UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 10-K
(Mark One)
☑ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the year ended December 31, 2025
o TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period from to
Commission File Number 001-36362
BioLife Solutions, Inc.
(Exact name of registrant as specified in its charter)
| | | | | |
| Delaware | 94-3076866 |
(State or other jurisdiction of
incorporation or organization) | (IRS Employer
Identification No.) |
3303 Monte Villa Parkway, Suite 310, Bothell, Washington, 98021
(Address of registrant’s principal executive offices, Zip Code)
(425) 402-1400
(Telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading symbol | Name of exchange on which registered |
Common stock, par value $0.001 per share | BLFS | The Nasdaq Stock Market, LLC |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark whether the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☑ No o
Indicate by check mark whether the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No ☑
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☑ No o
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (S232.405 of this chapter) during the preceding 12 months (or for such shorter period that the Registrant was required to submit such said files). Yes ☑ No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☑ Accelerated filer o Non-accelerated filer o Smaller reporting company o Emerging Growth Company o
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☑
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes o No ☑
As of the registrant’s most recently completed second fiscal quarter, the aggregate market value of common equity (based on closing price on June 30, 2025 of $21.54 per share) held by non-affiliates was approximately $845 million.
As of February 19, 2026, 48.3 million shares of the registrant’s common stock were outstanding.
Table of Contents
References throughout this Annual Report on Form 10-K to “BioLife Solutions, Inc.”, “BioLife”, “we”, “us”, “our”, or the “Company” refer to BioLife Solutions, Inc. and its subsidiaries, taken as a whole, unless the context otherwise indicates.
FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K (“Form 10-K” or “Annual Report”) contains forward-looking statements within the meaning of the safe harbor provisions of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). All statements, other than statements of historical fact, in this Annual Report, including, without limitation, statements regarding our strategy, future operations, future operating expenses, future financial position, future revenue, projected costs, prospects, plans, intentions, expectations, goals and objectives may be forward‑looking statements. The forward-looking statements in this Annual Report do not constitute guarantees of future performance, and actual results could differ materially from those expressed or implied in any forward-looking statements. Forward-looking statements can be identified by words such as “believe,” “expect,” “anticipate,” “contemplate,” “estimate,” “project,” “forecast,” “would,” “may,” “should,” “will,” “could,” “can,” “potential,” “possible,” “proposed,” “plan,” “develop,” “opportunity,” “intend,” “initiative,” “target,” “maintain,” “continue,” “strive,” “progress,” “aim,” or similar expressions.
The forward-looking statements in this Annual Report include, but are not limited to, statements about:
•the development, production and commercialization of our products and our ability to maintain reliable, high-quality products;
•our ability to compete effectively against current technologies and develop and market products that are competitive in the continually changing technological landscape;
•the ability of our customers to integrate our products into their bioproduction workflow process for cell and gene therapies;
•our ability to successfully increase our customer’s product yield and efficacy;
•the determination that our products are not subject to U.S. Food and Drug Administration ("FDA") or other regulatory approvals and the possibility that we could be subject to regulatory approvals in the future;
•the potential utility of and market for our products and services;
•our ability to implement our business strategy and anticipated business and operations (including with respect to acquired businesses);
•our future financial and operational performance;
•our ability to protect our proprietary position and the validity and enforceability of our patents and trade secrets;
•our anticipated future growth strategy;
•the expected benefits and other statements relating to our divestitures and acquisitions, capital requirements, intellectual property, suppliers, joint venture partners, future financial and operating results;
•our ability to protect our information systems and networks and the proprietary and confidential information in our possession;
•the impact of adverse changes in economic, political and market conditions in the areas that we serve, the U.S. and globally, including but not limited to, disruption in our supply chain, inflation in pricing for key materials or labor, the imposition of trade tariffs or other adverse changes resulting from epidemics, pandemics, and outbreaks of contagious diseases, natural disasters, economic or political instability, terrorist attacks and wars, including the ongoing war in Ukraine, conflict in the Middle East, U.S. involvement in Venezuela, or other adverse geopolitical developments;
•interest rates and interest rate fluctuations and their potential impact on the general economy and our profitability;
•potentially adverse impacts of climate change including increased frequency and severity of natural disasters and extreme weather events such as hurricanes, tornados, wildfires (exacerbated by drought), flooding, and extreme heat
•the increase in laws and regulations aimed at reducing greenhouse gas emissions and other measures to mitigate the impact of climate change, and related customer expectations and other environmental liabilities;
•legislative, regulatory and tax law and/or policy developments;
•declines in revenue relative to historical levels that we are unable to offset;
•our ability to effectively manage our operations, operating expenses, capital expenditures, debt service requirements and cash paid for income taxes and liquidity; and
•the availability and terms of any capital financing agreements.
We intend that such forward-looking statements be subject to the safe harbors for such statements. These forward-looking statements are based on the current beliefs, expectations and projections of our management and speak only as of the date of this Annual Report or, in the case of documents referred to or incorporated by reference, the date of those documents. You should not place undue reliance on these forward-looking statements, which are subject to significant known and unknown risks, uncertainties and other factors, including those described in Part I, Item 1A, “Risk Factors” in this Annual Report which are in some cases, beyond our control and which could materially affect results. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. If underlying assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results may differ materially from current expectations and projections.
We do not plan to release publicly any revisions to these forward-looking statements to reflect new information, future events or changed circumstances or otherwise after the date of this Annual Report, except as may be required by law. If we update one or more forward-looking statements, no inference should be drawn that we will make additional updates with respect to such updated forward-looking statements or other forward-looking statements.
This Annual Report includes trademarks, tradenames and service marks that are the property of other organizations. Solely for convenience, trademarks and tradenames referred to in this Annual Report appear without the ® and ™ symbols, but those references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights, or that the applicable owner will not assert its rights, to these trademarks and tradenames.
SUMMARY RISK FACTORS
Our business is subject to numerous risks and uncertainties, including those described in Part I, Item 1A Risk Factors in this Annual Report. You should carefully consider these risks and uncertainties when investing in our common stock. The principal risks and uncertainties affecting our business include the following:
•If our products or the products of our competitors do not perform as expected or the reliability of the technology on which our products are based is questioned, we could experience lost revenue, delayed or reduced market acceptance of our products, increased costs, and damage to our reputation.
•We operate in a highly competitive industry and if we cannot compete effectively, our business, financial condition and operating results could be materially and adversely affected.
•Despite our increasingly diversified customer base, we depend on a limited number of customers and products in a limited number of market sectors. If we lose any of these large customers or if there are disruptions in the sales of these products, our net product revenue and operating results could decline significantly.
•We expect our operating results to fluctuate significantly from period to period.
•If intangible assets and goodwill become impaired, we may have to take significant charges against earnings.
•Our success will depend on our ability to attract and retain key personnel.
•Our business may be subject to product liability claims or product recalls, which could be expensive and could result in a diversion of management’s attention.
•Difficulties in manufacturing could have an adverse effect upon our expenses and our product revenues.
•We are dependent on our suppliers and third-party manufacturers, including single-source and sole-source suppliers, and disruptions in our supply chain could adversely affect our ability to manufacture and deliver products.
•While we are not currently subject to FDA or other regulatory approvals, if our products become subject to regulatory requirements, the manufacture and sale of our products may be delayed or prevented, or we may become subject to increased expenses.
•We and our customers are subject to various international governmental regulations. Compliance with or changes in such regulations may cause us to incur significant expenses, and if we fail to maintain satisfactory compliance with certain regulations, we may be forced to recall products and cease their manufacture and distribution, and we could be subject to civil or criminal penalties.
•Healthcare reform measures could adversely affect our business and financial results.
•Insurance coverage is increasingly difficult to obtain or maintain.
•We are and may become the subject of various claims, litigation or investigations which could have a material adverse effect on our business, financial condition, or results of operations or the price of our common stock.
•Our business and operations could be negatively affected by securities litigation or stockholder activism, which could impact the trading price and volatility of our common stock and may constrain capital deployment opportunities and adversely impact our ability to expand our business.
•Our acquisitions expose us to risks that could adversely affect our business, and we may not achieve the anticipated benefits of acquisitions of businesses or technologies.
•Our recent divestitures expose us to ongoing risks and uncertainties, one or more of which could have a material adverse effect on our business, financial condition, results of operations, cash flows, and/or stock price.
•Our proprietary rights may not adequately protect our technologies and products.
•Expiration of our patents may subject us to increased competition and reduce our opportunity to generate product revenue.
•We may not be able to protect our intellectual property rights throughout the world.
•We may incur substantial costs as a result of litigation or other proceedings relating to patent and other intellectual property rights, and we may be unable to protect our rights to, or use of, our technology.
•Even if we are granted a patent, in certain circumstances we may be unable to protect our rights to, or use of, our technology.
•We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
•Our inability to protect our information systems and networks and the proprietary and confidential information in our possession from continually evolving cybersecurity risks or other technological risks, including as a result of breaches of our associated third parties' information technology systems, could materially adversely impact our business, financial condition and results of operations, in addition to our reputation and relationships with our employees, customers, suppliers and business partners.
•The development, deployment and use of artificial intelligence ("AI") technologies present new risks and challenges that could adversely impact our business
•Our stock price and volume may be volatile, and purchasers of our securities could incur substantial losses.
•A significant percentage of our outstanding common stock is held by one stockholder, and this stockholder therefore has significant influence on us and our corporate actions.
•We have not paid dividends on our common stock in the past and do not expect to pay dividends in the foreseeable future. Any return on investment may be limited to the value of our common stock.
•Certain provisions in our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
•Our Amended and Restated Bylaws designate the Court of Chancery of the State of Delaware and U.S. federal district courts as the exclusive forums for certain types of actions and proceedings that may be initiated by our stockholders, which limits our stockholders’ ability to choose the judicial forum for disputes with us or our directors, officers, or other employees.
•Though our internal control over financial reporting for the year ended December 31, 2025 was concluded to be operating effectively, we have historically identified material weaknesses in our internal control over financial reporting. We have remediated our historical material weaknesses, though if we are unable to maintain an effective system of internal control over financial reporting or disclosure controls and procedures, we may not be able to accurately and timely report financial results or prevent fraud, and our ability to meet our reporting obligations and the trading price of our common stock could be negatively affected.
