prior to commercial sales. FemBloc is approved in the European Union, UK and New Zealand. FemaSeed Intratubal Insemination is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel, Australia and New Zealand. FemVue, a companion diagnostic for fallopian tube assessment via ultrasound, is U.S. FDA-cleared and approved in Europe, UK, Canada, Japan, Israel, Australia and New Zealand. FemVue Controlled, a diagnostic solution for controlled contract delivery to evaluate fallopian tube status, is U.S. FDA-cleared. FemCerv, an endocervical tissue sampler for cervical cancer diagnosis, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel and New Zealand. FemCath for selective fallopian tube evaluation is U.S. FDA-cleared and approved in Europe, Canada and Israel. FemChec, a companion diagnostic product for FemBloc’s ultrasound-based confirmation test, is U.S. FDA-cleared and approved in Europe, UK, Canada, Israel, Australia and New Zealand. There can be no assurance the Company’s other products in development will receive the necessary approvals/ clearances. If the Company is denied regulatory approval/ clearance or approval/ clearance is delayed, it might have a materially adverse impact on the Company.
The medical device industry is characterized by frequent and extensive litigation and administrative proceedings over patent and other intellectual property rights. Whether a product infringes a patent involves complex legal and factual issues, the determination of which is often difficult to predict, and the outcome may be uncertain until the court has entered final judgment and all appeals are exhausted. The Company’s competitors may assert that its products or the use of its products are covered by United States or foreign patents held by them. If such relevant patents are upheld as valid and enforceable and the Company is found to infringe, the Company could be prevented from selling its products unless it can obtain a license to use technology or ideas covered by such patents or are able to redesign its products to avoid infringement. A license may not be available at all or on commercially reasonable terms, and it may not be able to redesign its products to avoid infringement.
The Company relies on some single source suppliers to provide certain components of its products commercially available and those under development. The Company purchases these components on a purchase order basis. If the Company overestimates its component requirements, it could have excess inventory, which would increase its costs and result in write‑downs harming its operating results. If the Company underestimates its requirements, it may not have an adequate supply, which could interrupt the manufacturing of its products.
(c)
Fair Value of Financial Instruments
The Company applies a fair value hierarchy that requires the use of observable market data, when available, and prioritizes the inputs to valuation techniques used to measure fair value in the following categories:
Level 1 – Valuation is based upon quoted prices for identical instruments traded in active markets.
Level 2 – Valuation is based upon quoted prices for similar instruments in active markets, quoted prices for identical or similar instruments in markets that are not active, and model‑based valuation techniques for which all significant assumptions are observable in the market.
Level 3 – Valuation is generated from model‑based techniques that use significant assumptions not observable in the market. These unobservable assumptions reflect the Company’s own estimates of assumptions market participants would use in pricing the asset or liability.
Certain of the Company’s financial instruments, including cash and cash equivalents, accounts receivable and other liabilities approximate their fair value because of the short‑term maturity of these financial instruments.
(d)
Cash and Cash Equivalents
The Company considers all
highly liquid investments with an initial maturity of three months or less when
purchased to be cash equivalents. Cash equivalents consist of amounts invested
in money market mutual funds and are stated at fair value. See Note 2(t) for
information on concentration of credit risk.
The Company grants trade credit to customers in the normal course of business and does not require collateral or any other security to support its receivables. Management reviews its accounts receivable monthly for any collection issues. Potentially uncollectible accounts are written off to bad debt expense when it is determined that the likelihood a customer account is uncollectible is probable. As of December 31, 2025 and 2024, the Company’s reserves for uncollectible accounts were $14,013 and $10,000, respectively. Trade accounts receivable are recorded at the invoiced amount, net of allowance for credit loss and do not bear interest. Amounts collected on trade accounts receivable are included in net cash provided by operating activities in the statements of cash flows.
Inventories are stated at the lower of cost or net realizable value. Cost, which includes amounts related to materials, labor and overhead, is determined on a first‑in, first‑out basis. Net realizable value is the estimated selling price in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation.
Inventory stated at cost, consisted of the following as of December 31:
| | | | | | | | | |
| |
| |
2025
| |
| |
2024
| |
|
Materials
|
| $ | 2,060,521 | |
| | 1,308,863 | |
|
Work in progress
|
| | 1,266,542 | |
| | 982,630 | |
|
Finished goods
|
| | 2,413,186 | |
| | 754,830 | |
|
Inventory
|
| $ | 5,740,249 | |
| | 3,046,323 | |
The Company has research tax credits that are available to the Company to offset future payroll withholding liabilities. As of December 31, 2025 and 2024, the total amount of these credits is $948,292 and $1,030,809, respectively. The Company has included these amounts on the accompanying balance sheets as follows as of December 31:
| | | | | | | | | |
| |
| |
2025 | |
| |
2024
| |
|
Prepaid and other current assets
|
| $ | 249,394 | |
| | 315,897 | |
|
Other long-term assets
|
| | 698,898 | |
| | 714,912 | |
|
Research tax credits available to the Company
|
| $ | 948,292 | |
| | 1,030,809 | |
(h)
Property and Equipment
Property and equipment are carried at cost less accumulated depreciation and, if applicable, impairment charges. Expenditures which materially increase value or extend useful lives of assets are capitalized, while maintenance and repairs which do not improve or extend the lives of the respective assets are charged to operations when incurred. Gains and losses on the retirement or disposal of individual assets are included in the results of operations.
Depreciation and amortization are computed using the straight‑line method over estimated useful lives of assets as follows:
| | |
|
Leasehold improvements
|
Shorter of lease term(s) or useful life
|
|
Office equipment
|
5 years |
|
Furniture and fixtures
|
7 years |
|
Machinery and equipment
|
5 to 7 years |
Depreciation expense for the years ended 2025 and 2024 was $295,859 and $277,178, respectively. In 2025, the Company disposed of property and equipment at an original cost of $296,338 with a net book value of $62,650, which is recorded in operating expenses on the statements of comprehensive loss. In 2024, the Company disposed of property and equipment at an original cost of $82,507 with a net book value of $676, which is recorded in operating expenses on the statements of comprehensive loss.
(i)
Impairment of Long-Lived Assets
The Company reviews long‑lived assets, including property and equipment and definite lived intangibles, for impairment whenever events or changes in business circumstances indicate that the carrying amount of the asset group may not be fully recoverable. An impairment loss would be recognized when estimated undiscounted future cash flows expected to result from the use of the asset group and its eventual disposition is less than its carrying amount. Impairment, if any, is measured as the amount by which the carrying amount of a long‑lived asset group exceeds its fair value. The Company has not recorded any impairment losses to date.
The Company records operating leases as right-of-use assets and operating lease liabilities in its balance sheets for all operating leases with terms exceeding one year. Right-of-use assets represent the right to use an underlying asset for the lease term, including extension options considered reasonably certain to be exercised, and operating lease liabilities to make lease payments. Right-of-use assets and operating lease liabilities are recognized based on the present value of lease payments over the lease term. To the extent that lease agreements do not provide an implicit rate, the Company uses its incremental borrowing rate based on information available at the lease commencement date to determine the present value of lease payments. The expense for operating lease payments is recognized on a straight-line basis over the lease term and is included in operating expenses in the Company’s statements of comprehensive loss. Non-lease components included in lease agreements are accounted for separately. The Company records finance leases as right-of-use assets and finance lease liabilities in its balance sheets for all finance leases with terms exceeding one year, similar to operating leases, and records interest expense and depreciation expense on the right-of-use asset in the statements of comprehensive loss.
Intangible assets consist of patent and trademark application costs and related legal fees, carried at cost less accumulated amortization and, if applicable, impairment charges. Amortization is computed using the straight‑line method over a weighted-average useful life of three years and is recorded in depreciation and amortization expense within the results of operations. Intangible assets consist of the following as of December 31:
| | | | | | | | | |
| |
| |
2025
| |
| |
2024
| |
|
Cost
|
| $ | 1,869,257 | |
| | 1,755,009 | |
|
Accumulated amortization
|
| | (1,734,343 | ) |
| | (1,689,091 | ) |
|
Net book value
|
| $ | 134,914 | |
| | 65,918 | |
Amortization expense for intangible assets for the years ended December 31, 2025 and 2024 was $46,175 and $20,140, respectively.
(l)
Deferred Offering Costs
Deferred offering costs, which consist mainly of legal, consulting, and accounting fees directly attributable to a strategic financing transaction, are capitalized in accordance with Staff Accounting Bulletin (SAB) Topic 5.A Expenses of Offering, codified in Accounting Standards Codification (ASC) 340-10-S99-1 Other Assets and Deferred Costs.
During 2022, the Company
incurred $232,845 in deferred offering costs in connection with prospectuses
filed in July 2022, including an offering to sell up to $150 million in stock,
debt securities and warrants, and an Equity Distribution Agreement entered into
with Piper Sandler which included an at-the-market (ATM) facility. These
deferred offering costs were offset against the total proceeds from the issuance
of common stock available under the prospectuses. As of December 31, 2025, and 2024, the Company
offset $15,637 and $3,143, respectively of deferred offering costs in
connection with the gross proceeds issued under the prospectuses. The shelf
expired in 2025 and the remaining balance of $196,267 was expensed as other
expense in the statements of comprehensive loss.
During 2025, the Company
incurred $73,785 in deferred offering costs in connection with a prospectus
filed in July 2025, including an offering to sell up to $150 million in stock,
debt securities and warrants. As of December 31, 2025, deferred offering costs
capitalized were $73,785 and are included in other assets in the accompanying
balance sheet. As of December 31, 2024, deferred offering costs capitalized
were $211,654 and are included in prepaid and other current assets in the
accompanying balance sheet. These costs will be offset against proceeds from the issuance of common stock sales and will be expensed if the prospectus is terminated or aborted.
Accrued compensation costs include incentive compensation and unused paid time off. Accrued clinical trial expenses include research and development costs for third-party services, largely related to the Company’s clinical trials, which are estimated based upon the services provided but not yet invoiced. These costs, at times, may be a significant component of the research and development expenses and the Company makes estimates in determining the accrued expense each period. As actual costs become known, the Company adjusts its accrual. Other accrued expenses include director fees, taxes and other miscellaneous accrued expenses.
Accrued expenses consisted of the following as of December 31:
| | | | | | | | | |
| |
| |
2025
| |
| |
2024
| |
|
Incentive and other compensation costs
|
| $ | 870,910 | |
| | 650,768 | |
|
Clinical trial costs
|
| | 259,293 | |
| | 354,762 | |
|
Director fees
|
| | 71,250 | |
| | 70,000 | |
|
Other
|
| | 64,320 | |
| | 75,519 | |
|
Accrued expenses
|
| $ | 1,265,773 | |
| | 1,151,049 | |
As part of the regulatory
approval process for taking its products to market or conducting post-market
clinical studies to support marketing efforts for products with regulatory
clearance, the Company enters into certain Clinical Trial Agreements (CTAs) which
include, among other things, the compensation and payment schedule the
participating medical institutions and investigators will receive for all costs
in connection with the clinical trial (or study) under the terms of the CTA. As
individual participants are enrolled in the study by the participating medical
institution or investigator, the Company pays certain per study fees according
to the CTA for the duration of the trial. As invoices are received by the
Company from the medical institution or investigator, the Company retains any
agreed upon percentage of total invoiced costs, generally ranging between 5% -
15%, which is withheld from payment until the end of the study. These retained
amounts are recorded as clinical holdback, a liability, on the accompanying
balance sheets, and all expenses incurred in connection with these CTA
activities are expensed as services are provided, which are included as
research and development expenses on the accompanying statements of comprehensive
loss.
The Company accounts
for its convertible notes based on an assessment of the convertible note terms
and applicable guidance ASC 470-20, Debt with Conversion and Other Options
(ASC 470-20) and ASC 815-40, Derivatives and Hedging—Contracts in Entity’s
Own Equity (ASC 815). The Company evaluates all features contained in
financing agreements to determine if there are any embedded derivatives that
require separate accounting from the underlying agreement. An embedded
derivative that requires separation is accounted for as a separate asset or
liability from the host agreement. The derivative asset or liability is
accounted for at fair value, with changes in fair value recognized in the
statement of comprehensive loss at each reporting period.