•Changes in tax laws and regulations, including international tax developments, could adversely affect our financial condition and results of operations.
•Our ability to use net operating loss ("NOL") and tax credit carryforwards and certain built-in losses to reduce future tax payments is limited by provisions of the Internal Revenue Code of 1986, as amended (the “Internal Revenue Code”), and it is possible that certain transactions or a combination of certain transactions may result in material additional limitations on our ability to use our NOL and tax credit carryforwards.
•Natural disasters, geopolitical unrest, war, terrorism, public health issues or other catastrophic events could disrupt the supply, delivery or demand of products, which could negatively affect our operations and performance.
•Tariffs and other trade policies could have a substantial impact on our business.
•Unfavorable currency exchange rate fluctuations may lead to lower operating margins or may cause us to raise prices for our products and services, which could result in reduced sales.
•Public health crises have adversely affected, and could in the future adversely affect, our business, financial condition, results of operations, and cash flows.
•Global climate change and related legal and regulatory developments could negatively affect our business, financial condition and results of operations.
PART I
ITEM 1. BUSINESS
The following discussion of our business contains forward-looking statements that involve risks and uncertainties (see the section entitled “Forward-Looking Statements” herein). Our actual results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including those factors set forth under “Risk Factors” and elsewhere in this Form 10-K.
Overview
We are a life sciences company that develops, manufactures, and markets bioproduction products and services which are designed to improve quality and de-risk biologic manufacturing, distribution, and transportation in the cell and gene therapy ("CGT") industry. Our products are used in basic and applied research and commercial manufacturing of biologic-based therapies. Customers use our products to maintain the health and function of biologic material during sourcing, manufacturing, and distribution.
We currently operate as one bioproduction products and services business which supports several steps in the biologic material manufacturing and delivery process. Our portfolio of tools and services focuses on biopreservation media and cell processing products. We have in-house expertise in cryobiology and the broader CGT workflow, and continue to evaluate opportunities to maximize the value of our product platforms for our extensive customer base through organic growth innovations, partnerships, and acquisitions.
Recent divestitures and acquisitions
On October 6, 2025, the Company entered into a Limited Liability Company Membership Interest Purchase Agreement (the “SAVSU Purchase Agreement”), by and between the Company and Peli BioThermal LLC, a Delaware limited liability company (“SAVSU Buyer”), for the sale by the Company of all of the issued and outstanding limited liability company membership interests (the “SAVSU Interests”) of SAVSU Cleo Technologies, LLC, a Delaware limited liability company ("SAVSU"), to SAVSU Buyer (the “SAVSU Divestiture”). SAVSU contained our evo cloud connected “smart” shipping container products that provided passive storage and transport for temperature-sensitive biologics and pharmaceuticals. Upon the execution of the SAVSU Purchase Agreement, the SAVSU business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented.
On April 4, 2025, pursuant to a Stock Purchase Agreement (the “PanTHERA Purchase Agreement”), by and among the Company, Casdin Partners Master Fund L.P. and each other person listed on Schedule A thereto (the “PanTHERA Sellers”), 2699979 Alberta LTD., an Alberta corporation and a wholly owned subsidiary of the Company (“PanTHERA Buyer Sub”), PanTHERA CryoSolutions Inc., an Alberta corporation (“PanTHERA”) and Dr. Jason Acker, solely in his capacity as Sellers’ Representative, the Company acquired the remaining 90% of the issued and outstanding shares of common stock of PanTHERA not owned by the Company from the PanTHERA Sellers (the “PanTHERA Transaction”). PanTHERA contains a patented Ice Recrystallization Inhibitor (“IRI”) GEN 2 cryopreservation technology that we expect to ultimately enhance the Company’s core capabilities in biopreservation and within the CGT market upon achievement of commercial viability.
On November 14, 2024, the Company entered into a Stock Purchase Agreement (the “CBS Purchase Agreement”), by and among the Company, Standex International Corporation, a Delaware corporation (“CBS Buyer”), and Arctic Solutions, Inc., a Delaware corporation and a wholly owned subsidiary of the Company (doing business as Custom Biogenic Systems, or “CBS”), for the sale by the Company of all of the issued and outstanding shares of common stock (the “CBS Shares”) of CBS to CBS Buyer (the “CBS Divestiture”). CBS contained our LN2 cryogenic freezers and other accessory products utilizing a dry storage method. Upon the execution of the CBS Purchase Agreement, the CBS business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented.
On November 12, 2024, the Company entered into a Stock Purchase Agreement (the “SciSafe Purchase Agreement”), by and among the Company, Subzero Purchaser Corp., a Delaware corporation (“SciSafe Buyer”), SciSafe, Inc., a Delaware corporation and an indirect, wholly owned subsidiary of the Company (“Seller”), and SciSafe, Inc., a New Jersey corporation and an indirect wholly owned subsidiary of the Company (“SciSafe”), for the sale by Seller of all of the issued and outstanding shares of common stock (the “SciSafe Shares”) of SciSafe to SciSafe Buyer (the “SciSafe Divestiture”). SciSafe contained our biological and pharmaceutical storage and cold chain logistics services. Upon the execution of the
SciSafe Purchase Agreement, the SciSafe business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented.
On April 17, 2024, the Company sold all of the issued and outstanding shares of common stock of Global Cooling, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Global Cooling”), to GCI Holdings Company, LLC, an Ohio limited liability company (“GCI Holdings”) pursuant to a Stock Purchase Agreement, dated April 17, 2024 (the “Global Cooling Purchase Agreement”), by and between the Company and GCI Holdings (the “Global Cooling Divestiture”). Global Cooling contained our portfolio of ultra-low temperature freezers. Upon the execution of the Global Cooling Purchase Agreement, the Global Cooling business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented.
The Company is presenting SAVSU, CBS, SciSafe, and Global Cooling within this Annual Report as discontinued operations for all periods presented within the Consolidated Balance Sheets and Consolidated Statements of Operations. The Consolidated Statements Of Comprehensive Loss, Consolidated Statements of Shareholders' Equity, and Consolidated Statements of Cash Flows are presented on a consolidated basis for both continuing operations and discontinued operations. All amounts, percentages, and disclosures for all periods presented in this Annual Report reflect only the continuing operations of the Company unless otherwise noted. See Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the divestitures described above.
Our products
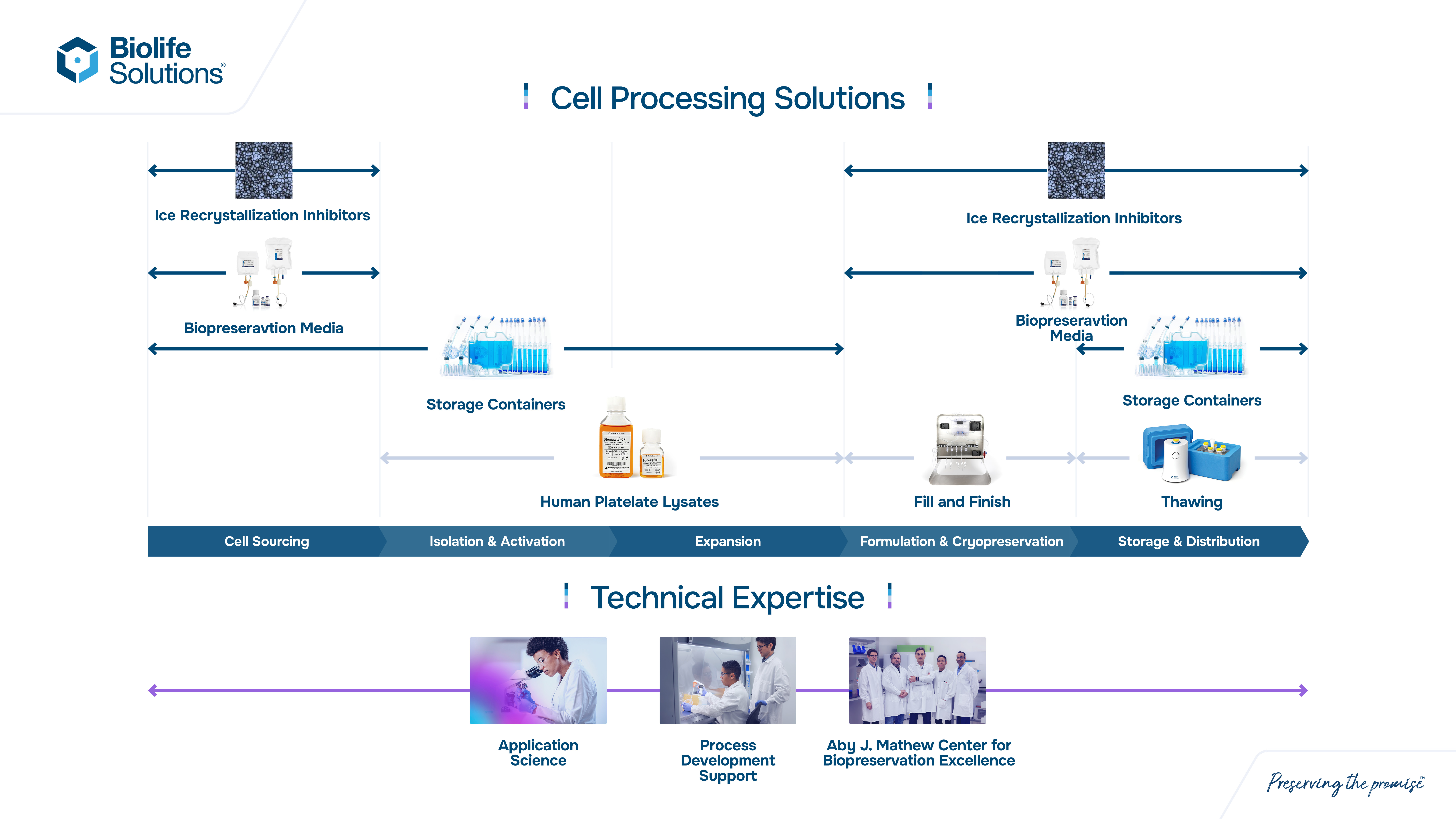
Our bioproduction products and services are comprised of one revenue line that contains three main offerings:
•Cell processing and other products
◦Biopreservation media
◦Human platelet lysate media (“hPL”), cryogenic and ultralow temperature containers, and automated cell-processing fill machines
◦Automated thawing devices
Cell processing
Biopreservation media
Our proprietary biopreservation media products, HypoThermosol® FRS and CryoStor® Freeze Media, are formulated to mitigate preservation-induced, delayed-onset cell damage and death which result when cells and tissues are subjected to reduced temperatures. Our technology can provide our CGT customers with significant shelf-life extension of biologic source material and final cell products, and can also greatly improve post-preservation cell and tissue viability and function. Our biopreservation media are serum-free, protein-free, fully defined, and manufactured under current Good Manufacturing Practices ("cGMP"). We strive to source wherever possible the highest available grade, Multi-compendial raw materials. Our US FDA Type II Master File applicable to our biopreservation products has been cross referenced over 750 times by our customers, and we believe our cell processing products are utilized in several hundred active clinical trials worldwide.