The convertible notes were
recorded net of debt issuance costs and any discounts. The Company has
historically issued warrants in conjunction with issuing convertible notes (see
Note 7). Debt issuance costs are amortized over the term of the convertible
notes using the effective interest method in addition to the discount initially
recognized for the fair value of warrants issued. The amortization of debt
issuance costs and discount is included in interest expense in the statements
of comprehensive loss. If the debt is retired early, the associated debt
discount will be recognized immediately as interest expense in the statements
of comprehensive loss.
(p)
Common Stock Warrants
The Company accounts for its
common stock warrants as equity-classified or liability-classified instruments
based on an assessment of the warrants’ specific terms and applicable
authoritative guidance in ASC 480, Distinguishing Liabilities from Equity
(ASC 480) and ASC 815. The assessment considers whether the warrants are (i)
freestanding financial instruments pursuant to ASC 480, (ii) meet the
definition of a liability pursuant to ASC 480, and (iii) meet all of the
requirements for equity classification under ASC 815, including whether the
warrants are indexed to the Company’s own common stock and whether the warrant
holders could potentially require “net cash settlement” in a circumstance
outside of the Company’s control, among other conditions for equity classification.
This assessment, which requires the use of professional judgment, is conducted
at the time of warrant issuance and as of each subsequent quarterly period end
date while the warrants are outstanding.
For warrants that meet all of
the criteria for equity classification, the warrants are required to be
recorded as a component of additional paid-in capital, on the balance sheets at
the time of issuance. For warrants that do not meet all the criteria for equity
classification, the warrants are required to be recorded at their initial fair
value on the date of issuance and on each balance sheet date thereafter.
The Company’s warrants liabilities are measured at fair value using a simulation model which takes
into account, as of the valuation date, factors including the current exercise
price, the expected life of the warrant, the current price of the Company's common
stock, the expected volatility, the risk-free interest rate for the term of the
warrant and the likelihood of achieving certain future milestone events and the
related impact to the price of the Company's common stock. The warrants liabilities are revalued at each reporting period and changes in fair value are
recognized in other (expense) income in the statements of comprehensive loss. The
selection of the appropriate valuation model and the inputs and assumptions
that are required to determine the valuation requires significant judgment and
requires management to make estimates and assumptions that affect the reported
amount of the related liability and reported amounts of the change in fair
value. Actual results could differ from those estimates, and changes in these
estimates are recorded when known. See
Note 7 for additional information on warrants issued by the Company.
(q)
Any Market Purchase Agreement (“AMPA”)
The Company evaluates all features contained in financing agreements to determine if there are any embedded derivatives that require separate accounting from the underlying agreement. An embedded derivative that requires separation is accounted for as a separate asset or liability from the host agreement. The derivative asset or liability is accounted for at fair value, with changes in fair value recognized in the statement of comprehensive loss. The Company determined that certain features under the AMPA contained embedded derivatives. The derivative liability associated with the AMPA were deemed de minimus at both the issuance date and as of December 31, 2025.
The Company’s policy is to recognize revenue when a customer obtains control of the promised goods under ASC 606, Revenue from Contracts with Customers. The amount of revenue recognized reflects the consideration to which the Company expects to be entitled to receive in exchange for these goods, and the Company has elected to exclude amounts collected from customers for all sales (and other similar) taxes from the transaction price. Revenue is recognized upon shipment of the Company’s goods based upon contractually stated pricing at standard payment terms typically ranging from 30 to 60 days. All revenue is recognized point in time and no revenue is recognized over time. There was no revenue recognized during the years ended December 31, 2025 and 2024 from performance obligations satisfied or partially satisfied in prior periods. Additionally, there were no unsatisfied performance obligations as of December 31, 2025 and 2024.
The majority of products sold directly to U.S. customers are shipped via common carrier, and the customer pays for shipping and handling and assumes control Free on Board (FOB) shipping point. Products shipped to the Company’s international distributors are in accordance with their respective agreements; however, the shipping terms are generally EX-Works, reflecting that control is assumed by the distributor at the shipping point. Returns are only accepted with prior authorization from the Company. Items to be returned must be in original unopened cartons and are subject to a 30% restocking fee. As of December 31, 2025, the Company has not had a history of significant returns.
The following table summarizes the Company’s sales by geographic region for the years ending December 31:
| | | | | | | | | |
|
Primary geographical markets
|
| |
2025
| |
| |
2024
| |
|
U.S.
|
| $ | 1,393,555 | |
| | 1,073,508 | |
|
International
|
| | 899,758 | |
| | 555,600 | |
|
Total
|
| $ | 2,293,313 | |
| | 1,629,108 | |
(s)
License, Manufacturing, and Supply Agreements
The Company entered into a FemVue License, Manufacturing, and Supply Agreement with Bayer Yakuhin, Ltd., a wholly owned subsidiary of Bayer AG, in 2012. The Company sells products based on purchase orders provided by Bayer Yakuhin in accordance with their agreement. Control and risk of ownership transfer at the time of shipment and the Company records revenue at that time.
The Company entered in distributor agreements with Comercial Medico Quirurgica, SA (“Comercial”) and Durgalab in September 2024. The Company sells products based on purchase orders provided by Comercial and Durgalab in accordance with their agreements. Control and risk of ownership transfer at the time of shipment and the Company records revenue at that time.
The Company entered in a distributor agreement with Kebomed Europe AG (“Kebomed”) in September 2025. The Company sells products based on purchase orders provided by Kebomed in accordance with their agreement. Control and risk of ownership transfer at the time of shipment and the Company records revenue at that time.
(t)
Concentration of Credit Risk
The Company generates revenue from sales directly to U.S. customers and to the Company’s international distributors with all prices in U.S. dollars. For the year ended December 31, 2025, one customer accounted for revenues of 21% of total revenues. As of December 31, 2025, one customer had an accounts receivable balance of 76% of total receivables, which was paid subsequent to year end. No other customer had revenue or accounts receivable in excess of 10% of total receivables. For the year ended December 31, 2024, two customers accounted for revenues of 15% and 12% of total revenues. As of December 31, 2024, three customers had balances of 23%, 21% and 12% of total receivables.
(u)
Research and Development
The Company’s research and development expenses consist of engineering, product development, clinical, quality assurance and regulatory expenses and are expensed as incurred. These expenses include direct expenses related to employee compensation, including salary, benefits and stock-based compensation; expenses related to consulting fees, testing fees, materials, and supplies; and activities conducted by third-party service providers, which include the conducting of preclinical studies and clinical trials.
The Company’s sales and marketing expenses consist of direct expenses related to employee compensation, including salary, benefits and stock-based compensation, advertising and marketing, business development and travel.
(w)
General and Administrative
The Company’s general and administrative expenses include accounting, human resources, and general corporate expenses. These expenses are primarily related to employee compensation, including salary, benefits, and stock‑based compensation. General corporate expenses generally relate to office rent, utilities, insurance, legal, and professional fees.
Advertising costs are expensed as incurred. Advertising costs were $262,270 and $360,958 for the years ended December 31, 2025 and 2024, respectively. They are reflected in sales and marketing expenses in the statements of comprehensive loss.
(y)
Stock-Based Compensation
The Company recognizes all employee and nonemployee stock-based compensation as a cost in the financial statements. Equity-classified awards are measured at the grant-date fair value of the award. The Company estimates grant-date fair value using the Black-Scholes option pricing model and records forfeitures as they are incurred. Details of the stock‑based compensation and accounting treatment are discussed in Note 10.
The Company utilizes the asset‑and‑liability method of accounting for income taxes as set forth in ASC 740, Income Taxes. Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes, as well as the net operating loss, capital loss, and tax credit carryforwards. Valuation allowances are established against deferred tax assets if it is more likely than not that they will not be realized.
ASC 740 prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. The standard requires that the Company recognize in its financial statements the impact of a tax position if that position is more likely than not of being sustained on audit, based on the technical merits of the position. The Company has determined it had no unrecognized tax benefits as of December 31, 2025 and 2024.
As of December 31, 2025, the 2022 through 2025 tax years remain subject to examination by federal and most state tax authorities. The use of net operating losses generated in tax years prior to 2022 may also subject returns for those years to examination.
(aa)
Net Loss per Share Attributable to Common Stockholders
Basic net loss per share attributable to common stockholders is calculated by dividing the net loss attributable to common stockholders by the weighted-average number of shares of common stock outstanding during the period, without consideration of common stock equivalents. The net loss attributable to common stockholders is calculated by adjusting the net loss of the Company for the cumulative dividends, if any. Diluted net loss per share attributable to common stockholders is the same as basic net loss per share attributable to common stockholders since the effect of potentially dilutive securities is anti-dilutive given the net loss position of the Company.
(ab)
Recently Issued Accounting Pronouncements – Recently Adopted
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. The ASU requires the annual financial statements to include consistent categories and greater disaggregation of information in the rate reconciliation, and income taxes paid disaggregated by jurisdiction. ASU 2023-09 is effective for the Company’s annual reporting periods beginning after December 15, 2024. The ASU may be applied either with a prospective method or a fully retrospective method of transition. Early adoption is permitted. The Company adopted the ASU prospectively on January 1, 2025 and it did not have a material impact on its operations.
(ac)
Recently Issued Accounting Pronouncements – Not Yet Adopted
In November 2024, the FASB issued ASU 2024-03, Disaggregation of Income Statement Expenses (Subtopic 220-40). The standard requires public business entities to disclose additional information about specific expense categories included in relevant income statement captions, including purchases of inventory, employee compensation, depreciation, amortization of intangible assets, and certain other expenses. The ASU 2024-03 is effective for the Company’s annual reporting periods beginning after December 15, 2026. Early adoption is permitted. The ASU may be applied either with a prospective method or a fully retrospective method of transition. Management is currently assessing the impact of this standard on the Company’s financial statements and will adopt the ASU on January 1, 2027.
In December 2025, the FASB issued ASU 2025-11, Interim Reporting (Topic 270): Narrow-Scope Improvements. The amendments clarify the applicability of the interim reporting guidance in Topic 270 and improve the navigability of interim disclosure requirements by providing a more comprehensive listing of disclosures required in interim financial statements. The ASU is effective for the Company’s interim periods in annual reporting periods beginning after December 15, 2027. Early adoption is permitted. The ASU may be applied either with a prospective method or a fully retrospective method of transition. Management is currently assessing the impact of this standard on the Company’s financial statements and will adopt the ASU on January 1, 2028.
No other new accounting pronouncements not yet effective are expected to have a material impact on the Company’s financial statements.
(3)
Fair Value of Financial Instruments
The following
fair value hierarchy table presents information about the Company’s assets and
liabilities measured at fair value on a recurring basis:
| |
| | | |
| | | |
| | | |
| |
Fair value measurement at
Reporting Date
|
| |
| |
(Level 1)
| |
| |
(Level 2)
| |
| |
(Level 3)
| |
|
As of December 31, 2025
|
| | | |
| | | |
| | | |
|
Liabilities:
|
| | | |
| | | |
| | | |
|
AMPA liability
|
| | | |
| | | |
| $ | - | |
|
Conversion Option liability
|
| | | |
| | | |
| $ | 2,014,000 | |
|
Warrants liabilities
|
| | | |
| | | |
| $ | 4,943,000 | |
The fair value of the AMPA
(see Note 9(f)) was measured at inception and at the December 31, 2025
reporting period using
Level 3 inputs. The fair value was deemed de minimis at both date of
issuance date and as of the December 31, 2025 reporting period.