Stability (i.e. shelf-life) and functional recovery are crucial aspects of academic research and clinical practice in the biopreservation of biologic-based source material, intermediate derivatives, and isolated/derived/expanded cellular products and therapies. Limited stability is especially critical in the CGT field, where harvested cells and tissues will lose viability over time if not maintained appropriately at normothermic body temperature (37ºC) or stored in a hypothermic or cryogenic state in an effective preservation medium.
Chilling (hypothermia) is used to reduce metabolism and delay degradation of harvested cells and tissues. However, subjecting biologic material to hypothermic or cryogenic environments and subsequently rewarming them may also induce damaging molecular stress and structural changes. Although cooling successfully reduces metabolism (i.e., lowers demand for energy), various levels of cellular damage and death occur when using suboptimal methods. Biopreservation media can mitigate the damage from exposure to hypothermic or cryogenic temperatures and subsequent rewarming.
Traditional biopreservation media range from simple “balanced salt” (electrolyte) formulations to complex mixtures of electrolytes, energy substrates such as sugars, osmotic buffering agents, and antibiotics. The resulting limited stability from
the use of these traditional biopreservation media formulations is a significant shortcoming that our optimized proprietary products address with great success.
Our scientific research activities over the last 20+ years enabled a detailed understanding of the molecular basis for the hypothermic and cryogenic (low-temperature induced) damage/destruction of cells through apoptosis and necrosis. This research led directly to the development of our HypoThermosol FRS and CryoStor technologies. Our proprietary biopreservation media products are specifically formulated to:
•Minimize cell and tissue swelling
•Reduce free radical levels upon formation
•Maintain appropriate low temperature ionic balances
•Provide regenerative, high-energy substrates to stimulate recovery upon warming
•Avoid the creation of an acidic state (acidosis)
•Inhibit the onset of apoptosis and necrosis
A key feature of our biopreservation media products is their “fully-defined” profile. All of our cGMP products are serum-free, protein-free and are formulated and filled using aseptic processing. We strive to use USP/Multi-compendial grade or the highest quality available synthetic components. All of these features benefit prospective customers by facilitating the qualification process required to incorporate our products into their regulatory filings.
Competing biopreservation media products are often formulated with isotonic media cocktails, animal serum, and potentially a single sugar or human protein. A key differentiator of our proprietary HypoThermosol FRS and CryoStor formulations is the engineered optimization of the key ionic component concentrations for low-temperature environments. This is in contrast to media optimized for normothermic body temperature (around 37°C), as found in culture media or saline-based isotonic formulas. While competing cryopreservation freeze media is often comprised of a single permeating cryoprotectant such as dimethyl sulfoxide (“DMSO”), our CryoStor formulations incorporate multiple permeating and non-permeating cryoprotectant agents, which allows for multiple mechanisms of protection and reduces the dependence on a single cryoprotectant. We believe that our products offer significant advantages over in-house ("home brew") formulations or commercial “generic” biopreservation media. These advantages include time savings, more consistent and higher quality of components, more rigorous quality control release testing, cost effectiveness, and improved preservation efficacy.
The results of independent testing demonstrate that our biopreservation media products significantly extend shelf-life and improve cell and tissue post-thaw viability and function. Our products have demonstrated improved biopreservation outcomes, including greatly extended shelf-life and post-thaw viability and yield across a broad array of cell and tissue types.
Human platelet lysate media, cryogenic vials and automated cell-processing fill machines
Our bioproduction products portfolio includes human platelet lysates for cell expansion, which reduces risk and improves downstream performance over fetal bovine serum, human serum, and other chemically defined media, CellSeal® closed systems that are purpose-built rigid containers used in CGT that can be filled manually or with high throughput systems, CryoCase™ cryo-compatible transparent rigid containers designed for closed-system fill and retrieval, and automated cell processing machines that bring multiple processes traditionally performed by manual techniques under a higher level of control to protect therapies from loss or contamination.
Automated thawing devices
The ThawSTAR® line includes thawing products that control the temperature and timing of the thawing process of biologic material. Our customizable, automated, water-free thawing products use algorithmic programmed heating plates to consistently bring biologic material from a frozen state to a liquid state in a controlled and consistent manner, helping reduce damage during the temperature transition while delivering critical process consistency across cell batches. Use of ThawSTAR products can also reduce risk of contamination versus using a traditional water bath.
Our market opportunity
The CGT market has been rapidly expanding, treating diseases once thought incurable. According to the Alliance for Regenerative Medicine (“ARM”), “2026 State of the Industry Briefing” there were over 1,900 ongoing clinical trials
globally utilizing regenerative medicine at year-end 2025. Additionally, ARM reported an expected $2.0 billion in revenues over the next five years from recently developed and FDA approved CGT therapies, with continued investment in CGT development from the world's largest biopharma companies throughout 2025. ARM also reported there was approximately $11.1 billion invested in the regenerative medicine market in 2025, with an expectation of continued regulatory approvals for CGTs during 2026.
The technologies developed within the CGT market change the ways physicians treat patients. The manufacturing, distribution and the delivery process of these therapies is significantly different from many other types of treatments. We believe we are well positioned to address many of the unique manufacturing challenges in the process of delivering CGTs.
The bioproduction process
Our various products and services currently integrate into several steps in our customers’ bioproduction workflow process for cell and gene therapies. See the diagram below for an illustration of this process and our product roles. We offer products that integrate into the critical steps of preservation and thawing under controlled conditions.
Complementary products portfolio
Expanding Participation in Customers’ Workflow
Our strategy
We are focused on the development, production, and commercialization of differentiated, best-in-class products and services that facilitate the manufacturing and delivery of cell and gene therapies and biologic materials. Our products are designed to increase our customers’ product yield and efficacy. We are committed to supporting our customers with strong customer service and applications expertise.
We leverage our numerous relationships with leading cell and gene therapy companies that use our offering of bioproduction products and services to cross-sell other parts of the portfolio. Over the last several years, we have built a
strong reputation as a trusted supplier of critical tools used in cell and gene therapy and biopharma manufacturing. We believe that our relationships and reputation could enable us to drive further incremental revenue growth through the sale of additional products and services to a captive customer base. Our products are designed to increase our customers’ product yield and functionality while reducing their risk, and we are committed to supporting our customers with strong service in addition to scientific and technical expertise in the applications of our products.
Business Operations
Research and development
Our research and development activities are focused on evaluating new, potentially disruptive technologies which may add value throughout the cell and gene therapy manufacturing and delivery workflow. We routinely assess and analyze the strengths and weaknesses of competitive and adjacent products, and are engaged in business development discussions on an ongoing basis. We strive to continue to anticipate customer needs in providing enabling technologies in the CGT space.
Sales and marketing
We market and sell our products through direct sales and third-party distribution. We have experienced field-based sales employees who market our growing product portfolio on a direct basis. Our technical applications engineers and customer care support teams have extensive experience providing support both prior and subsequent to the sale of products.
Our products are also marketed and distributed by regional distributors under non-exclusive agreements. In 2025, 2024, and 2023, sales to third-party distributors accounted for 34%, 34%, and 38% of our revenue, respectively.
The following table represents the Company’s total revenue by geographic area (based on the location of the customer):
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| Revenue by customers’ geographic locations | 2025 | | 2024 | | 2023 |
| United States | 80 | % | | 75 | % | | 81 | % |
| Europe, Middle East, Africa (EMEA) | 14 | % | | 19 | % | | 12 | % |
| Other | 6 | % | | 6 | % | | 7 | % |
| Total revenue | 100 | % | | 100 | % | | 100 | % |
Manufacturing
Cell processing – We maintain and operate two independent cGMP clean room production suites for manufacturing sterile biopreservation media products in Bothell, Washington. Our quality management system (“QMS”) in Bothell is certified to the ISO 13485:2016 standard. Our QMS takes guidance from applicable sections of 21 CFR Part 820 – Quality System Regulation for Good Manufacturing Practice of medical devices, 21 CFR Parts 210 and 211 – cGMP for Finished Pharmaceuticals, FDA Guidance – Sterile Drug Products, Volume 4, EU Guidelines Annex 1 – Manufacture of Sterile Medicinal Products, ISO 13408 – Aseptic Processing of Healthcare Products, and ISO 14644 – Clean Rooms and Associated Controlled Environments.
We also maintain and operate one cGMP clean room production suite for manufacturing hPL media in Indianapolis, Indiana. Our QMS in Indianapolis is certified to the ISO 9001:2015 standard. Our QMS takes guidance from applicable sections of 21 CFR Part 820 – Quality System Regulation for Good Manufacturing Practice of medical devices, 21 CFR Parts 210 and 211 – cGMP for Finished Pharmaceuticals, Volume 4, EU Guidelines Annex 2 – Manufacture of Biological active substances and Medicinal Products for Human Use and ISO 14644 – Clean Rooms and Associated Controlled Environments.
We seek to manage single-source supplier risk by regularly assessing the quality and capacity of our suppliers, implementing supply and quality agreements where appropriate, and actively managing lead times and inventory levels of sourced components. Pursuant to our customer and quality agreements, we are required to notify customers of any changes to our raw materials. For certain components without a secondary supplier, we estimate that it would take up to six months to find and qualify a second source. Order quantities and lead times for externally sourced components are based on our
forecasts, which are derived from historical demand and anticipated future demand. Lead times for components may vary depending on the size of the order, specific supplier requirements, and current market demand for the materials and parts.
We practice continuous improvement based on routine internal audits through our own monitoring of process outputs, external feedback, and audits performed by our partners and customers. In addition, we maintain a business continuity management system that focuses on key areas such as contingency planning, safety stocks and off-site storage of raw materials and finished goods to ensure continuous supply of our products.
Thaw systems – Our ThawSTAR automated, water-free thawing products are produced by a contract manufacturing organization ("CMO") based in the United States. We believe this CMO has the skills, experience and capacity needed to meet our quality standards and demand expectations for the product line. We estimate that it would take up to six months to find and qualify an alternative CMO. To date, we have not experienced significant difficulties in obtaining our automated thaw products from our CMO.
Product regulatory status
Our products are not subject to any specific FDA or other international marketing regulations for drugs, devices, or biologics. We are not required to sponsor formal prospective, controlled clinical trials in order to establish safety and efficacy. However, to support our current and prospective clinical customers, we manufacture and release our products in compliance with cGMP and other relevant quality standards.
To assist customers with their regulatory applications, we maintain Type II Master Files at the FDA for CryoStor, HypoThermosol FRS, BloodStor 27, Stemulate, nLiven PR, T-Liven PR, CellSeal Closed System containers, and our Cell Thawing Media products, which provide the FDA with information regarding our manufacturing facility and process, our quality system, stability and safety, and any additional testing that has been performed. Customers engaged in clinical and commercial applications may notify the FDA of their intention to use our products in their product development and manufacturing process by requesting a cross-reference to our master files.
Intellectual property
The following table lists our granted and pending patents. We have also obtained certain trademarks and tradenames for our products to distinguish our genuine products from our competitors’ products and we maintain certain details about our processes, products, and strategies as trade secrets. While we believe that the protection of patents and trademarks is important to our business, we also rely on a combination of trade secrets, nondisclosure and confidentiality agreements, scientific expertise, and continuing technological innovation to maintain our competitive position. Despite these precautions, it may be possible for unauthorized third parties to copy certain aspects of our products and/or to obtain and use information that we regard as proprietary (see Part I, “Item 1A. Risk Factors” in this Annual Report for additional details). The laws of some foreign countries in which we sell our products do not protect our proprietary rights to the same extent as do the laws of the United States.
| | | | | | | | | | | | | | | | | |
| Issued Patents | | Patents Applied For | | Registered Trademarks |
| Cell processing and other products | 87 | | | 83 | | | 45 | |
Competition
Our bioproduction products and services compete on the basis of value proposition, performance, quality, cost effectiveness, and application suitability with numerous established technologies. Additional products using new technologies that may be competitive with our products may also be introduced. Many of the companies selling or developing competitive products have greater financial and human resources, R&D, manufacturing, and marketing experience than we do. They may undertake their own development of products that are substantially similar to or compete with our products, and they may succeed in developing products that are more effective or less costly than any that we may develop. These competitors may also prove to be more successful in their production, marketing and commercialization activities. We cannot be certain that the research, development, and commercialization efforts of our competitors will not render any of our existing or potential products obsolete.
Human capital
We view our team members as the key to our success. As of December 31, 2025, we had 155 full-time team members and no part-time team members. Our Company culture encourages strong team collaboration and an emphasis on participation in challenging tasks that expand our team's skill sets. We believe in an open-door policy at all levels of the organization and establish Company-wide quarterly townhall meetings to foster a collaborative, connected environment in which anyone can contribute to our success. Our human capital strategy revolves around retaining top talent and maintaining high engagement across our Company. We consider relations with our team members to be good and welcome feedback from all levels of the Company on how to make improvements in our business processes and Company culture.
Team Member Engagement
We value a high level of engagement from our team members. We endeavor to foster a culture of mutual respect and ensure that team members feel valued. We compete for local talent with companies that are both in our market and within proximity to our primary office locations, as well as for our remote workforce within our industry and across other industries. We offer a hybrid work model that enables our team members to work remotely and on-location as their responsibilities allow, and we support team events to build internal collaboration and engagement. We support our team members’ participation in Company social events to build community engagement and foster a sense of responsibility to the communities in which we work.
Compensation and Benefits
We view our compensation and benefits practices as critical to our recruitment, retention, and engagement efforts. We prioritize competitive health benefits for our team members and employ a pay-for-performance compensation model, paying at or above market rates for our positions. We also offer incentive programs for certain employees focused on incentivizing and retaining talent that include stock grants and bonus incentives and provide a generous paid time off program including additional holidays granted throughout the year.
Training and Development
We have a strong belief in promotion from within our Company and provide training and development opportunities to support our talented professionals in their career growth. We offer internal training, peer-to-peer learning opportunities, and mentorship of our team members.
Health and Safety
We provide a safe and healthy work environment that encourages team members to speak up and identify potential safety hazards. We conduct employee ergonomics assessments specifically in our manufacturing processes to limit and eliminate potential injuries from repetitive movements. We provide training for specific safety requirements and modify processes as needed to assure that our team members’ health and safety is a part of every aspect of our business. We provide appropriate personal protective equipment and training for the use of that equipment for safe use.
Corporate history
We were incorporated in Delaware in 1987 under the name Trans Time Medical Products, Inc. In 2002, the Company, then known as Cryomedical Sciences, Inc. was engaged in manufacturing and marketing cryosurgical products. The entity was merged with our wholly owned subsidiary, BioLife Solutions, Inc., which was engaged as a developer and marketer of biopreservation media products for cells and tissues. Following the merger, we changed our name to BioLife Solutions, Inc.
Principal offices; available information
Our principal executive offices are located at 3303 Monte Villa Parkway, Suite 310, Bothell, Washington 98021 and the telephone number is (425) 402-1400. We maintain a website at www.biolifesolutions.com. The information contained on or accessible through our website is not part of this Annual Report on Form 10-K and is not incorporated in any manner into this Annual Report. Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Exchange Act, are available free of charge on our website as soon as reasonably practicable after we electronically file such reports with, or furnish
those reports to, the Securities and Exchange Commission (the “SEC”). The SEC also maintains an Internet site that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC at http://www.sec.gov.
ITEM 1A. RISK FACTORS
Investing in our common stock involves a high degree of risk. You should carefully consider the risks and uncertainties described below, together with all of the other information contained in this Annual Report, including our financial statements and related notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” before deciding to invest in our common stock. If any of the following risks materialize, our business, financial condition, results of operation and prospects will likely be materially and adversely affected. In that event, the market price of our common stock could decline and you could lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial also may impair our business operations or financial condition.
Risks related to our business and operations
If our products or the products of our competitors do not perform as expected or the reliability of the technology on which our products are based is questioned, we could experience lost revenue, delayed or reduced market acceptance of our products, increased costs, and damage to our reputation.
Our success depends on the market’s confidence that we can provide reliable, high-quality products to our customers. We believe that customers in our target markets are likely to be particularly sensitive to product defects and errors. Our reputation and the public image of our products and technologies may be impaired if our products or similar products of our competitors fail to perform as expected. In the future, if our products experience, or are perceived to experience, a material defect or error, this could result in loss or delay of revenues, delayed or reduced market acceptance, damage to our reputation, diversion of development resources, legal claims, increased insurance costs or increased service and warranty costs, any of which could harm our business, financial condition or results of operations. Such defects or errors could also narrow the scope of the use of our products, which could hinder our success in the market. Even after any underlying concerns or problems are resolved, any lingering concerns in our target market regarding our technology or any manufacturing defects or performance errors in our products could continue to result in lost revenue, delayed or reduced market acceptance, damage to our reputation, increased service and warranty costs and claims against us.
We operate in a highly competitive industry and if we cannot compete effectively, our business, financial condition and operating results could be materially and adversely affected.
The life sciences industry is highly competitive and subject to rapid technological change. We anticipate that we will continue to face increased competition as existing companies may choose to develop new or improved products and as new companies enter the market with new technologies, any of which could compete with our products or even render our products obsolete. While there are technological and marketing barriers to entry, we cannot guarantee that these barriers will be sufficient to defend our market share against current and future competitors.
Many of our competitors are significantly larger than us and have greater financial, technical, research, marketing, sales, distribution and other resources than us and may have longer operating histories. These companies may develop technologies that are superior alternatives to our products or may be more effective at commercializing and marketing their technologies. There may also be other companies which are currently developing competitive products and services, or which may in the future develop technologies and products that are comparable, superior or less costly than our own. We may need to improve our existing technologies or develop new technologies for our products to remain competitive. Our future success depends on our ability to compete effectively against current technologies, as well as to respond effectively to technological advances by developing and marketing products that are competitive in the continually changing technological landscape. Our competitors may succeed in developing or marketing technologies and products that are more effective or commercially attractive than any that are being developed or marketed by us, or may succeed in obtaining regulatory approval, or introducing or commercializing any such products, prior to us. Such developments could have a material adverse effect on our business, financial condition and results of operations. Also, even if we can compete successfully, we may not continue do so in a profitable manner.
Despite our increasingly diversified customer base, we depend on a limited number of customers and products in a limited number of market sectors. If we lose any of these large customers or if there are disruptions in the sales of these products, our net product revenue and operating results could decline significantly.
During the years ended December 31, 2025, 2024, and 2023, we derived approximately 29%, 32%, and 29% of our revenue from three customers, respectively. In the years ended December 31, 2025, 2024, and 2023, we derived approximately 82%, 80%, and 82% of our revenue from CryoStor products, respectively. Our principal customers may
vary from period to period and such customers may not continue to purchase products from us at current levels or at all. Further, the inability of some of our customers to consummate anticipated purchases of our products due to changes in end-user demand, and other unpredictable factors that may affect customer ordering patterns could lead to significant reductions in net product revenue which could harm our business.
We expect our operating results to fluctuate significantly from period to period.
Our revenue, operating margins and other operating results have varied significantly in the past and may continue to fluctuate from period to period in the future due to a variety of factors, many of which are beyond our control. Factors relating to our business that may contribute to these fluctuations include, but are not limited to, changes in the timing and terms of product orders and service contracts by our customers as a result of our customer concentration or otherwise, changes in the demand for the mix of products and services that we offer, the timing and market acceptance of our new product and service introductions, delays or problems in the planned introduction of new products or services, or in the performance of any such products following delivery to customers or the quality of such services, new products, services or technological innovations by our competitors, and potential supply chain issues, which can, among other things, render our products and services less competitive due to the rapid technological changes in the markets in which we provide products and services, impact our ability to reduce our costs in response to decreased demand for our products and services, impact our ability to accurately estimate customer demand, including the accuracy of demand forecasts used by us, create disruptions in our manufacturing process or in the supply of components to us, and lead to write-offs for excess or obsolete inventory, competitive pricing pressures, and increased investment into our infrastructure to support our growth, including capital equipment, research and development, as well as selling and marketing initiatives to support continuous product and services innovation, technological capability enhancements and sales efforts, among other factors described elsewhere in this Annual Report. If our quarterly operating results fail to meet expectations of investors or research analysts, the price of our common stock may decline.