The fair value of the
Conversion Option liability and Warrants liabilities related to the 2025 Notes
(see Note 7b) were measured at inception and at the December 31, 2025 reporting
period. The change in fair value between the periods was recognized as gains in
other (expense) income in the statements of comprehensive loss and are
summarized below:
| | | | | | | | | |
| |
| |
Fair Value of
Conversion Option
Liability
| |
| |
Fair Value of
Warrants Liabilities | |
| Fair value at issuance |
| $ | 2,517,000 | |
| $ | 5,674,000 | |
| Change in fair value (gain) |
| | (503,000 | ) |
| | (731,000 | ) |
|
Balance at December 31, 2025
|
| $ | 2,014,000 | |
| $ | 4,943,000 | |
The December 31, 2025 fair
value of the Conversion Option liability and Warrants liabilities were
estimated using the Black-Scholes option pricing model Level 3 inputs, with the
following assumptions:
| | | | | | | | | |
| | | | Conversion
Option
Liability | | | | Warrants
Liabilities | |
| Expected term (in years) | | | 2.84 | | | | 2.84 | |
| Risk‑free interest rate | | | 3.41 | % | | | 3.41 | % |
| Dividend yield | | | — | % | | | — | % |
| Expected volatility | | | 20 | % | | | 20 | % |
| Exercise price | | $ | 0.73 | | | $ | 0.81-1.10 | |
| Stock price | | $ | 0.58 | | | $ | 0.58 | |
| | | | | | | | | |
| Black-Scholes value | | $ | 0.047 | | | $ | 0.005-0.030 | |
The valuations of the
Conversion Option liability and Warrants liabilities at issuance were
calibrated such that the aggregate fair values of all instruments issued
together (including the 2025 Notes which are not subsequently remeasured at
fair value) equaled total gross proceeds received of $12 million in the
November 2025 Financing (see Note 7). This calibration resulted in an expected
volatility assumption of 20% which was also used in the valuation as of
December 31, 2025. If the Company used the historical volatility of its common
stock as the expected volatility assumption, the estimated value of the
Conversion Option liability and Warrants liabilities would be higher. Similarly, the expected terms of the
Conversion Option and Warrants were based on the Company’s option to mandate
conversion of the Notes upon achieving certain milestones (see Note 7) as well
as the expectation that the Warrants will be exercised upon a significant
increase in the price of the Company’s common stock. If the Company used the contractual term of
the Conversion Option and Warrants as the expected term, the estimated value of
the Conversion Option liability and Warrants liabilities would be higher.
We review the fair value hierarchy classification of our applicable assets and liabilities on a quarterly basis. Changes in the observability of valuation inputs may result in a reclassification for certain financial assets or liabilities. During the year ended December 31, 2025, there were no transfers between Level 1, Level 2 and Level 3.
The carrying amounts of cash and cash
equivalents, accounts receivable, customer deposits, accounts payable and
accrued expenses approximate fair value due to their short-term nature. As of
December 31, 2025, the fair value of the 2025 notes (excluding the conversion
option, see Note 7b), calculated using a discounted cash flow analysis using
Level 3 inputs, was approximately $3.8 million. As of December 31, 2024, the
fair value of the Company’s 2023 Notes (see Note 7a), calculated using a discounted
cash flow analysis using Level 3 inputs, was approximately $6.5 million. The
Company uses a Black-Scholes option valuation model to determine the grant date
fair value of employee stock options which uses Level 2 inputs. See Note 10 for
a description of inputs used.
(4)
Cash and Cash Equivalents
As of December 31, 2025 and 2024, money market funds included in cash and cash equivalents on the balance sheets were $7,110,655 and $3,451,761, respectively. The Company considers all highly-liquid investments with a remaining maturity of three months or less at the date of purchase to be cash.
(5)
Commitments and Contingencies
As of December 31, 2025, the Company has the right of use for its facilities located in Suwanee, GA under a long-term operating lease agreement, as amended (Lease Agreement), which expires in April 2029. The Company has the option (Extension Option) to extend the term for one consecutive term of five years each at the greater of the then current prevailing rental rate or current base rent rate, as agreed by both parties, and upon certain terms and conditions. The Company must provide written notice of its intent to exercise this extension option at between nine and 12 months prior to the expiration date of April 2029. Under the terms of the lease agreement, the Company’s monthly rent is subject to increases on an annual basis. As of December 31, 2025, the Company’s monthly rent payment was $52,614.
Operating right-of-use assets and lease liabilities consist of the following as of December 31:
| | | | | | | | | |
| |
| |
2025
| |
| |
2024
| |
|
Lease right-of-use assets
|
| $ | 1,297,121 | |
| | 1,805,543 | |
| | | | | | | | | |
|
Lease liabilities:
|
| |
2025
| |
| |
2024
| |
|
Lease liabilities – current portion
|
| $ | 487,624 | |
| | 517,967 | |
|
Lease liabilities – long-term portion
|
| | 1,030,476 | |
| | 1,518,100 | |
|
Total
|
| $ | 1,518,100 | |
| | 2,036,067 | |
As of December 31, 2025 and
2024, the weighted-average discount rate for all operating leases with initial
terms of more than one year was approximately 10% and the weighted-average
remaining term for operating leases was 3.3 years and 4.3 years, respectively.
The operating lease agreement for the Company’s facility includes non-lease costs, such as common area maintenance, which are recorded as variable lease costs. Operating lease expenses are included in general and administrative expenses in the Company’s statements of comprehensive loss and are summarized as follows for the years ending December 31:
| | | | | | | | | |
|
Lease cost:
|
| |
2025
| |
| |
2024
| |
|
Operating lease cost
|
| $ | 619,796 | |
| | 619,796 | |
|
Short-term lease cost
|
| | 7,765 | |
| | 7,765 | |
|
Variable lease cost
|
| | 139,646 | |
| | 148,109 | |
|
Total
|
| $ | 767,207 | |
| | 775,670 | |
The following table summarizes the Company’s undiscounted cash payment obligations for its operating lease liabilities with initial terms of more than twelve months as of December 31, 2025:
|
Operating leases:
|
| | | |
|
2026
|
| $ | 654,514 | |
|
2027
|
| | 680,694 | |
|
2028
|
| | 707,922 | |
|
2029
|
| | 243,835 | |
|
2030
|
| | — | |
|
Total undiscounted lease payments -operating leases
|
| | 2,286,965 | |
|
Less: imputed interest
|
| | (768,865 | ) |
|
Lease liability
|
| | 1,518,100 | |
|
Less: current portion of lease liability
|
| | (487,624 | ) |
|
Lease liability, less current portion
|
| $ | 1,030,476 | |
(b)
Clinical Trial Agreements (CTAs)
As part of the regulatory approval process for taking its products to market or conducting post-market clinical studies to support marketing efforts for products with regulatory clearance, the Company enters into a CTA to compensate each participating medical institution and investigator. Generally, upon executing a CTA with a participating medical institution or investigator, the Company pays a fee for institutional review board (IRB) approval that usually requires annual renewals and one-time site startup costs. As individual participants are enrolled in the clinical trial by the participating medical institution or investigator, the Company pays certain per subject fees according to the CTA for the duration of the trial. Expenses incurred in connection with these CTA activities are expensed as services are provided and are included in research and development expenses on the accompanying statements of comprehensive loss.
Occasionally, the Company may be a party to legal claims or proceedings of which the outcomes are subject to significant uncertainty. In accordance with ASC 450, Contingencies, the Company will assess the likelihood of an adverse judgment for any outstanding claim as well as ranges of probable losses. When it has been determined that a loss is probable and the amount can be reasonably estimated, the Company will record a liability. For the years ended December 31, 2025 and 2024, there were no material legal contingencies requiring accrual or disclosure.
The Company, as permitted under Delaware law and in accordance with its bylaws, indemnifies its officers and directors for certain events or occurrences, subject to certain limits, while the officer or director who is or was serving at the Company’s request in such capacity. The Company entered into employment agreements with its officers, which provides for indemnification protection in the executive’s capacity as an officer for actions taken within the scope of employment. The maximum amount of potential future indemnification is unlimited; however, the Company has obtained director and officer insurance that limits its exposure. The Company believes the fair value for these indemnification obligations is minimal. Accordingly, the Company has not recognized any liabilities relating to these obligations as of December 31, 2025 and 2024.
AFCO Credit Corporation (AFCO)
In July 2025, the Company executed a promissory note with AFCO to finance certain insurance premiums totaling $367,450, requiring the Company to pay $31,672 in a down payment and make monthly installment payments. The annual interest rate was 7.44% and the monthly installment is $31,672, which represents principal and interest. The Note was paid in full without penalty in November 2025.
Interest expense in connection with the AFCO Promissory Note was $8,614 for the year ended December 31, 2025.
(7)
Convertible Notes with Warrants
(a) Convertible
Notes with Warrants (November 2023 Financing)
On
November 21, 2023, the Company issued (i) senior unsecured convertible notes in
an aggregate principal amount of $6,850,000 (“2023 Notes”), convertible into
shares of common stock at a conversion price of $1.18 per share, (ii) Series A
Warrants to purchase up to an aggregate of 5,805,083 shares of common stock at
an exercise price of $1.18 per share, and (iii) Series B Warrants, together
with the Series A Warrants, and, together with the convertible notes, to
purchase up to an aggregate of 5,805,083 shares of common stock at an exercise
price of $1.475 per share. The financing resulted in aggregate gross proceeds
of $6,850,000, before $525,144 of issuance costs.
The 2023 Notes accrued
interest at a rate of 6.0% per annum, payable annually, in cash or shares of
common stock at the Company’s option. In November 2024, the Company paid
$111,000 of accrued interest in cash and $300,000 accrued interest in 315,790
shares of common stock. In November 2025, the Company paid $407,841 in accrued
interest in 516,256 shares of common stock.
As
noted above, the 2023 Notes were convertible into shares of common stock at the
election of the holder at any time at an initial conversion price of $1.18. The
conversion feature did not meet the requirements for separate accounting and
was not accounted for as a derivative instrument. As of December 31, 2024, the
2023 Convertible Notes had not been converted into shares of common stock. In
April 2025, $85,000 of convertible notes were converted into 72,033 shares of
common stock.
The
2023 Notes matured on November 21, 2025, and were repaid in full by the Company
with proceeds from the November 2025 Financing discussed below. No gain or loss was recorded upon repayment
of the 2023 Notes.
Warrants
The Series A Warrants are exercisable immediately and expire five years from the date of issuance. The Company has the right to call the exercise of the Series A Warrants if the closing price of the common stock exceeds 200% of the Series A Exercise Price for 10 consecutive trading days and the daily dollar trading volume of the common stock exceeds $1,000,000 per day during the same period and certain equity conditions are satisfied.
The Series B Warrants expired in November 2024.
The Series A Warrants are classified as a component of permanent equity because they are freestanding financial instruments that are legally detachable and separately exercisable from the shares of common stock from which they are issued, are immediately exercisable, do not embody an obligation for the Company to repurchase its shares, and permit the holders to receive a fixed number of shares of common stock upon exercise.
The Series A and Series B
Warrants were valued using the Black-Scholes option pricing model with the
following assumptions:
| | | | | | | | | |
| | | | Series A Warrants | | | | Series B Warrants | |
| Expected term (in years) | | | 5 | | | | 1 | |
| Risk‑free interest rate | | | 4.55 | % | | | 5.24 | % |
| Dividend yield | | | — | % | | | — | % |
| Expected volatility | | | 104.89 | % | | | 113.84 | % |
| Exercise price | | $ | 1.18 | | | $ | 1.48 | |
| Stock price | | $ | 0.95 | | | $ | 0.95 | |
| | | | | | | | | |
| Black-Scholes value | | $ | 0.55 | | | $ | 0.28 | |
The
November 2023 Financing proceeds of $6,850,000 net of $525,144 in issuance costs
were allocated to the convertible notes and Series A and Series B Warrants
using the relative fair value method. The proceeds allocated to the warrants
was $2,219,165 and was recorded as a component of stockholders’ equity.
For the year ended December 31, 2025, the Company recognized total interest expense on the 2023 Notes of $1,794,113, including coupon interest expense of $363,315, amortization of debt discount and issuance costs of $1,430,798. For the year ended December 31, 2024, the Company recognized total interest expense on the convertible notes of $1,603,575, including coupon interest expense of $411,001 amortization of debt discount and issuance costs of $1,192,574. The effective interest rate for the discount amortization was 32.3%. The Notes and accrued interest, net of unamortized discount costs was $47,841 and $5,406,228 as of December 31, 2025 and 2024, respectively. See Note 3 for further detail regarding fair value of the 2023 Notes.
(b) Convertible Notes with Warrants
(November 2025 Financing)
In November 2025, the Company
issued (i) senior secured convertible notes (“2025 Notes”) in an aggregate
principal amount of $12,000,000, convertible into shares of 16,378,563 common
stock at a conversion price of $0.73 per share, (ii) Series A-1 Warrants to
purchase up to an aggregate of 16,378,563 shares of common stock at an exercise
price of $0.81 per share, subject to adjustments, (iii) Series B-1 Warrants to
purchase an aggregate of 16,378,563 shares of common stock at an exercise price
of $0.92 per share, subject to adjustments, and (iv) Series C-1 Warrants to
purchase an aggregate of 16,378,563 shares of common stock at an exercise price
of $1.10 per share, subject to adjustments (Series A-1 Warrants, Series B-1
Warrants and Series C-1 Warrants are collectively the “Warrants”). The
financing resulted in aggregate gross proceeds of $12,000,000, before $718,528
of issuance costs.