If intangible assets and goodwill become impaired, we may have to take significant charges against earnings.
As of December 31, 2025 the net carrying value of our goodwill and other intangible assets totaled $212.8 million. We periodically review our goodwill and the estimated useful lives of our identifiable intangible assets, taking into consideration any events or circumstances that might result in either a diminished fair value, or for intangible assets, a revised useful life. These events and circumstances include significant changes in the business climate, legal factors, operating performance indicators, advances in technology and competition. Any reduction or impairment of value of intangible assets and goodwill will result in a charge against earnings, which could materially adversely affect our results of operations and shareholders’ equity in future periods.
Our success will depend on our ability to attract and retain key personnel.
Our success in implementing our business strategy depends largely on the skills, experience and performance of key members of our executive management team and others in key management positions. The continuing service of our executive management team and other key management positions, together with our ability to attract and retain such management personnel, is critical to our ability to implement our business strategy. There is substantial competition to attract such key management personnel and the loss of one or more of these individuals could have a material adverse effect on our business and operating results.
In addition, a critical factor to our business is our ability to attract and retain essential engineering, scientific, sales and management personnel. Our future success depends to a significant degree upon the continued services of key scientific and technical personnel. We are continually at risk of losing such personnel or being unable to hire additional engineering, scientific, sales and management personnel. If we fail to attract and retain such personnel, our sales efforts will be hindered and we will not be able to achieve our growth objectives.
Our business may be subject to product liability claims or product recalls, which could be expensive and could result in a diversion of management’s attention.
Our business exposes us to potential product liability risks that are inherent in designing, manufacturing, and marketing our products. In particular, we are a supplier of bioproduction products to the cell and gene therapy industry. Our products are used in basic and applied research, and commercial manufacturing of biologic-based therapies and must meet stringent requirements. Products and services as sophisticated as ours could contain undetected errors or defects, especially when first introduced or when new equipment or versions are released. Customers use our products to maintain the health and
function of biologic material during sourcing, manufacturing, storage, and distribution of cells and tissues, and component failures, manufacturing flaws, design defects or inadequate disclosure of product-related risks with respect to these or other products we manufacture or sell could result in an unsafe condition or injury.
As a result, we face an inherent risk of damage to our reputation if one or more of our products are, or are alleged to be, defective. We may be exposed to risks from product liability and warranty claims in the event that our products actually or allegedly fail to perform as expected or the use of our products results, or is alleged to result, in bodily injury and/or property damage. The outcome of litigation, particularly any class-action lawsuits, is difficult to quantify. Plaintiffs often seek recovery of very large or indeterminate amounts, including punitive damages. The magnitude of the potential losses relating to these lawsuits may remain unknown for substantial periods of time and the cost to defend against any such litigation, whether or not we are found liable, may be significant. Accordingly, we could experience product liability losses in the future and incur significant costs to defend these claims. While we maintain product liability insurance coverage, which we deem to be adequate based on historical experience, we cannot assure you that coverage will be available for such risks in the future or that, if available, it would prove sufficient to cover potential claims or that the present amount of insurance can be maintained in force at an acceptable cost to us.
In addition, if any of our products are, or are alleged to be, defective, we may voluntarily participate, or be required by applicable regulators, to participate in a recall of that product if the defect or the alleged defect relates to safety. We cannot assure you that we will be successful in initiating appropriate market recall or market withdrawal efforts that may be required in the future or that these efforts will have the intended effect of preventing product malfunctions and the accompanying product liability that may result. In the event of a recall, we may experience lost sales and be exposed to individual or class-action litigation claims and reputational risk. Product liability, warranty and recall costs may have a material adverse effect on our business, financial condition and results of operations.
Difficulties in manufacturing could have an adverse effect upon our expenses and our product revenues.
We currently manufacture all of our biopreservation media products and other related components. We currently outsource the manufacturing of certain thaw products, and certain cold chain products. Manufacturing our products is difficult and complex. To support our current and prospective clinical customers, we and our outsource manufacturers comply with, and intend to continue to comply with, cGMP in the manufacture of our products. Our ability to adequately manufacture and supply our products in a timely matter is dependent on the uninterrupted and efficient operation of our facilities and those of third parties manufacturing certain of our products or producing raw materials and supplies upon which we rely in our manufacturing. Manufacturing our products may be impacted by:
•availability or contamination of raw materials and components used in the manufacturing process, particularly those for which we have no other source or supplier;
•the ongoing capacity of our facilities and those of our outside manufacturers;
•our and our outside manufacturers’ ability to comply with existing and new regulatory requirements, including cGMP;
•inclement weather and natural disasters;
•changes in forecasts of future demand for product components;
•potential facility contamination by microorganisms or viruses;
•updating of manufacturing specifications;
•product quality success rates and yields;
•labor strikes; and
•global viruses, pandemics and epidemics.
If efficient manufacturing and supply of our products is interrupted, we may experience delayed shipments or supply constraints. If we are at any time unable to provide an uninterrupted supply of our products to customers, our customers may be unable to supply their end-products incorporating our products to their patients and other customers, which could materially and adversely affect our product revenue and results of operations. In addition, if we are unable to procure a component from one of our outside manufacturers, we may be required to enter into arrangements with one or more alternative manufacturing companies, which may cause delays in producing components or result in significant increase in expenses.
We are dependent on our suppliers and third-party manufacturers, including single-source and sole-source suppliers, and disruptions in our supply chain could adversely affect our ability to manufacture and deliver products.
We are dependent on our suppliers and third-party manufacturers to provide quality products and components. Some of the materials, supplies, and services used in our product manufacturing, as well as some of our products, are sourced from single- or sole-source suppliers. As a result, our ability to negotiate favorable terms with those suppliers may be limited, and if those suppliers experience operational, financial, quality, or regulatory difficulties, or if those suppliers and/or their facilities refuse to supply us or cease operations temporarily or permanently, or if those suppliers take unreasonable business positions, we could be forced to cease product manufacturing until the suppliers resume operations, until alternative suppliers could be identified and qualified, or permanently if the suppliers do not resume operations and no alternative suppliers could be identified and qualified.
We cannot assure you that, in the future, our current or alternative sources for materials, supplies, and services used in our product manufacturing, as well as some of our products, will be able to meet all our demands on a timely basis. Unavailability of necessary components could require us to re-engineer our products to accommodate available substitutions, which could increase costs to us and/or have a material adverse effect on our manufacturing schedules, products’ performance and market acceptance. We might not be able to find a sufficient alternative supplier in a reasonable amount of time, or on commercially reasonable terms, if at all. If we fail to obtain an alternative supplier for the components of our products, our operations could be disrupted.
The global supply chain is subject to disruption due to labor, geopolitical, trade and monetary issues, which may be exacerbated by ongoing instability in Ukraine and the Middle East. Although we have yet to experience any material effects of this impact on our supply chain or operations, we face the potential risk that upstream disruptions may occur. Risks relating to the lingering effects of global supply chain disruptions may even continue after current conflicts have subsided.
While we are not currently subject to FDA or other regulatory approvals, if our products become subject to regulatory requirements, the manufacture and sale of our products may be delayed or prevented, or we may become subject to increased expenses.
While none of our products are subject to FDA regulation, we comply with cGMP requirements and other relevant quality standards to support our current and prospective clinical customers. However, we cannot assure you that our products will not be subject to FDA regulation in the future, and we may develop products in the future that subject us to regulation by the FDA and similar foreign regulatory agencies. The regulations enforced by the FDA and similar foreign regulatory agencies govern a wide variety of product-related activities, including the research, development, testing, manufacture, quality control, approval, clearance, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, marketing, post-approval monitoring and reporting, pricing, and export and import of pharmaceutical products. If we or any of our customers, suppliers or distributors fail to comply with applicable regulatory requirements, we may face, among other things, warning letters; adverse publicity affecting both us and our customers; investigations or notices of non-compliance, fines, injunctions, and civil penalties; import or export restrictions; partial suspensions or total shutdown of production facilities or the imposition of operating restrictions; increased difficulty in obtaining required FDA clearances or approvals or foreign equivalents; seizures or recalls of our products or those of our customers; or the inability to sell our products and services. Any such FDA or other foreign regulatory agency actions could disrupt our business and operations, lead to significant remedial costs and have a material adverse impact on our financial position and results of operations.
We and our customers are subject to various international governmental regulations. Compliance with, or changes in, such regulations may cause us to incur significant expenses, and if we fail to maintain satisfactory compliance with certain regulations, we may be forced to recall products and cease their manufacture and distribution, and we could be subject to civil or criminal penalties.
We and our customers are subject to various significant international, federal, state and local regulations, including but not limited to regulations in the areas of health and safety, employment, labor and immigration, import/export controls, trade restrictions and anti-competition. In addition, as an international organization, we are subject to data privacy and security laws, regulations, and customer-imposed controls in numerous jurisdictions as a result of having access to and processing confidential, personal, or sensitive data in the course of our business. The EU’s General Data Protection Regulation ("GDPR"), which became effective in May 2018, applies to our activities related to products and services that we offer to EU customers and workers. The GDPR established new requirements regarding the handling of personal data and includes significant penalties for non-compliance. Other governmental authorities around the world have passed or are considering similar types of legislative and regulatory proposals concerning data protection. Each of these privacy, security and data
protection laws and regulations could impose significant limitations and increase our cost of providing our products and services where we process end user personal data and could harm our results of operations and expose us to significant fines, penalties, and other damages.
We must also comply with complex foreign and U.S. laws and regulations, such as the U.S. Foreign Corrupt Practices Act, the U.K. Bribery Act, and other local laws prohibiting corrupt payments to governmental officials, anti-competition regulations and sanctions imposed by the U.S. Office of Foreign Assets Control and other similar laws and regulations. Violations of these laws and regulations could result in fines and penalties, criminal sanctions, restrictions on our business conduct and on our ability to offer our products in one or more countries, and could also materially affect our brand, our ability to attract and retain employees, our international operations, our business and our operating results. Although we have implemented policies and procedures designed to ensure compliance with these laws and regulations, there can be no assurance that our employees, contractors, or agents will not violate our policies. These regulations are complex, change frequently and have tended to become more stringent over time. We may be required to incur significant expenses to comply with these regulations or to remedy any violations of these regulations. Any failure by us to comply with applicable government regulations could also result in the cessation of our operations or portions of our operations, product recalls or impositions of fines and restrictions on our ability to carry on or expand our operations.
Healthcare reform measures could adversely affect our business and financial results.
In response to perceived increases in healthcare costs in recent years, the efforts of governmental and third-party payors to contain or reduce the costs of healthcare and, more generally, to reform the U.S. healthcare system may adversely affect the business and financial condition of pharmaceutical and biotechnology companies, including ours. Specifically, in both the United States and some foreign jurisdictions, there have been a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sell our products profitably, including by limiting the prices we are able to charge for our products or the acceptance and availability of our products. Efforts by governments and other third-party payors to contain or reduce the costs of healthcare through various means may limit our commercial opportunities and adversely affect our operating results and result in a decrease in the price of our common stock or limit our ability to raise capital. We anticipate additional uncertainty as debates about healthcare and public health continue.
Insurance coverage is increasingly difficult to obtain or maintain.
While we currently maintain product liability insurance, directors’ and officers’ liability insurance, general liability insurance, and other types of insurance, first- and third-party insurance is increasingly more difficult to obtain and maintain and has become more costly and narrower in scope, and we may be required to assume more risk in the future. We cannot predict the magnitude of potential liabilities and claims that may be made against us which could exceed the limits of these policies. Additionally, our insurance coverage may not protect us against all liability because our policies typically have various exceptions to the claims covered and also require us to assume some costs of the claim even though a portion of the claim may be covered. If we are subject to third-party claims or suffer a loss or damage in excess of our insurance coverage, we may be required to share that risk in excess of our insurance limits. A partially or completely uninsured claim, if successful and of significant magnitude, could have a material adverse effect on our business, financial condition and results of operations. Furthermore, any first- or third-party claims made on our insurance policies may impact our future ability to obtain or maintain product liability insurance coverage at reasonable costs, if at all.
We are and may become the subject of various claims, litigation or investigations which could have a material adverse effect on our business, financial condition, or results of operations or the price of our common stock.
We are and may become subject to various claims, including “whistleblower” complaints, litigation or investigations, including commercial disputes and employee claims, and from time to time may be involved in governmental or regulatory investigations or similar matters. Some of these claims may relate to the activities of businesses that we have acquired, even though these activities may have occurred prior to our acquisition of such businesses. Any claims asserted against us or our management, regardless of merit or eventual outcome, could harm our reputation, distract our management and have an adverse impact on our relationship with our existing or prospective clients, distribution partners and other third parties and could lead to additional related claims. Furthermore, there is no guarantee that we will be successful in defending ourselves in pending or future litigation or similar matters under various laws. Any judgments or settlements in any pending litigation or future claims, litigation or investigation could have a material adverse effect on our business, financial condition, or results of operations or the price of our common stock.
Our business and operations could be negatively affected by securities litigation or stockholder activism, which could impact the trading price and volatility of our common stock and may constrain capital deployment opportunities and adversely impact our ability to expand our business.
Our business and operations could be negatively affected if we become subject to any securities litigation or from continued stockholder activism, which could cause us to incur significant expenses, hinder the execution of our business and growth strategy, constrain our capital deployment opportunities, and impact the price of our common stock. In the past, following periods of volatility in the market price of a company’s securities, securities class action litigation has often been brought against that company. Stockholder activism, which can take many forms or arise in a variety of situations, has been increasing recently. Volatility in the price of our common stock, our cash balance, our financial performance or other reasons may cause us to become the target of securities litigation or continue to be the target of stockholder activism.
Securities litigation and stockholder activism, including potential proxy contests, could result in substantial costs and divert the attention of our management and our board of directors (our “Board”) and resources from our business. Additionally, such securities litigation and stockholder activism could give rise to perceived uncertainties as to our future, adversely affect our relationships with service providers and make it more difficult to attract and retain qualified personnel. Also, we may be required to incur significant legal fees and other expenses related to any securities litigation and activist stockholder matters. Further, the price of our common stock could be subject to significant fluctuation or otherwise be adversely affected by the events, risks and uncertainties of any securities litigation and stockholder activism. In addition, stockholder activism may constrain our capital deployment opportunities and may limit the types of investments that are available to us.
Risks related to our acquisition and divestiture activities
Our acquisitions expose us to risks that could adversely affect our business, and we may not achieve the anticipated benefits of acquisitions of businesses or technologies.
From time to time, potential acquisition opportunities may become available to us, and we may periodically engage in discussions or negotiations relating to potential acquisitions, including acquisitions that may be material in size or scope to our business. As a part of our growth strategy, we have made, and may continue to make, selected acquisitions of other companies and technologies and continue to evaluate expansion through acquisitions of other companies or technologies, which may carry numerous risks and operational, financial, and managerial challenges, including, but not limited to, the following, any of which could adversely affect our business, financial condition, or results of operations:
•difficulties in integrating new operations, technologies, products, and personnel;
•problems maintaining uniform procedures, controls, and policies with respect to our financial accounting systems;
•lack of synergies or the inability to realize expected synergies and cost-savings;
•difficulties in managing geographically dispersed operations, including risks associated with entering foreign markets in which we have no or limited prior experience;
•underperformance of any acquired technology, product, or business relative to our expectations and the price we paid;
•negative near-term impacts on financial results after an acquisition, including acquisition-related earnings charges;
•the potential loss of key strategic partners of acquired companies;
•claims by terminated employees and shareholders of acquired companies or other third parties related to the transaction;
•the assumption or incurrence of additional debt obligations or expenses, or use of substantial portions of our cash;
•diversion of management’s attention and company resources from existing operations of the business;
•inconsistencies in standards, controls, procedures, and policies;
•cash expenses and non-cash accounting charges incurred in connection with acquisitions, including unanticipated costs associated with the amortization of intangible assets;
•the impairment of intangible assets as a result of technological advancements, or worse-than-expected performance of acquired companies;
•assumption of, or exposure to, historical liabilities of the acquired business, including unknown contingent or similar liabilities, including product liability, that are difficult to identify or accurately quantify; and
•risks associated with acquiring intellectual property, including potential disputes regarding acquired companies’ intellectual property.
Additionally, the successful integration of acquired businesses requires significant efforts and expense across all operational areas, including sales and marketing, research and development, manufacturing, finance, legal, and information technologies. Acquisitions and strategic investments and alliances may require us to integrate and collaborate with a different company culture, management team, business model, business infrastructure and sales and distribution methodology, and assimilate and retain geographically dispersed, decentralized operations and personnel. Depending on the size and complexity of an acquisition, our successful integration of the entity depends on a variety of factors, including introducing new or modified products and meeting revenue targets as expected, the retention of key employees and key customers, increased exposure to certain governmental regulations and compliance requirements and increased costs and use of resources. The integration of acquired businesses is also likely to result in our systems and internal controls becoming increasingly complex and more difficult to manage. Any difficulties in the assimilation of acquired businesses into our control system could harm our operating results or cause us to fail to meet our financial reporting obligations. We are unable to guarantee the success of any acquisitions that we complete and such acquisitions may not be, or remain, profitable. Our failure to successfully address the foregoing integration risks may prevent us from achieving the anticipated benefits from any acquisition in a reasonable time frame, or at all.
Even if we are able to successfully integrate acquired businesses, our ability to realize the anticipated business opportunities, growth prospects, cost savings, synergies, and other benefits of acquisitions depends on a number of factors including our ability to:
•leverage our infrastructure to sell and cross-market acquired products;
•drive adoption of acquired products in new markets;
•bring acquired products to new geographic markets;
•comply with new governmental regulations, including obtaining regulatory approvals in relevant markets;
•execute on development timelines for acquired products;
•manage inventories for product lines;
•carry, service, and manage significant debt and repayment obligations; and
•manage the unforeseen risks and uncertainties related to these transactions, including any related to intellectual property rights.
Many of these factors are outside of our control and any one of them could result in increased costs, decreased revenues, and diversion of management’s time and energy. The benefits of these transactions may not be achieved within the anticipated time frame or at all. Any of these factors could negatively impact our earnings per share, decrease or delay the expected accretive effect of the transaction, and negatively impact the price of our common stock.
Our recent divestitures expose us to ongoing risks and uncertainties, one or more of which could have a material adverse effect on our business, financial condition, results of operations, cash flows, and/or stock price.
In recent years, we completed several divestitures, including the SAVSU Transaction, the CBS Divestiture, the SciSafe Divestiture, and the Global Cooling Divestiture. These divestitures expose us to continued financial and operational risks, such as through retained liabilities and indemnification obligations, transition service obligations, potential disputes with buyers regarding purchase price adjustments or indemnification claims, loss of revenue and customers, stranded costs and inefficiencies, potential litigation and employee retention challenges.
For example, pursuant to the Global Cooling Purchase Agreement, we are required to indemnify Global Cooling for preexisting legal contingencies. Prior to the Global Cooling Divestiture, two lawsuits were filed by previous customers seeking payment for losses related to Global Cooling’s commercial freezer products. These lawsuits are described in further detail under Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
In addition, in connection with the SciSafe Divestiture, we remain liable and responsible for the full performance and observance of all of the provisions, covenants, and conditions in one of SciSafe’s operating leases. In the case of a breach
or violation of any provision of the lease by SciSafe Buyer, we are deemed to be in default of the lease provisions. Simultaneously, we received indemnification pursuant to any obligation owed by us under this operating lease. This indicates we undertake the obligation to stand ready to perform over the term of the guarantee in the event of the specified triggering events noted above, or if conditions, such as breach or default, occur. However, the non-contingent aspect of the guarantee enables us to recover any losses from SciSafe Buyer.
These risks, or any future unforeseen risks, could have a material adverse impact on our business, financial condition, results of operations, cash flows or the price of our common stock.