The 2025 Notes accrue interest at a rate of 8.5% per annum, payable annually in-kind by increasing the principal balance outstanding. The Company granted a security interest in
substantially all of its assets to the holders of the 2025 Notes. The 2025 Notes mature in November 2035. The Company has a call option where it may voluntarily redeem the 2025 Notes after seven and one half years from Note issuance at 112% of the then-outstanding principal.
The 2025 Notes are convertible into shares of common stock at the election of the holder at any time (“Conversion Option”) at an initial conversion price of $0.73 (“Conversion Price”). The Conversion Price is adjustable proportionally for certain events including stock splits, reverse stock splits and stock dividends (each a “Share Combination Event”). Additionally, upon a Share Combination Event, the conversion price will also be further adjusted to the lowest dollar volume-weighted average price (VWAP) of the Company’s common stock during the five-day period following any Share Combination Event, but only if the conversion price (after the standard proportional adjustment) is higher than the market price.
The Company may require
conversion of the 2025 Notes, in whole or in part, if at any time (i) a
Registration Statement is effective, (ii) for the immediately preceding thirty
(30) consecutive trading days, the VWAP has exceeded 450% of the then-effective
Conversion Price on each such trading day, (iii) the total daily trading volume
of the Common Stock is at least $1,500,000.00, and (iv) certain equity
conditions are satisfied (effective registration statement is available,
sufficient authorized shares are available, etc.). The holders of the 2025
Notes may require prepayment of principal and accrued interest upon certain
Events of Default (failure to timely pay amounts owed under the 2025 Notes,
failure to deliver shares upon conversion of the 2025 Notes or exercise of the
related Warrants, failure to maintain an effective registration statement
related to the common shares underlying the 2025 Notes and related Warrants,
etc.), and the Company must redeem the 2025 Notes if involuntary bankruptcy
proceedings are initiated against the Company and not dismissed within 60 days
or if the Company voluntarily initiates bankruptcy. Additionally, in the event
of Fundamental Transaction, the Company must redeem the 2025 Notes, and if the
Fundamental Transaction occurs in the first 5 years of note issuance, the
redemption will include a premium.
If the Company offers future rights such as convertible securities, options, or warrants to purchase common stock to common stockholders (“Purchase Rights”), the holder/investor of the 2025 Notes are likewise entitled to receive such Purchase Rights as if the Notes were converted and the corresponding shares of common stock were outstanding.
As of December 31, 2025, the 2025 Notes have not been converted into shares of common stock.
Warrants
The Series A-1 Warrants are exercisable immediately and expire ten years from the date of issuance. The Series B-1 Warrants are exercisable immediately and expire at the earlier of (i) the tenth anniversary of the issuance of the Warrants, or (ii) 45 days after the Company discloses two consecutive quarters of at least $3.25 million in sales. The Series C-1 Warrants are exercisable immediately and expire at the earlier of (i) the tenth anniversary of the issuance of the Warrants, or (ii) 45 days after the Company discloses FDA approval of FemBloc.
The exercise price of the Warrants are adjustable proportionally in the event of a Share Combination Event. Additionally, similar to the Conversion Option in the 2025 Notes, upon a Share Combination Event, the exercise prices of the Warrants will also be further adjusted to the lowest VWAP of the Company’s common stock during the five-day period following any Share Combination Event, but only if the exercise price (after the standard proportional adjustment) is higher than the market price. The number of Warrants exercisable will also be adjusted such that the aggregate exercise price remains constant.
In the event of a Fundamental Transaction within the Company’s control, the holders of the Warrants are entitled to put the unexercised portion of the Warrants to the Company in exchange for cash at their then Black-Scholes value. If the Fundamental Transaction is outside of the Company’s control, the holders of the Warrants may receive consideration for the unexercised portion of the Warrants equal to the then Black-Scholes value in the same form (cash or equity) as other common shareholders pursuant to the Fundamental Transaction.
If the Company offers future rights such as convertible securities, options, or warrants to purchase common stock to common stockholders (“Purchase Rights”), the holders of the Warrants are likewise entitled to receive such Purchase Rights as if the Warrant were exercised and the corresponding shares of common stock were outstanding.
As of December 31, 2025 the Warrants have not been exercised.
The 2025 Notes
and Warrants are freestanding financial instruments that are legally detachable
and separately exercisable. Due to the adjustments to the conversion price of
the 2025 Notes and the exercise prices of the Warrants upon a Share Combination
Event beyond a standard proportional adjustment (but only if the market price
of the Company’s common stock is lower than the Conversion Price of the 2025
Notes or exercise prices of the Warrants) and due to the requirement that
shareholder approval be obtained for conversion and exercises that would exceed
19.99% of the outstanding shares of the Company, both the Conversion Option and
the Warrants were determined to be not indexed to the Company’s common stock.
As a result, at issuance, the Conversion Option was bifurcated from the 2025
Notes, and both the Conversion Option and Warrants were recognized as
derivative liabilities on the Company’s balance sheet. The Conversion Option
and Warrants will be remeasured at fair value every reporting period. The Notes
will be accreted to their maturity value based on the effective interest
method. The accretion is recorded as a credit to interest expense and was
($43,748) for the year ended December 31, 2025. The effective interest rate of
the 2025 Notes is approximately 21.9%.
See Note 3 for
further detail regarding fair value of the 2025 Notes, Conversion Option and
Warrants liabilities.
The following summarizes the carrying values of the November
2025 and November 2023 Convertible Notes as of December 31, 2025 and 2024:
| |
|
Stated
Interest
Rate
|
|
|
2025
|
|
|
2024
|
|
|
November 2023 Convertible Notes,
including related parties
|
|
|
6.0 |
% |
|
$ |
— |
|
|
$ |
6,850,000 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| Issuance of November 2025 Convertible Notes |
|
|
8.5 |
% |
|
|
12,000,000 |
|
|
|
— |
|
|
PIK interest on November 2025 Notes
|
|
|
|
|
|
|
132,139 |
|
|
|
— |
|
|
Less
unamortized discount and debt issuance costs
|
|
|
|
|
|
|
(8,953,275 |
) |
|
|
(1,443,772 |
) |
|
Total Convertible Notes Payable
|
|
|
|
|
|
|
3,178,864 |
|
|
|
5,406,228 |
|
|
Convertible
Notes Payable, short term
|
|
|
|
|
|
|
— |
|
|
|
(5,406,228 |
) |
|
Convertible Notes Payable
|
|
|
|
|
|
$ |
3,178,864 |
|
|
$ |
— |
|
No amounts
are due under the 2025 Notes until they mature in November 2035. In March 2026, the Company modified the 2025
Notes and related Warrants to remove the non-standard adjustment to the
conversion price and exercise prices upon a Share Combination Event (See Note
16).
(8) Income
Taxes
The
Company adopted ASU 2023-09, Income Taxes (Topic 740):
Improvements to Income Tax Disclosures on January 1, 2025 on a prospective
basis. Accordingly, the presentation of certain prior year disclosures below
differs from 2025 disclosures.
Income
tax expense consists of the following:
| | | | | |
|
Income
tax expense from continuing operations
|
| |
2025
| |
|
Current
tax expense
|
| | | |
|
Federal
|
| $ | — | |
|
State
and local
|
| | 1,297 | |
|
Total
current tax expense
|
| $ | 1,297 | |
| |
| | | |
|
Deferred
tax expense
|
| | | |
|
Federal
|
| | (4,205,724 | ) |
|
State
and local
|
| | 13,844 | |
|
Total
deferred tax expense
|
| $ | (4,191,880 | ) |
| |
| | | |
| Valuation allowance |
| | 4,191,880 | |
| |
| | | |
|
Total
income tax expense
|
| | | |
|
Federal
|
| | — | |
|
State
and local
|
| | 1,297 | |
|
Total
income tax expense
|
| $ | 1,297 | |
The
current tax provisions and deferred tax provisions, as reflected in the
financial statements are as follows as of December 31, 2024:
| | | | | |
| |
|
|
2024
| |
|
Current
federal taxes
|
|
$ | — | |
|
Current
state taxes
|
|
| 9,602 | |
|
Current
tax provision
|
|
| 9,602 | |
|
Deferred
federal taxes
|
|
| (4,433,940 | ) |
|
Deferred
state taxes
|
|
| (342,310 | ) |
|
Valuation
allowance change
|
|
| 4,776,250 | |
|
Deferred
tax provision
|
|
| — | |
|
Total
income tax expense provision
|
|
$ | 9,602 | |
As further described in Note
2, Summary of Significant Accounting Policies, the Company has elected
to prospectively adopt the guidance in ASU No. 2023-09, Income Taxes (Topic
740): Improvements to Income Taxes Disclosures, or ASU 2023-09. The
following table is a reconciliation of the federal statutory rate to the
Company’s effective rate for the year ended December 31, 2025:
| | | | | | | | | |
| | | | | | | | | |
| |
| |
2025 |
|
| |
| |
Total
| |
| |
%
| |
| |
| |
| |
| | | |
|
Loss from continuing operations
before income tax expense
|
| $ |
(18,626,590 | ) |
| | | |
| |
| |
| |
| | | |
|
Tax provision at U.S.
federal statutory rate
|
| |
(3,911,584 | ) |
| | 21.00 | % |
|
Federal
|
| |
| |
| | | |
|
Permanent differences
|
| |
(73,174 | ) |
| | 0.40 | % |
|
Research and development credit
|
| |
(415,511 | ) |
| | 2.20 | % |
|
Other deferred adjustments
|
| |
194,817 | |
| | (1.00 | )% |
|
Valuation allowance
|
| |
4,205,724 | |
| | (22.60 | )% |
|
Changes in unrecognized tax benefits
|
| |
| |
| | | |
|
State income taxes, net of federal
income tax benefit
|
| |
1,025 | |
| | 0.00 | % |
|
Income tax expense
|
| $ |
1,297 | |
| | —
| % |
The following is a
reconciliation of income tax expense at the statutory federal income tax rate
and income taxes as for the year ended December 31, 2024 in accordance with
guidance prior to the adoption of ASU 2023-09:
| |
| |
2024
| |
|
Federal income tax at statutory federal rate
|
| | 21 | % |
|
Permanent differences
|
| | — | |
|
Research and development credit
|
| | 3 | |
|
Other deferred adjustments
|
| | — | |
|
State income tax expense (net of federal benefit)
|
| | 2 | |
|
Valuation allowance
|
| | (26 | ) |
|
Effective tax rate
|
| | — | % |
Under the prospective
adoption of ASU 2023-09, income taxes paid by jurisdiction as of December 31,
2025, are as follows:
| | | | | |
| |
| |
2025
| |
|
U.S. Federal
|
| $ |
— | |
|
New
York
|
| |
2,000 | |
|
California
|
| |
1,600 | |
|
New
Hampshire
|
| |
1,100 | |
|
Georgia
|
| |
1,000 | |
|
New
Jersey
|
| |
500 | |
|
Massachusetts
|
| |
456 | |
|
Other
States
|
| |
150 | |
|
Total
U.S. State and Local
|
| |
6,806 | |
|
Total
income taxes paid
|
| $ |
6,806 | |
Deferred tax assets
(liabilities) consisted of the following as of December 31, 2025 and 2024:
| | | | | | | | | |
| |
| |
2025
| |
| |
2024
| |
|
Deferred
tax asset arising from
|
| |
| |
| | | |
|
Net
operating loss carryforwards
|
| $ |
26,318,782 | |
| | 23,030,273 | |
|
Accrued
expenses (compensation)
|
| |
109,914 | |
| | 138,806 | |
|
Intangibles
|
| |
98,355 | |
| | 93,426 | |
|
Property
and equipment
|
| |
67,159 | |
| | 80,313 | |
|
Research
and development expense capitalization
|
| |
3,835,472 | |
| | 3,326,621 | |
|
Research
and development tax credits
|
| |
4,139,328 | |
| | 3,839,058 | |
|
Share-based
compensation expense
|
| |
375,773 | |
| | 257,891 | |
|
Lease
liabilities
|
| |
344,986 | |
| | 471,733 | |
|
Other
|
| |
10,796 | |
| | 5,210 | |
|
Deferred
tax asset
|
| |
35,300,565 | |
| | 31,243,331 | |
|
Deferred
tax liability arising from:
|
| |
| |
| | | |
| Uniform capitalization |
| |
— | |
| | (11,092 | ) |
|
Right-of-use
assets
|
| |
(294,769 | ) |
| | (418,323 | ) |
|
Deferred
tax liability
|
| |
(294,769 | ) |
| | (429,415 | ) |
|
Valuation
allowance
|
| $ |
35,005,796 | |
| | 30,813,916 | |
|
Net
deferred tax asset
|
| $ |
— | |
| | — | |
Deferred income taxes reflect the net tax effects
of temporary differences between the carrying amount of assets and liabilities
for financial reporting purposes and the amounts used for income tax purposes,
and the Company is required to reduce its deferred tax assets by a valuation
allowance if it is more likely than not that some or all of its deferred tax
assets will not be realized. Management must use judgment in assessing the
potential need for a valuation allowance, which requires an evaluation of both
negative and positive evidence. The weight given to the potential effect of
negative and positive evidence should be commensurate with the extent to which
it can be objectively verified. In determining the need for and amount of the
valuation allowance, if any, the Company assesses the likelihood that it will
be able to recover its deferred tax assets using historical levels of income,
estimates of future income and tax planning strategies. As a result of
historical cumulative losses, the Company determined that, based on all
available evidence, there was substantial uncertainty as to whether it will
recover recorded net deferred taxes in future periods. Accordingly, the Company
recorded a valuation allowance against all of its net deferred tax assets as of
December 31, 2025 and 2024. The change in valuation allowance was $4,191,880 and
$4,776,250 for the years ended December 31, 2025 and 2024, respectively.
Beginning on January 1, 2022, the Tax Cuts and Jobs Act,
enacted in December 2017, eliminated the option to deduct research and
development expenditures in the current period and requires taxpayers to
capitalize and amortize U.S.-based and non-U.S. based research and development
expenditures over five and fifteen years, respectively. This legislation does
not impact the Company’s current tax obligations.
As
of December 31, 2025 and 2024,
respectively, the Company has $120,129,845 and $104,880,492 of federal net
operating loss carryforwards and $4,139,328 and $3,723,817 of federal research
and experimentation tax credits, respectively, and state net operating loss
carryforwards of $20,275,840 and $17,675,024, respectively. The utilization of
such net operating loss carryforwards and the realization of tax benefits in
future years depend predominately upon having taxable income. Under the
provisions of the Internal Revenue Code, certain substantial changes in the
Company’s ownership may result in a limitation on the amount of net operating
loss carryforwards and tax credit carryforwards that may be used in future
years.
The
Company’s net operating losses may be subject to Section 382 of the Internal
Revenue Code which provide for a limitation on the annual use of net operating
losses following certain ownership changes that could limit the Company’s
ability to utilize these carryforwards. The Company has completed an analysis
covering the period February 19, 2004 through December 31, 2018, to determine
if such ownership changes have occurred and concluded it was more likely than
not that there were changes in ownership during the period, with the most
recent change of ownership occurring on December 16, 2016. Further analyses
will be performed prior to recognizing the benefits of any losses or credits in
the financial statements, and the Company is in the process of determining the
limitations that Section 382 will have on the Company’s net operating loss
carryforwards and research credits. In general, the annual use limitation
equals the aggregate value of the Company’s stock at the time of the ownership
change multiplied by a specified tax-exempt interest rate.
The following schedule
indicates the expiration year, as of December 31, for the Company’s federal net
operating loss carryforwards available to future years without taking into
account any Section 382 limitations as of December 31, 2025:
|
2026
|
| $ |
1,213,130 | |
|
2027
|
| |
2,082,043 | |
|
2028
|
| |
2,536,605 | |
|
2029
|
| |
2,235,045 | |
|
2030
|
| |
4,132,949 | |
|
2031
|
| |
3,160,709 | |
|
2032
|
| |
3,533,521 | |
|
2033
|
| |
2,987,848 | |
|
2034
|
| |
2,516,728 | |
|
2035
|
| |
4,777,558 | |
|
2036
|
| |
4,503,474 | |
|
2037
|
| |
6,869,819 | |
|
2038
|
| |
11,194,631 | |
|
Indefinitely
|
| |
68,385,785 | |
|
Total
|
| $ |
120,129,845 | |
The FASB issued
authoritative guidance on accounting for uncertainty in income taxes, which
clarifies the accounting for income taxes, by prescribing a minimum recognition
threshold that a tax position is required to meet before recognition in the
financial statements. The guidance also provides direction on recognition,
measurement, classification, interest, and penalties, accounting in interim
periods, disclosure, and transition. Management has determined there are no
uncertain tax positions. Accordingly, these financial statements do not include
any adjustments or disclosures related to uncertain tax positions.
(9) Stockholders’
Equity
The Company’s Board of
Directors approved in January 2017 the Tenth Amended and Restated Certificate
of Incorporation authorizing the Company to issue for all classes of stock
169,000,000 shares at $0.001 par value per share, of which 95,853,558 shares were
designated common stock and 73,146,442 shares were designated Preferred Stock.
In June 2021, the
Company issued 2,650,000 shares of common stock in connection with the
Company’s IPO of its common stock at $13.00 per share. Net proceeds to the
Company, after deducting underwriting discounts, commissions, and legal
expenses, was $31,613,500. Offering costs incurred by the Company were
$2,016,143, which include legal expenses incurred and paid by the underwriters
of $425,000. Immediately prior to the closing of the IPO, all of the
convertible Series A preferred stock and redeemable convertible Series B and
Series C preferred stock automatically converted into 8,116,343 shares of
common stock.
The Company filed an
Eleventh Amended and Restated Certificate of Incorporation (the “Amended and
Restated Certificate”) with the Secretary of State of the State of Delaware in
connection with the completion of the IPO on June 22, 2021. The Amended and Restated
Certificate amends and restates the Company’s existing certificate of
incorporation in its entirety to, among other things: (i) authorize 200,000,000
shares of common stock; (ii) eliminate all references to the
previously-existing series of preferred stock (Series A, B and C); and (iii)
authorize 10,000,000 shares of undesignated preferred stock that may be issued
from time to time by the Board in one or more series.
On July
1, 2022, we entered into an Equity Distribution Agreement (the “Equity
Distribution Agreement”) with Piper Sandler & Co. (“Piper Sandler” or the
“Sales Agent”) and filed a related prospectus establishing an “at-the-market”
facility, pursuant to which we may offer and sell shares of our common stock
from time to time through the Sales Agent. In October 2023, the Sales Agent was
authorized to sell shares for aggregate proceeds up to $16.7 million at current
market prices until all shares are sold. During the year ended December 31, 2025,
we sold approximately 5.6 million shares of common stock for aggregate proceeds
of $7.0 million. In December 2025, we filed a new prospectus pursuant to which
we may sell up to $9.8 million under the Equity Distribution Agreement. In
July 2025, the Company filed a prospectus to sell up to $150 million in stock,
debt securities and warrants. As of December 31,
2025, the amount we are authorized to sell is subject to baby-shelf
limitations. The Company may only offer one-third of the public float during
and trailing twelve-month period. Based on our public float, as of the date of
the filing of this Annual Report on Form 10-K, we are only permitted to utilize
a shelf registration statement, including the registration statement under
which our at the market offering is operated, subject to Instruction I.B.6 to
Form S-3, which is referred to as the “baby shelf” rule. For so long as our
public float is less than $75.0 million, we may not sell more than the
equivalent of one-third of our public float during any 12 consecutive months
pursuant to the baby shelf rules.
(a) Common
Stock
The holders of the
common stock shall have the exclusive right to vote for the election of
directors and on all other matters requiring stockholder action, each
outstanding share entitling the holder thereof to one vote on each matter
properly submitted to the stockholders of the Company for their vote; provided,
however, that, except as otherwise required by law, holders of common stock, as
such, shall not be entitled to vote on any amendment to the Amended and
Restated Certificate (or on any amendment to a certificate of designations of
any series of preferred stock) that alters or changes the powers, preferences,
rights or other terms of one or more outstanding series of preferred stock if
the holders of such affected series of preferred stock are entitled to vote,
either separately or together with the holders of one or more other such
series, on such amendment pursuant to this Amended and Restated Certificate (or
pursuant to a certificate of designations of any series of preferred stock).
Dividends may be
declared and paid or set apart for payment upon the common stock out of any
assets or funds of the Company legally available for the payment of dividends,
but only when and as declared by the Board of Directors or any authorized
committee thereof.
In the event of our
liquidation or dissolution, the holders of common stock are entitled to receive
proportionately the Company’s net assets available for distribution to
stockholders after the payment of all debts and other liabilities and subject
to the prior rights of any outstanding preferred stock. Holders of common stock
have no preemptive, subscription, redemption, or conversion rights.
(b) Convertible
Preferred Stock
As of December 31, 2025,
no shares of convertible preferred stock have been issued and/or outstanding,
and no dividends have been declared or paid since inception.
(c) Redeemable
Convertible Preferred Stock
As of December 31, 2025,
no shares of redeemable convertible preferred stock have been issued and/or
outstanding, and no dividends have been declared or paid since inception.
(d) Preferred
Stock
Under the terms of the
Amended and Restated Certificate, the Company’s Board of Directors is
authorized to direct the Company to issue shares of preferred stock in one or
more series without stockholder approval. The Board of Directors has the
discretion to determine the rights, preferences, privileges and restrictions,
including voting rights, dividend rights, conversion rights, redemption
privileges and liquidation preferences, of each series of preferred stock.
The purpose of
authorizing the Board of Directors to issue preferred stock and determine its
rights and preferences is to eliminate delays associated with a stockholder
vote on specific issuances. The issuance of preferred stock, while providing
flexibility in connection with possible acquisitions, future financings, and
other corporate purposes, could have the effect of making it more difficult for
a third-party to acquire, or could discourage a third-party from seeking to
acquire, a majority of the outstanding voting stock. As of December 31, 2025
and 2024, no shares of preferred stock are outstanding.
(e) June
2025 Financing
In June 2025, the Company
sold 3,600,000 shares of common stock in an underwritten public offering at
$0.85 per share. The Company also sold 1,686,275 shares of common stock in a
separate concurrent private placement at a price of $0.85 per share to certain
existing institutional stockholders and a price of $1.02 per share to certain
directors and officers. The offering resulted in aggregate gross proceeds of
$4,510,001, before $804,940 of transaction costs.
Additionally,
common warrants to purchase 105,726 shares of common stock were issued to the
underwriter as a commission for services performed. The underwriter warrants
are exercisable beginning December 2, 2025, have a 5-year term and an exercise
price of $1.0625 per share. The warrants are classified as a component of
permanent equity because they are freestanding financial instruments that are
legally detachable and separately exercisable from the shares of common stock
from which they are issued, are immediately exercisable, do not embody an obligation
for the Company to repurchase its shares and permit the holders to receive a
fixed number of shares of common stock upon exercise. The warrants were valued
at $56,035 using Black-Scholes assumptions. As of December 31, 2025, 105,726
underwriter warrants remain outstanding.
(f) Any Market Purchase Agreement (“AMPA”)
On June 30, 2025, the Company
entered into an Any Market Purchase Agreement (“Purchase Agreement” or “AMPA”) with
Alumni Capital LP (“Alumni”) whereby the Company has the right, but not
obligation, to sell to Alumni up to an aggregate of $10 million in shares of common
stock in a series of purchases until December 31, 2026. The Company may elect
that Alumni purchase up to $1 million in shares of common stock (or up to $5
million if mutually agreed) at either (i) the lowest traded price for four
previous business days, multiplied by 90% or (ii) up to the lesser of (a) $1
million in shares of common stock or (b) 100% of the average daily trading
volume of common stock for previous two business days at lowest daily dollar
volume-weighted-average price, multiplied by 97%. The Company is limited to
issuances to Alumni or 19.99% of the shares of common stock outstanding
immediately prior to the execution of the Purchase Agreement. The Purchase
Agreement allows the Company to raise equity on a periodic basis at its
discretion depending on a variety of factors including market conditions, the
trading price of the common stock, and use of proceeds for operating
activities. Due to certain pricing and settlement provisions, the Purchase
Agreement includes an embedded put option contract. The Company accounts for
the Purchase Agreement as a derivative, with a fair value remeasured each reporting period. The difference between
the discounted purchase price and fair value of the shares will be expensed in
the period the transaction occurs as a non-cash, nonoperating financing cost. See Note 3 for further detail regarding fair value of the AMPA.
The Company incurred a 2% commitment fee of $200,000
and an additional $86,295 in expenses as of December 31, 2025, which are
included as other expense in the statements of comprehensive loss.