Further, in connection with these divestitures, we incurred significant cash and non-cash expenses, including transaction costs, severance payments, earn-out or adjustment settlements, and stock compensation acceleration. Any future divestitures may result in similar costs and risks.
Risks related to our intellectual property, cyber security, and artificial intelligence
Our proprietary rights may not adequately protect our technologies and products.
Our technology is critical to the implementation of our business plan, and we are dependent on our patent rights and other intellectual property rights to maintain our competitive position. Our policy is to seek to protect our proprietary position and our commercial success will depend on our ability to obtain patents and/or regulatory exclusivity and maintain adequate protection for our technologies and products in the United States and other countries. We will be able to protect our proprietary rights from unauthorized use by third parties only to the extent that our proprietary technologies and products are covered by valid and enforceable patents or are effectively maintained as trade secrets.
We intend to apply for additional patents covering both our technologies and products, as we deem appropriate. We may, however, fail to apply for patents on important technologies or products in a timely fashion, if at all. Our existing patents and any future patents we obtain may not be sufficiently broad to prevent others from practicing our technologies or from developing competing products and technologies. In addition, the patent positions of life science industry companies are highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. As a result, the validity and enforceability of our patents cannot be predicted with certainty. In addition, we cannot guarantee that:
•we were the first to make the inventions covered by each of our issued patents and pending patent applications;
•we were the first to file patent applications for these inventions;
•others will not independently develop similar or alternative technologies or duplicate any of our technologies;
•any of our pending patent applications will result in issued patents;
•any of our patents will be valid or enforceable;
•any patents issued to us will provide us with any competitive advantages, or will not be challenged by third parties; and
•we will develop additional proprietary technologies that are patentable, or the patents of others will not have an adverse effect on our business.
The actual protection afforded by a patent varies on a product-by-product basis, from country to country and depends on many factors, including the type of patent, the scope of its coverage, the availability of regulatory related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patents. Our ability to maintain and solidify our proprietary position for our products will depend on our success in obtaining effective claims and enforcing those claims once granted. Our issued patents and those that may be issued in the future, or those licensed to us, may be challenged, invalidated, unenforceable or circumvented, and the rights granted under any issued patents may not provide us with proprietary protection or competitive advantages against competitors with similar products. We also rely on trade secrets to protect some of our technology, especially where it is believed that patent protection is inappropriate or unobtainable. However, trade secrets are difficult to maintain. While we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, or scientific and other advisors may unintentionally or willfully disclose our proprietary information to competitors. Enforcement of claims that a third party has illegally obtained and is using trade secrets is expensive, time consuming and uncertain. In addition, non-U.S. courts are sometimes less willing than U.S. courts to protect trade secrets. If our competitors independently develop equivalent knowledge, methods, and know-how, we would not be able to assert our trade secrets against them and our business could be harmed.
Expiration of our patents may subject us to increased competition and reduce our opportunity to generate product revenue.
The patents for our products have varying expiration dates and, when these patents expire, we may be subject to increased competition and we may not be able to recover our development costs. In some of the larger economic territories, such as the United States and Europe, patent term extension/restoration may be available. We cannot, however, be certain that an extension will be granted or, if granted, what the applicable time or the scope of patent protection afforded during any extended period will be. If we are unable to obtain patent term extension/restoration or some other exclusivity, we could be subject to increased competition and our opportunity to establish or maintain product revenue could be substantially reduced or eliminated. Furthermore, we may not have sufficient time to recover our development costs prior to the expiration of our U.S. and non-U.S. patents.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting, and defending patents on all our products in every jurisdiction would be prohibitively expensive. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products. These products may compete with our products and may not be covered by any patent claims or other intellectual property rights.
The laws of some non-U.S. countries do not protect intellectual property rights to the same extent as the laws of the United States, and many companies have encountered significant problems in protecting and defending such rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement of our patents. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business.
We may incur substantial costs as a result of litigation or other proceedings relating to patent and other intellectual property rights, and we may be unable to protect our rights to, or use of, our technology.
Our success will depend to a significant degree on our ability to secure and protect intellectual property rights and enforce patent and trademark protections relating to our technology. While we believe that the protection of patents and trademarks is important to our business, we also rely on a combination of copyright, trade secret, nondisclosure and confidentiality agreements, know-how and continuing technological innovation to maintain our competitive position. From time to time, litigation may be advisable to protect our intellectual property position. However, these legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. If we choose to go to court to stop someone else from using the inventions claimed in our patents or our licensed patents, that individual or company has the right to ask the court to rule that these patents are invalid and/or should not be enforced against that third party. These lawsuits are expensive and would consume time and other resources even if we were successful in stopping the infringement of these patents. In addition, there is a risk that the court will decide that these patents are invalid or unenforceable and that we do not have the right to stop the other party from using the inventions. There is also the risk that, even if the validity or enforceability of these patents is upheld, the court will refuse to stop the other party on the grounds that such other party’s activities do not infringe our rights. Any adverse outcome in litigation relating to the validity of our patents, or any failure to pursue litigation or otherwise to protect our patent position, could materially harm our business and financial condition. In addition, confidentiality agreements with our employees, consultants, customers, and key vendors may not prevent the unauthorized disclosure or use of our technology. It is possible that these agreements will be breached or that they will not be enforceable in every instance, and that we will not have adequate remedies for any such breach.
If a third party asserts that we infringed its patents or other proprietary rights, we could face a number of risks that could seriously harm our results of operations, financial condition and competitive position, including:
•patent infringement and other intellectual property claims, which would be costly and time consuming to defend, whether or not the claims have merit, and which could delay a product and divert management’s attention from our business;
•substantial damages for past infringement, which we may have to pay if a court determines that our product or technologies infringe a competitor’s patent or other proprietary rights;
•a court prohibiting us from selling or licensing our technologies unless the third party licenses its patents or other proprietary rights to us on commercially reasonable terms, which it is not required to do; and
•if a license is available from a third party, we may have to pay substantial royalties or lump-sum payments or grant cross licenses to our patents or other proprietary rights to obtain that license.
We cannot predict the extent to which we might be required to seek licenses or alter our products or services so that they no longer infringe the rights of others. We also cannot guarantee that licenses will be available or the terms of any licenses we may be required to obtain will be reasonable. Similarly, changing our products, services or processes to avoid infringing the rights of others may be costly or impractical and could detract from the value of our products and services. If a judgment of infringement were obtained against us, we could be required to pay substantial damages and a court could issue an order preventing us from selling one or more of our products or offering certain of our services. Further, the cost and diversion of management attention brought about by such litigation could be substantial, even if we were to prevail. Any of these events could result in significant expense to us and may materially harm our business and our prospects.
Even if we are granted a patent, in certain circumstances we may be unable to protect our rights to, or use of, our technology.
The biotechnology industry has produced a proliferation of patents, and it is not always clear to industry participants, including us, which patents cover various types of products or methods of use. The coverage of patents is subject to interpretation by the courts, and the interpretation is not always uniform. If we are sued for patent infringement, we would need to demonstrate that our products or methods of use either do not infringe the patent claims of the relevant patent, and/or that the patent claims are invalid, and/or that the patent is unenforceable, and we may not be able to do this. Proving invalidity, in particular, is difficult since it requires a showing of clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents.
U.S. patent laws as well as the laws of some foreign jurisdictions provide for provisional rights in published patent applications beginning on the date of publication, including the right to obtain reasonable royalties, if a patent subsequently issues and certain other conditions are met.
Because some patent applications in the United States may be maintained in secrecy until the patents are issued, because patent applications in the United States and many foreign jurisdictions are typically not published until 18 months after filing, and because publications in the scientific literature often lag behind actual discoveries, we cannot be certain that others have not filed patent applications for technology covered by our issued patents or our pending applications, or that we were the first to invent the technology.
Patent applications filed by third parties that cover technology similar to ours may have priority over our patent applications and could further require us to obtain rights to issued patents covering such technologies. If another party files a U.S. patent application on an invention similar to ours, we may elect to participate in or be drawn into an interference proceeding declared by the U.S. Patent and Trademark Office to determine priority of invention in the United States. The costs of these proceedings could be substantial, and it is possible that such efforts would be unsuccessful, resulting in a loss of our U.S. patent position with respect to such inventions. Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. In addition, any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise the funds necessary to continue our operations. We cannot predict whether third parties will assert these claims against us, or whether those claims will harm our business. If we are forced to defend against these claims, whether they are with or without any merit and whether they are resolved in favor of or against us, we may face costly litigation and diversion of management’s attention and resources. As a result of these disputes, we may have to develop costly non-infringing technology or enter into licensing agreements. These agreements, if necessary, may be unavailable on terms acceptable to us, if at all, which could seriously harm our business or financial condition.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
We employ individuals who previously worked with other companies, including our competitors or potential competitors. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise used or disclosed confidential information, including trade secrets or other proprietary information, of former employers or other third parties. We may also be subject to claims that former employers or other third parties have an ownership interest in our patents. Litigation may be necessary to defend against these claims. We may not be successful in
defending these claims, and even if we are successful, litigation could result in substantial costs and be a distraction to our management and other employees. Any litigation or the threat thereof may adversely affect our ability to hire employees, and we may lose valuable intellectual property rights if we fail in defending any such claims. A loss of key personnel or their work product could diminish or prevent our ability to commercialize our products, which could have an adverse effect on our business, results of operations and financial condition.
Our inability to protect our information systems and networks and the proprietary and confidential information in our possession from continually evolving cybersecurity risks or other technological risks, including as a result of breaches of our associated third parties' information technology systems, could materially adversely impact our business, financial condition and results of operations, in addition to our reputation and relationships with our employees, customers, suppliers and business partners.
In conducting our business, we collect, process, transmit and store sensitive, proprietary and confidential information about our employees, customers, vendors, and other parties, including business and personal information, which may be entitled to protection under a number of regulatory regimes. This information may include, but is not limited to, account access credentials, credit and debit card numbers, bank account numbers, social security numbers, driver’s license numbers, names and addresses and other types of sensitive business or personal information. Some of this information is also processed and stored by our third-party service providers to whom we outsource certain functions and other agents, including our customers, which we refer to collectively as our associated third parties.