(g) August 2025 Financing
On August 25, 2025, the
Company entered into a securities purchase agreement pursuant to which the
Company sold (i) 10,434,586 shares of common stock in a public offering and to
certain Company officers, (ii) pre-funded warrants to purchase 11,750,000 shares
of common stock, and (iii) common warrants to purchase 22,184,586 shares of
common stock. Additionally, common warrants to purchase 443,692 shares of
common stock were issued to the underwriter as compensation for services
performed.
The pre-funded warrants,
common warrants and underwriter warrants were exercisable immediately following
the closing date of the offering. The pre-funded warrants have an unlimited
term and an exercise price of $0.0001 per share. The common warrants have a
5-year term and an exercise price of $0.36 per share, except for common
warrants sold to certain Company officers, which have an exercise price of
$0.5151 per share. The underwriter warrants have a 5-year term and exercise
price of $0.45 per share. The offering resulted in aggregate gross proceeds of
$7,998,826, before $930,132 of transaction costs.
The pre-funded warrants and
common warrants are classified as a component of permanent equity because they
are freestanding financial instruments that are legally detachable and
separately exercisable from the shares of common stock with which they were issued,
are immediately exercisable, do not embody an obligation for the Company to
repurchase its shares, and permit the holders to receive a fixed number of
shares of common stock upon exercise.
The common stock was valued
at $3,770,001, based on the Company’s stock price. The pre-funded warrants and
common warrants were valued at $4,230,000 and $5,324,301, respectively, using
the following Black-Scholes assumptions:
| | | | | | | | | | | | | |
| | | | Pre-funded warrants | | | | Common warrants | | | | Underwriter warrants | |
| | | | | | | | | | | | | |
| Expected term (in years) | | | 4 | | | | 4 | | | | 4 | |
| Risk‑free interest rate | | | 3.64 | % | | | 3.64 | % | | | 3.64 | % |
| Dividend
yield | | | — | % | | | — | % | | | — | % |
| Expected volatility | | | 90.63 | % | | | 90.63 | % | | | 90.63 | % |
| Exercise price | | $ | 0.0001 | | | | $0.36- $0.5151 | | | $ | 0.45 | |
| Stock price | | $ | 0.36 | | | | $0.36- $0.5151 | | | $ | 0.36 | |
| Black-Scholes value | | $ | 0.36 | | | | $0.24-0.27 | | | $ | 0.22 | |
The net proceeds of
$7,068,694 were allocated to the common stock, pre-funded warrants and common
warrants using the relative fair value method and were recorded to
stockholders’ equity.
As of December 31, 2025, 6,750,000
pre-funded warrants, 12,965,476 common warrants and 443,692 placement agent
warrants remain outstanding.
(h) Warrants
As of December 31, 2025,
the Company has warrants outstanding to purchase shares of common stock, which
are all exercisable in whole or in part, with expiration dates as follows:
| | | | | | | | | | | | | | | | | |
|
Expiration date
|
| |
2025
|
| |
2024
|
|
Number
of
warrants
outstanding
| |
| |
Exercise
price
| |
| |
Number
of
warrants
outstanding
| |
| |
Exercise
price
| |
|
April
2025
|
| |
— | |
| $ |
— | |
| |
55,177 | |
| $ |
7.25 | |
|
December
2026
|
| |
128,934 | |
| $ |
9.45 | |
| |
128,934 | |
| $ |
9.45 | |
|
January
2027
|
| |
12,705 | |
| $ |
9.45 | |
| |
12,705 | |
| $ |
9.45 | |
|
April
2028
|
| |
68,809 | |
| $ |
1.53 | |
| |
68,809 | |
| $ |
1.53 | |
|
November
2028
|
| |
5,805,083 | |
| $ |
1.18 | |
| |
5,805,083 | |
| $ |
1.18 | |
|
June
2030
|
| |
105,726 | |
| $ |
1.06 | |
| |
— | |
| $ |
— | |
|
August
2030
|
| |
13,409,168 | |
|
$
|
0.36-.5151 | |
| |
— | |
| $ |
— | |
|
November
2035
|
| |
49,135,689 | |
|
$
|
0.81-1.10 | |
| |
— | |
| $ |
— | |
|
Pre-funded
warrants
|
| |
6,750,000 | |
| |
no
expiration
| |
| |
— | |
| |
— | |
|
Total
|
| |
75,416,114 | |
| |
| |
| |
6,070,708 | |
| |
| |
In
April 2025, 55,177 warrants to purchase shares of common stock expired.
(i) Dividends
As of December 31, 2025,
no dividends have been declared or paid since inception.
(10) Equity
Incentive Plans
(a) Stock
Option Plans – Prior to the IPO
Prior to the IPO in June
2021, the Company maintained two equity incentive plans, the 2004 Stock
Incentive Plan, as amended, or 2004 Plan, and the 2015 Stock-Based Incentive
Compensation Plan, or 2015 Plan, which provided the Company’s employees,
non-employee directors, consultants and independent contractors the opportunity
to participate in the equity appreciation of the business through the receipt
of stock options to purchase shares of common stock. New grants ceased being
made under the 2004 Plan upon the adoption of the 2015 Plan; however,
outstanding stock options under the 2004 Plan may continue to be exercised in
accordance with their terms. The Company adopted the 2015 Plan in April 2015,
which contains substantially similar terms and conditions as the 2004 Plan. The
2015 Plan initially had 1,176,681 shares of common stock reserved for issuance
under the 2015 Plan and was administered by the compensation committee of the
Board of Directors. Upon the closing of the IPO, no further awards will be made
under the 2015 Plan; however, outstanding stock options under the 2015 Plan may
continue to be exercised in accordance with their terms.
(b) Stock
Option Plans – Post the IPO
In June
2021, in connection with the IPO, the 2021 Equity Incentive Plan (2021 Plan)
became effective, which was adopted by the Board of Directors in February 2021
and approved by stockholders in March 2021. The 2021 Plan is
administered by our compensation committee.
Under the 2021 Plan, the
Company may grant awards in respect of shares of common stock to employees,
consultants, and non-employee directors pursuant to option awards, stock
appreciation right, or SAR, awards, restricted stock awards, restricted stock
unit, or RSU, awards, performance stock awards, performance stock unit, or PSU,
awards, and other stock-based awards.
The total number of
shares of common stock available for awards under the 2021 Plan is 1,111,111,
provided that such number shall be automatically increased on each January 1,
beginning on January 1, 2022, by 4% of the outstanding number of shares of common
stock on the immediately preceding December 31 or such lesser number of shares
as determined by the Board of Directors. The aggregate number of shares of our
common stock that will be available for issuance under awards granted pursuant
to the 2021 Plan shall also be increased by the number of shares underlying the
portion of an award granted under the 2015 Plan that is cancelled, terminated
or forfeited or lapses after the effective date of the 2021 Plan. No more than
1,111,111 shares of common stock issued under the 2021 Plan may be issued
pursuant to the exercise of incentive stock options (ISO), provided that such
number shall be automatically increased on each January 1, beginning on January
1, 2022, by the lesser of 4% of the outstanding number of shares of common
stock on the immediately preceding December 31 or 555,555 shares of common
stock. Shares of common stock issued by us in connection with the assumption or
substitution of outstanding grants or under certain stockholder approved plans
from an acquired company shall not reduce the number of shares of common stock
available for awards under the 2021 Plan. Shares of common stock underlying the
portion of an award that is forfeited or otherwise terminated for any reason
whatsoever, in any case, without the issuance of shares of common stock, will
be added back to the number of shares of common stock available for grant under
the 2021 Plan. No non-employee director may be granted awards under the 2021
Plan in any one calendar year covering a number of shares of common stock that
have a fair market value on the grant date in excess of $350,000 in the first
calendar year of such non-employee director’s initial service as a non-employee
director and $200,000 in any other calendar year of such non-employee
director’s service as a non-employee director.
Options granted under
the 2021 Plan may be either ISOs or non-qualified stock options. The price at
which shares of common stock may be purchased upon exercise shall be determined
by the compensation committee but shall not be less than the fair market value
of one share of common stock on the date of grant, or, in the case of an ISO
granted to a ten-percent stockholder, less than 110% of the fair market value
of a share of common stock on the date of grant. The compensation committee may
grant options that have a term of up to 10 years, or, in the case of an ISO
granted to a ten-percent stockholder, five years. The award agreement shall
specify the exercise price, term, vesting requirements, including any
performance goals, and any other terms and conditions applicable to the granted
option. Unless otherwise provided in an award agreement or an effective
employment, consulting, severance or similar agreement with us or a subsidiary,
upon a participant’s termination of service for any reason, the unvested portion
of each award of options granted generally will be forfeited with no
compensation due to the participant.
Activity under the stock
option plans was as follows:
| |
| |
Number
of
options
| |
| |
Weighted
average
exercise
price
| |
| |
Aggregate
Intrinsic Value
| |
|
Outstanding
at December 31, 2023
|
| | 2,102,030 | |
| $ |
2.00 | |
| | | |
|
Granted
|
| | 1,359,821 | |
| |
1.07 | |
| | | |
|
Forfeited
|
| | (487,632 | ) |
| |
1.84 | |
| | | |
|
Outstanding
at December 31, 2024
|
| | 2,974,219 | |
| $ |
1.60 | |
| | 847,758 | |
|
Granted
|
| | 1,639,915 | |
| |
1.08 | |
| | | |
|
Forfeited
|
| | (783,743 | ) |
| |
1.15 | |
| | | |
|
Outstanding
at December 31, 2025
|
| | 3,830,391 | |
| $ |
1.47 | |
| | 92,127 | |
| |
| | | |
| |
| |
| | | |
| Vested and exercisable at December 31, 2025 |
| | 2,021,208 | |
| $ |
1.87 | |
| | 75,115 | |
The
intrinsic value represents the amount by which the market price of the
underlying stock at December 31, 2025 exceeds the exercise price of an option.
Stock options granted
under the 2021 Plan for the years ended December 31, 2025 and 2024 were as
follows:
| |
| |
2025
| |
| |
2024
| |
|
Employee
|
| | 1,569,515 | |
| | 1,286,921 | |
|
Nonemployee
|
| | 70,400 | |
| | 72,900 | |
|
Total
|
| | 1,639,915 | |
| | 1,359,821 | |
The Black-Scholes
weighted-average assumptions for all stock option awards granted during 2025
and 2024 were as follows:
| | | | | | | | | | | | | | | | | |
| | | | 2025 | | | | 2024 | |
| | | | 2021 Plan | | | | Inducement
Grants | | | | 2021 Plan | | | | Inducement
Grants | |
| Fair Value of Awards | | $ | 0.83 | | | | 0.69 | | | | 0.91 | | | | 0.94 | |
| Grant Price | | $ | 1.08 | | | | 0.9 | | | | 1.07 | | | | 1.1 | |
| | | | | | | | | | | | | | | | | |
| Expected term (in years) | | | 6.04 | | | | 6.25 | | | | 6.20 | | | | 6.25 | |
| Risk‑free interest rate | | | 4.34 | % | | | 4.03 | % | | | 4.04 | % | | | 4.10 | % |
| Dividend yield | | | — | % | | | — | % | | | — | % | | | — | % |
| Expected volatility | | | 91.17 | % | | | 88.50 | % | | | 109.61 | % | | | 109.64 | % |
The intrinsic values
represent the dollar value of the exercised stock options whereby the fair
market value of the underlying common stock exceeded the exercise price of the
stock option as of the exercise date. There were
no options exercised during 2025 and 2024.
The options outstanding
and vested and currently exercisable by exercise prices as of December 31, 2025
were as follows:
| | | | | | | | | | | | | | | | | | | | | | |
| | Options outstanding | | | Options vested and exercisable |
| | Exercise price | | | Outstanding (in shares) | | | | Weighted average remaining life years | | | | Number of options vested | | | | Weighted average Exercise price | | | | Weighted average remaining life years | |
| $ | 0.47 - 0.49 | | | 1,021,300 | | | | 6.31 | | | | 832,500 | | | $ | 0.49 | | | | 6.04 | |
| | 0.50 - 0.80 | | | 523,596 | | | | 7.84 | | | | 159,381 | | | | 0.76 | | | | 6.80 | |
| | 0.81 - 1.33 | | | 1,579,479 | | | | 8.37 | | | | 450,359 | | | | 1.10 | | | | 6.80 | |
| | 1.34 - 1.74 | | | 204,846 | | | | 1.06 | | | | 87,435 | | | | 1.67 | | | | 1.62 | |
| | 1.75 - 3.14 | | | 103,550 | | | | 2.40 | | | | 93,913 | | | | 2.59 | | | | 2.01 | |
| | 3.15 - 3.60 | | | 237,614 | | | | 1.34 | | | | 237,614 | | | | 3.24 | | | | 1.34 | |
| | 3.61 - 13.00 | | | 160,006 | | | | 4.73 | | | | 160,006 | | | | 9.94 | | | | 4.73 | |
| | | | | 3,830,391 | | | | 6.61 | | | | 2,021,208 | | | | 1.87 | | | | 5.23 | |
As of
December 31, 2025, the total number of shares of common stock reserved for
future awards under the 2021 Plan is 3,701,726.
Since 2023, the Company has granted 250,000 inducement grants, a stock option grant to certain employees for the right to purchase shares, which were approved by the Compensation Committee. The inducement grants vest in equal installments over four years provided the employee remains employed by the Company on the vesting date.
The inducement grants are summarized as follows:
| | | | | | | | | | | | | |
| | | Number of
options | | | Weighted
average
exercise
price | | | Weighted
average
remaining
life years | |
| Outstanding at December 31, 2024 | | | 250,000 | | | $ | 1.89 | | | | 8.07 | |
| Granted | | | 100,000 | | | | 0.90 | | | | 10.48 | |
| Forfeited | | | (212,500 | ) | | | 1.02 | | | | | |
| Outstanding at December 31, 2025 | | | 137,500 | | | $ | 2.52 | | | | 4.53 | |
| Vested and exercisable at December 31, 2025 | | | 112,500 | | | $ | 2.42 | | | | 4.17 | |
The Company uses the Black‑Scholes option pricing model to determine the fair value of stock awards granted to employees and non-employees. The determination of the fair value of share‑based payment awards granted using a pricing model is affected by the Company’s stock price as well as the assumptions regarding a number of complex and subjective variables as follows:
The expected term of stock options represents the period where the stock options are expected to remain outstanding. The Company’s historical share option exercise experience does not provide a reasonable basis upon which to estimate an expected term because of a lack of sufficient data. Therefore, the Company estimates the expected term for all options granted by using the simplified method provided by Compensation—Stock Compensation (Topic 718): Improvements to Accounting for Stock Compensation (ASC 718), which calculates the expected term as the average of the time-to-vesting and the contractual life of the options. The contractual term for options awarded since inception is 10 years for employees and non-employees.
(ii)
Risk‑Free Interest Rate
The risk‑free interest rate is based on U.S. Treasury zero‑coupon issues with remaining terms similar to the expected term on the options.
The Company has not declared or paid any cash dividends from inception through December 31, 2025 and does not plan to pay any cash dividends in the foreseeable future, and, therefore, used an expected dividend yield of zero in the valuation model.
Expected volatility measures the amount that a stock price has fluctuated or is expected to fluctuate during a period. The Company determines volatility based on an analysis of comparable companies.
The Company accounts for forfeitures as they occur.
(e)
Employee Stock Purchase Plan (ESPP)
In June 2021, in connection with the IPO, the ESPP became effective upon adoption by the Board of Directors in February 2021 and the stockholders approved the 2021 ESPP in March 2021. The ESPP is administered by the compensation committee.
The total number of shares of common stock available for purchase under the ESPP is 166,666, provided that such number is automatically increased on January 1 of each calendar year, from January 1, 2022 through January 1, 2031 by the least of (i) 1.0% of the total number of shares of common stock outstanding on December 31 of the immediately preceding calendar year, (ii) 222,222 shares of common stock or (iii) a number determined by the board of directors that is less than the foregoing clauses (i) and (ii).
Under the ESPP, the Company may specify offerings with durations of not more than 27 months and may specify shorter purchase periods within each offering. Each offering will have one or more purchase dates on which shares of common stock will be purchased for employees participating in the offering. An offering may be terminated under certain circumstances. No employee may purchase more than 12,254 shares of common stock under the ESPP during any offering period. Unless otherwise determined by the board of directors, shares of common stock will be purchased for accounts of employees participating in the ESPP at a price per share equal to the lower of (i) 85% of the fair market value of a share of common stock on the last date of an offering period or (ii) 85% of the fair market value of a share of common stock on the first day of such offering period.
As of December 31, 2025, 234,801 shares of common stock have been purchased under the ESPP to date, and the total number of shares of common stock reserved for future awards under the ESPP is 618,735.
(f)
Stock‑Based Compensation Expense
Stock‑based compensation expense recognized is based on the value of the portion of stock option awards that is ultimately expected to vest on a straight-line basis. Stock‑based compensation expense recognized in the Company’s statements of comprehensive loss during the years ended December 31, 2025, and 2024 includes compensation expense for stock‑based awards based on the fair value estimated in accordance with the provisions of ASC 718.
The following table shows the stock-based compensation expense related to vested stock option grants to employees and non-employees awarded under the stock plans and inducement grants by financial statement line item on the accompanying statements of comprehensive loss:
| | | | | | | | | |
| |
|
2025
| |
|
2024
| |
|
Research and development
|
| $ | 224,714 | |
| | 179,911 | |
|
Sales and marketing
|
| | 52,537 | |
| | 48,379 | |
|
General and administrative
|
| | 512,894 | |
| | 215,857 | |
|
Total share-based compensation expense
|
| $ | 790,145 | |
| | 444,147 | |
As of December 31, 2025, the
remaining amount of stock‑based compensation expense that is expected to be
recognized in future periods for employees and non-employees is $1,138,799
which includes $155,222 of compensation expense to be recognized upon achieving
a certain performance condition. The $983,577 of unrecognized expense is
expected to be recognized over a weighted-average period of 2.5 years.
The Company has a 401(k) defined contribution plan covering substantially all employees, meeting certain eligibility requirements. The Company has no required matching or other contribution requirements. For the years ended December 31, 2025 and 2024, the Company contributed $122,917 and $97,876 of voluntary employer matching contributions.
(12)
Related‑Party Transactions
In November 2023, the Company
issued unsecured convertible notes and accompanying Series A and Series B
Warrants (see Note 7a). The transaction included issuance of a $5 million
convertible note and 4,237,288 Series A and 4,237,288 Series B Warrants to
PharmaCyte Biotech, Inc. The interim CEO, President and Director of PharmaCyte
Biotech, Inc., Joshua Silverman, served on the Company’s board of directors until
March 2026. The Series B Warrants expired in November 2024. In November 2024, the Company paid PharmaCyte accrued interest on the convertible note of
$300,000 in equity of 315,790 common shares. In November 2025, the Company paid
PharmaCyte accrued interest on the convertible note of $300,000 in equity of
379,747 common shares. In November 2025, the convertible notes principal
balance of $5 million was repaid at maturity. No amounts were due to Pharmacyte
as of December 31, 2025. Convertible notes of $5M and accrued interest of
$32,500 were due to Pharmacyte as of December 31, 2024.
In the June 2025 Financing (see Note 9e), certain executive officers and a director of the Company of the purchased an aggregate of 98,040 shares of the Company’s common stock in a private placement at a price of $1.02 per share, for total gross proceeds of $100,001. Other investors in the private placement purchased common stock at a discounted price of $0.85 per share. No amounts were due to or from these related parties in connection with this transaction as of December 31, 2025.
In the August 2025 Financing (see Note 9g), certain executive officers of the Company purchased 87,363 common shares, with a common warrant, with a combined price of $0.5151. Gross proceeds from the transaction totaled $45,001. The price equaled the market price of the Company’s stock on the applicable purchase date. The common warrants have a 5-year term and an exercise price of $0.5151 per share. The warrants remain outstanding as of December 31, 2025. No amounts were due to or from these related parties in connection with this transaction as of December 31, 2025.
In the November 2025 Financing (see Note 7b), certain executive officers and a director of the Company purchased senior secured convertible notes with a combined principal balance of $100,000, convertible into common shares at a price of $0.73 per share. Additionally, in connection with the convertible notes, the officers and director were issued 136,488 Series A-1 Warrants, 136,488 Series B-1 Warrants and 136,488 Series C-1 Warrants at conversion exercise prices of $0.81, $0.92 and $1.10 per share, respectively. As of December 31, 2025, $100,000 in convertible notes were due to the officers and director, in addition to accrued interest of $1,251. All A-1, B-1 and C-1 warrants issued to the officers and director remain outstanding as of December 31 2025.
During the years ended December 31, 2025 and 2024, a family member of the CEO was employed and earned total cash compensation of $196,000 and $168,000, respectively. The compensation paid is consistent with market standards for comparable positions.
(13)
Net Loss per Share Attributable to Common Stockholders
The following table sets forth the computation of the basic and diluted net loss per share for the years ended December 31:
| | | | | | | | | |
| |
|
2025
| |
|
2024
| |
| |
| | | |
| | | |
|
Net loss attributable to common stockholders, basic and diluted
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
| |
| | | |
| | | |
| Weighted-average common shares and pre-funded warrants outstanding- basic and diluted |
| | 39,549,218 | |
| | 22,267,695 | |
|
Net loss per share attributable to common stockholders, basic and diluted
|
| $ | (0.47 | ) |
| | (0.85 | ) |
Since the shares underlying the November 2025 pre-funded warrants were issuable for little consideration of $0.0001 per share, they were considered outstanding for both basic and diluted loss per share from the date of issuance.
The following potentially dilutive securities have been excluded from the computations of diluted weighted-average shares outstanding because they would be anti-dilutive for the years ended December 31:
| | | | | | | | | |
| |
|
2025
| |
|
2024
| |
|
Options to purchase common stock
|
| | 3,967,891 | |
| | 3,224,219 | |
|
Warrants to purchase common stock, in connection with April 2023 financing
|
| | 68,809 | |
| | 68,809 | |
|
Warrants to purchase common stock, in connection with November 2023 financing
|
| | 5,805,083 | |
| | 5,805,083 | |
|
Warrants to purchase common stock, in connection with June 2025 financing
|
| | 105,726 | |
| | — | |
|
Warrants to purchase common stock, in connection with August 2025 financing
|
| | 13,409,168 | |
| | — | |
| November 2025 convertible
notes and PIK interest, if converted |
| | 16,558,917 | |
| | — | |
|
Warrants to purchase common stock, in connection with November 2025 financing
|
| | 49,135,689 | |
| | — | |
|
Warrants to purchase common stock
|
| | 141,639 | |
| | 196,816 | |
|
Total potential shares
|
| | 89,192,922 | |
| | 9,294,927 | |
In accordance with FASB ASC Topic 280, Segment Reporting, the Company has determined that it operates as a single business segment, which is the development and commercialization of therapeutic and diagnostic products that service women’s reproductive health needs (infertility and permanent birth control).
The determination of a single business segment is consistent with the financial information regularly provided to the Company’s chief operating decision maker (“CODM”). As a single reportable segment entity, the Company’s segment performance measure is net loss attributable to shareholders. The measurement of segment assets is reported on the balance sheet as total assets. The Company’s CODM is its Chief Executive Officer and Chief Financial Officer, who together review and evaluate net income for purposes of assessing performance, making operating decisions, allocating resources, and planning and forecasting for future periods.
Significant segment expenses, as provided to the CODM are as follows:
| | | | | | | | | |
| |
|
2025
| |
|
2024
| |
|
Sales
|
| $ | 2,293,313 | |
| | 1,629,108 | |
|
Cost of sales (excluding depreciation expense)
|
| | 872,400 | |
| | 544,903 | |
|
Research and development expense
|
| | 2,072,771 | |
| | 2,217,610 | |
|
Other research and development expenses
|
| | 5,504,933 | |
| | 5,998,933 | |
|
Total research and development expense
|
| | 7,577,704 | |
| | 8,216,543 | |
|
Sales and marketing expense
|
| | 4,443,807 | |
| | 4,030,150 | |
|
General and administrative expense
|
| | 6,646,037 | |
| | 6,325,999 | |
|
Depreciation and amortization expense
|
| | 342,034 | |
| | 297,318 | |
|
Total operating expenses
|
| | 19,009,582 | |
| | 18,870,010 | |
|
Total other expense
|
| | (1,037,921 | ) |
| | (1,021,221 | ) |
|
Loss before income taxes
|
| | (18,626,590 | ) |
| | (18,807,026 | ) |
|
Income tax expense
|
| | 1,297 | |
| | 9,602 | |
|
Net loss
|
| $ | (18,627,887 | ) |
| | (18,816,628 | ) |
The company incurred the following Other Expense, net for the years ended December 31, 2025 and 2024:
| |
| | | |
| | | |
| |
| Year Ended December 31, | |
| |
|
2025
| |
|
2024
| |
|
Interest income
|
| $ | 101,760 | |
| | 582,354 | |
|
Amortization of debt issuance costs and discount for 2025 and 2023 notes
|
| | (1,387,050 | ) |
| | (1,192,574 | ) |
|
Interest expense
|
| | (504,069 | ) |
| | (411,001 | ) |
|
Debt issuance costs for AMPA
|
| | (286,295 | ) |
| | — | |
|
Change in fair value of Conversion Option liability
|
| | 503,000 | |
| | — | |
|
Change in fair value of Warrants liabilities
|
| | 731,000 | |
| | — | |
|
Other expenses
|
| | (196,267 | ) |
| | — | |
|
Total other expense, net
|
| $ | (1,037,921 | ) |
| | (1,021,221 | ) |
In February and March 2026,
the Company sold 623,911 shares under the at-the-market facility, resulting in
gross cash proceeds of $352,922.
In February 2026, $76,452 of the 2025 Notes and accrued interest were converted into 103,429 shares of common stock.
In March 2026, the Company
modified the 2025 Notes and related Warrants to remove the non-standard
adjustment to the conversion price and exercise prices, respectively, upon a
Share Combination Event. As
consideration for this modification, the Company issued the holders of the 2025
Notes and related Warrants new Series D-1 Warrants which allow the holders to
purchase an aggregate number of 16,378,563 shares of the Company’s common
stock. The exercise price of the Series
D-1 Warrants is $0.58 per share, and the Warrants expire in March 2029.
Item 9.
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
Not applicable.
Evaluation of Disclosure Controls and Procedures
The term “disclosure controls and procedures,” as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act, refers to controls and procedures that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company’s management, including its principal executive officer and principal financial officer, as appropriate to allow timely decisions regarding required disclosure. As required by Rules 13a-15(b) and 15d-15(b) of the Exchange Act, our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures as of the end of the period covered by this Annual Report on Form 10-K. Based on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective as of December 31, 2025.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act). Our management, under the oversight of our board of directors, evaluated the effectiveness of our internal control over financial reporting as of December 31, 2025 based on the framework in Internal Control-Integrated Framework (2013), issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this evaluation, our management concluded that our internal control over financial reporting was effective as of December 31, 2025.
Our independent registered accounting firm will not be required to opine on the effectiveness of our internal control over financial reporting pursuant to the Sarbanes-Oxley Act until we are no longer an “emerging growth company” as defined in the JOBS Act.
Changes in Internal Control over Financial Reporting
There has been no change in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during the quarter ended December 31, 2025, that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Inherent Limitations on Effectiveness of Controls
Management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Our management, including our Chief Executive Officer and Chief Financial Officer, does not expect that our disclosure controls and procedures or internal control over financial reporting will prevent all errors and all fraud. A control system, no matter how well designed and implemented, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues within a company are detected. The inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple errors or mistakes. Controls can also be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the controls. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and may not be detected.
Rule 10b5-1 and Non-Rule 10b5-1 Trading Arrangements
During the twelve months ended December 31, 2025, none of our directors or officers adopted, terminated or modified a “Rule 10b5-1 trading arrangement” or “non-Rule 10b5-1 trading arrangement,” as defined in Item 408(a) of Regulation S-K of the Exchange Act.
Item 9C.
Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
None
Item 10.
Directors, Executive Officers and Corporate Governance.
The information required by this item is incorporated herein by reference to our definitive proxy statement for our 2026 Annual Meeting of Stockholders.
Code of Ethics
The board of directors has adopted a Code of Ethics applicable to all of our directors, officers and employees, including our principal executive officer, principal financial officer and principal accounting officer. A copy of the Code of Ethics is available on our website www.femasys.com.
Compensation of Directors and Executive Officers
The information required by this item is incorporated herein by reference to our definitive proxy statement for our 2026 Annual Meeting of Stockholders.
Item 12.
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
The information required by this item with respect to the security ownership of certain beneficial owners and the security ownership of management is incorporated herein by reference to our definitive proxy statement for our 2026 Annual Meeting of Stockholders.
Item 13.
Certain Relationships and Related Transactions, and Director Independence.
The information required by this item with respect to certain relationships and related transactions is incorporated herein by reference to our definitive proxy statement for our 2026 Annual Meeting of Stockholders.
Item 14.
Principal Accounting Fees and Services.
The information required by this item is incorporated herein by reference to our definitive proxy statement for our 2026 Annual Meeting of Stockholders.
| | | | | |
|
Exhibit
Number
|
Description of Document
|
Schedule/Form
|
Exhibit
|
Filing Date
|
| |
|
|
|
|
|
|
Eleventh Amended and Restated Certificate of Incorporation of Femasys Inc.
|
Form 8-K
|
3.1
|
June 22, 2021
|
|
|
Amended and Restated Bylaws of Femasys Inc.
|
Form 8-K
|
3.2
|
June 22, 2021
|
|
|
First Amendment to the Amended and Restated Bylaws of Femasys Inc., dated as of March 29, 2023
|
Form 8-K
|
3.1
|
March 30, 2023
|
|
|
Description of the Registrant’s Securities
|
Form 10-K
|
4.1
|
March 24, 2022
|
|
|
Form of Certificate of Common Stock
|
Form S-1
|
4.1
|
May 14, 2021
|
|
|
Form of indenture
|
Form S-3
|
4.3
|
July 1, 2022
|
|
|
Form of Pre-Funded Warrant
|
Form 8-K
|
4.1
|
April 20, 2023
|
|
|
Form of Common Stock Warrant
|
Form 8-K
|
4.2
|
April 20, 2023
|
|
|
Form of Placement Agent Warrant
|
Form 8-K
|
4.3
|
April 20, 2023
|
|
|
Form of Series A Warrant
|
Form 8-K
|
4.1
|
November 15, 2023
|
|
|
Underwriter Warrant
|
Form 8-K
|
4.1
|
June 2, 2025
|
|
|
Underwriter Common Stock Purchase Warrant
|
Form 8-K
|
4.1
|
August 27, 2025
|
|
|
Pre-Funded Common Stock Purchase Warrant
|
Form 8-K
|
4.2
|
August 27, 2025
|
|
|
Common Stock Purchase Warrant
|
Form 8-K
|
4.3
|
August 27, 2025
|
|
|
Form of Series A-1 Warrant
|
Form 8-K
|
4.1
|
November 7, 2025
|
|
|
Form of Series B-1 Warrant
|
Form 8-K
|
4.2
|
November 7, 2025
|
|
|
Form of Series C-1 Warrant
|
Form 8-K
|
4.3
|
November 7, 2025
|
|
|
Femasys Inc. 2021 Equity Incentive Plan, and forms of agreements thereunder
|
Form S-1
|
10.3
|
May 14, 2021
|
|
|
Femasys Inc. 2021 Employee Stock Purchase Plan
|
Form S-1
|
10.4
|
May 14, 2021
|
|
|
Amended and Restated Employment Agreement, by and between Femasys Inc. and Kathy Lee-Sepsick
|
Form S-1/A
|
10.6
|
June 14, 2021
|
|
|
Femasys Inc. Non-Employee Director Compensation Policy
|
Form S-1/A
|
10.11
|
June 14, 2021
|
|
|
Form of Indemnification Agreement between Femasys Inc. and its directors and officers
|
Form S-1
|
10.12
|
May 14, 2021
|
|
|
Master Services Agreement and Statement of Work for consulting services, effective August 12, 2021, by and between Femasys Inc. and Bespoke Medical Affairs Solutions, LLC
|
Form 10-Q
|
10.1
|
November 12, 2021
|
|
|
Employment Agreement, dated as of February 28, 2022, between Femasys Inc. and Dov Elefant
|
Form 8-K
|
10.1
|
February 24, 2022
|
|
|
Form of Inducement Stock Option Agreement
|
Form 8-K
|
10.2
|
February 24, 2022
|
| | | | | |
| 10.9 |
Equity Purchase Agreement dated July 1, 2022, between Femasys Inc. and Piper Sandler & Co. |
Form S-3 |
1.2 |
July 1, 2022 |
| 10.10 |
Purchase Agreement dated June 30, 2025, by and between Femasys Inc. and Alumni Capital LP. |
Form S-3 |
10.1 |
July 3, 2025 |
| 10.11 |
Separation Agreement with Daniel Currie |
Form 8-K |
10.1 |
October 17, 2025 |
|
|
Form of Senior Secured Convertible Note
|
Form 8-K
|
10.2
|
November 7, 2025
|
|
|
Form of Securities Purchase Agreement dated November 3, 2025 between Femasys Inc. and the other parties thereto
|
Form 8-K
|
10.1
|
November 7, 2025
|
|
|
Form of Registration Rights Agreement dated November 7, 2025 between Femasys Inc. and the other parties thereto
|
|
|
|
| |
|
|
|
|
|
|
Insider Trading Policy
|
|
|
|
|
|
Consent of KPMG LLP
|
|
|
|
|
|
Power of Attorney (included on signature page)
|
|
|
|
|
|
Certification of Principal Executive Officer Pursuant to Securities Exchange Act Rules 13a-14(a), as adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
|
|
|
|
|
|
Certification of Principal Financial Officer Pursuant to Securities Exchange Act Rules 13a-14(a), as adopted Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
|
|
|
|
| 32.1* |
Certification of Principal Executive Officer Pursuant to 18 U.S.C. Section 1350, as adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| 32.2* |
Certification of Principal Financial Officer Pursuant to 18 U.S.C. Section 1350, as adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
| 97.1 |
Clawback Policy |
Form 10-K |
97.1 |
March 28, 2024 |
| 101.INS* |
Inline XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document) |
|
|
|
| 101.SCH* |
Inline XBRL Taxonomy Extension Schema Document |
|
|
|
| 101.CAL* |
Inline XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
| 101.DEF* |
Inline XBRL Taxonomy Extension Definition Linkbase Document |
|
|
|
| 101.LAB* |
Inline XBRL Taxonomy Extension Label Linkbase Document |
|
|
|
| 101.PRE* |
Inline XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
| 104* |
Cover Page Interactive Data File (formatted as inline XRBL and contained in Exhibit 101) |
|
|
|
* Filed herewith
None.
Pursuant to the requirements of the Securities Act, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Suwanee, State of Georgia, on this 31 day of March 2026.
FEMASYS INC.
|
Dated: March 31, 2026
|
By: /s/ Kathy Lee-Sepsick
|
|
|
|
Kathy Lee-Sepsick
|
|
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Kathy Lee-Sepsick and Dov Elefant, and each of them as his or her true and lawful attorneys-in- fact and agents, each with the full power of substitution, for him or her and in his or her name, place or stead, in any and all capacities, to sign any and all amendments to this report, and to file the same, with exhibits thereto and other documents in connection therewith with the Securities and Exchange Commission, hereby ratifying and confirming all that said attorneys-in-fact, and each of them, or his or her substitute or substitutes may lawfully do or cause to be done by virtue hereof. Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | | |
|
Signature
|
|
Title
|
|
Date
|
| |
|
|
|
|
|
By: /s/ Kathy Lee-Sepsick
|
|
|
|
March 31, 2026
|
|
Kathy Lee-Sepsick
|
|
President and Chief Executive Officer (principal executive officer)
|
|
|
| |
|
|
|
|
|
By: /s/ Dov Elefant
|
|
|
|
March 31, 2026
|
|
Dov Elefant
|
|
Chief Financial Officer (principal financial and accounting officer)
|
|
|
| |
|
|
|
|
|
By: /s/ Charles Larsen
|
|
|
|
March 31, 2026
|
|
Charles Larsen
|
|
Chairperson of the Board of Directors
|
|
|
| |
|
|
|
|
|
By: /s/ Alistair Milnes
|
|
|
|
March 31, 2026
|
|
Alistair Milnes
|
|
Director
|
|
|
| |
|
|
|
|
|
By: /s/ Kenneth D. Eichenbaum
|
|
|
|
March 31, 2026
|
|
Kenneth D. Eichenbaum
|
|
Director
|
|
|
| |
|
|
|
|
|
By: /s/ Edward Uzialko, Jr.
|
|
|
|
March 31, 2026
|
|
Edward Uzialko, Jr.
|
|
Director
|
|
|
132