Although we take the security of our network systems and information seriously, there can be no assurance that the security measures we and our associated third parties employ will effectively prevent unauthorized persons from obtaining unauthorized access to our systems and information due to the evolving nature and intensity of cyberattacks and threats to data security, in light of new and sophisticated tools and methods used by criminals and cyberterrorists to penetrate and compromise systems, including computer viruses, malware, ransomware, phishing, misrepresentation, social engineering and forgery, which make it increasingly challenging to anticipate, harder to detect, and more difficult to adequately mitigate these risks. Additionally, threats to our systems and our associated third parties’ systems can derive and have derived from human error, fraud or malice on the part of employees or third parties, or may result from accidental technological failure, including as a result of natural disasters, power failures or other events beyond our control. While we have cyber security insurance, we may incur significant costs in the event of a successful cyber incident against us or in responding to and recovering from a cyber incident that are not covered by, or exceed the limits of, such insurance. Additionally, the cost and operational consequences of implementing, maintaining and enhancing further data or system protection measures could increase significantly to overcome increasingly intense, complex and sophisticated global cyber threats.
The frequency, intensity, and sophistication of cyberattacks and data security incidents has significantly increased in recent years and is constant. We are continually subject to cyberattacks and the risk of data security incidents, some of which have been successful. Such incidents include malicious third party attempts to identify and exploit system vulnerabilities and/or penetrate or bypass our security measures in order to gain unauthorized access to our networks and systems or those of our associated third parties. Such access has led and could lead in the future to the compromise of sensitive, business, personal or confidential information or instructions to transfer funds by us or customers to unauthorized recipients. Although we have experienced security breaches in the past, none of these breaches have resulted in a material liability or loss to us. As a result of such breaches, we proactively employ multiple methods at different layers of our systems to defend our systems against intrusion and attack and to protect the data we collect. However, we cannot be certain that these measures will be successful and sufficient to counter current and emerging technology threats that are designed to breach our systems in order to gain access to confidential information. Further, while we select our associated third parties carefully, and we seek to ensure that our customers adequately protect their systems and data, we do not control their actions and are not able to oversee their processes. Any problems experienced by our associated third parties, including those resulting from breakdowns or other disruptions in the services provided by such parties or cyber-attacks and security breaches, could adversely affect our ability to conduct our business and our financial condition.
The protection and security of our network systems and our own information, as well as information relating to our employees, customers, suppliers, business partners and others, is vitally important to us. Any failure of us or our associated third parties to maintain the security of our network systems and the proprietary, confidential, and personal data in our possession, including via the penetration of our network security and the misappropriation of proprietary, confidential and personal information, could result in costly investigations and remediation, business disruption, damage to our reputation, financial obligations to third parties, fines, penalties, regulatory proceedings and private litigation with potentially large costs, and also result in deterioration in our employees’, customers’, suppliers’ and business partners’ confidence in us and
other competitive disadvantages, and thus could have a material adverse effect on our business, financial condition and results of operations.
The development, deployment and use of AI technologies present new risks and challenges that could adversely impact our business.
AI is increasingly being used across the global business landscape, including in the life sciences and healthcare industries. We are beginning to integrate AI and machine learning technologies into our business to enhance our operations, products, technology, and services and expect our use of AI to increase as the technology rapidly evolves and improves. However, AI presents risks and challenges that could adversely impact our business.
The development, deployment, and use of AI, including within the life sciences industry, is still in its early stages, where the use of insufficiently developed AI technologies and premature deployment practices could result in unintended outcomes that harm our business. AI technologies may be developed using inaccurate, incomplete, flawed or biased algorithms, training methodologies or data, which could result in competitive harm, regulatory penalties, legal liability, or brand or reputational harm. Further, a failure to timely and effectively use or deploy AI and integrate it into new product offerings and services could negatively impact our competitiveness, particularly ahead of evolving industry trends and evolving consumer demands, and require investments that increase our costs.
Laws and regulations regarding AI technologies are rapidly evolving as well, including in the areas of intellectual property, cybersecurity, privacy, and data protection. As such, it remains uncertain how AI laws and regulations will impact our business or the associated cost or risks related to compliance therewith or with respect to incorporating compliance mechanisms appropriately and effectively into our operations. Compliance with new or changing laws, regulations, or industry standards relating to AI may impose significant operational and financial burdens and may limit our ability to develop, deploy, or use AI technologies in our business.
Risks related to our common stock
Our stock price and volume may be volatile, and purchasers of our securities could incur substantial losses.
The trading price and volume of our common stock, traded on the Nasdaq Capital Market ("Nasdaq") has been highly volatile and could fluctuate widely in price in response to various factors, many of which are beyond our control. In addition, the securities markets have from time-to-time experienced significant price and volume fluctuations that are unrelated to the operating performance of companies. These market fluctuations may also materially and adversely affect the market price of our common stock. For example, in the year ended December 31, 2025, the highest intra-day sale price of our common stock on Nasdaq was $29.62 per share and the lowest intra-day sale price of our common stock on Nasdaq was $19.10 per share. Our highest trading day volume was 1,625,800 shares traded and the lowest trading day volume was 105,900 shares traded. We may continue to incur substantial increases or decreases in our stock price and volume in the foreseeable future.
Our stock price and trading volume and the market prices and trading volume of many publicly traded companies, including companies in the life sciences industry, have been, and can be expected to be, highly volatile. The future market price and trading volume of our common stock could be significantly impacted by numerous factors, including, but not limited to:
•Future sales of our common stock or other capital raising events by us;
•Sales of our common stock by existing shareholders;
•Changes in our capital structure, including stock splits or reverse stock splits;
•Changes in our product offerings and business structure through acquisitions or divestitures, and public perception of our announced acquisitions and divestitures;
•Announcements of technological innovations for new commercial products by our present or potential competitors;
•Developments concerning proprietary rights;
•Adverse results in our field or with clinical tests of our products in customer applications;
•Adverse litigation;
•Unfavorable legislation or regulatory decisions;
•Public concerns regarding our products;
•Variations in quarterly operating results;
•General trends in the health care and biotechnology industries;
•Global viruses, epidemics, and pandemics; and
•Other factors outside of our control, including significant market fluctuations.
In addition, sales of a substantial number of shares of our common stock or other securities in the public markets (including an issuance by us of additional securities in a public offering or private placement), or the perception that these sales may occur, could cause the market price of our common stock or other securities to decline and could materially impair our ability to raise capital through the sale of additional securities. The sale of a large number of shares of our common stock or other securities also might make it more difficult for us to sell equity or equity-related securities in the future at a time and at the prices that we deem appropriate.
A significant percentage of our outstanding common stock is held by one stockholder, and this stockholder therefore has significant influence on us and our corporate actions.
As of December 31, 2025, based on our review of public filings and our records, one of our existing stockholders, Casdin Capital, LLC owned 5,957,165 shares of our common stock, representing 12.4% of the issued and outstanding shares of common stock. Accordingly, this stockholder has had, and will continue to have, significant influence in determining the outcome of any corporate transaction or other matter submitted to our stockholders for approval, including mergers, consolidations and the sale of all or substantially all our assets, election of directors and other significant corporate actions. In addition, without the consent of this stockholder where a stockholder vote may be necessary, we could be prevented from entering into transactions that could be beneficial to us.
We have not paid dividends on our common stock in the past and do not expect to pay dividends in the foreseeable future. Any return on investment may be limited to the value of our common stock.
We have never paid cash dividends on our common stock and do not anticipate paying cash dividends in the foreseeable future. Any future determination to pay dividends will be at the discretion of our Board, subject to compliance with covenants in current and future agreements governing our indebtedness, and will depend on our results of operations, financial condition, capital requirements, contractual arrangements and other factors that our Board deems relevant. Our current policy is to retain all funds and earnings for use in the operation and expansion of our business.
Certain provisions in our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Certain provisions in our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws may have the effect of delaying or preventing a change of control or changes in our management, including, among other things, provisions that:
•authorize our Board to issue, without further action by the stockholders, shares of preferred stock and to determine the price and other terms, including preferences and voting rights;
•restrict the ability of our stockholders to call a special meeting of stockholders except upon written request of the holders of 35% of the outstanding shares entitled to vote thereat;
•establish advance notice procedural mechanics and disclosure requirements applicable to stockholder nominations of directors and submissions of proposals regarding other business at stockholder meetings; and
•provide that any vacancies on our Board resulting from death, resignation, disqualification, removal or other cause or in increase in the authorized number of directors may be filled only by a majority of directors then in office, even though less than a quorum, or by the sole remaining director.
As a Delaware corporation, we are also subject to certain Delaware anti-takeover provisions. Under Delaware law, a corporation may not engage in a business combination with any holder of 15% or more of its capital stock unless the holder has held the stock for three years or, among other things, the Board has approved the transaction. Our Board could rely on Delaware law to prevent or delay an acquisition of us. These provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our Board, which is responsible for appointing the members of our management.
Our Amended and Restated Bylaws designate the Court of Chancery of the State of Delaware and U.S. federal district courts as the exclusive forums for certain types of actions and proceedings that may be initiated by our stockholders, which limits our stockholders’ ability to choose the judicial forum for disputes with us or our directors, officers, or other employees.
Our Amended and Restated Bylaws provide that, with certain limited exceptions, any:
•derivative action, suit or proceeding brought on our behalf;
•action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers, employees to us or our stockholders;
•civil action to interpret, apply or enforce any provision of the General Corporation Law of the State of Delaware;
•civil action to interpret, apply, enforce or determine the validity of the provisions of our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws; or
•action asserting a claim governed by the internal affairs doctrine;
will be exclusively brought in the Court of Chancery of the State of Delaware (or, if the Court of Chancery of the State of Delaware lacks jurisdiction, the sole and exclusive forum for such action shall be another state or federal court located within the State of Delaware).
Furthermore, unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States of America will be the sole and exclusive forum for the resolution of any complaint asserting a cause of action under the Securities Act.
The enforceability of similar choice of forum provisions in other companies’ certificates of incorporation or similar governing documents has been challenged in legal proceedings, and it is possible that a court could find the choice of forum provisions contained in our bylaws to be inapplicable or unenforceable, including with respect to claims arising under the U.S. federal securities laws.
This exclusive forum provision may limit the ability of a stockholder to commence litigation in a forum that the stockholder prefers, or may require a stockholder to incur additional costs in order to commence litigation in Delaware or U.S. federal district court, each of which may discourage such lawsuits against us or our directors or officers. Alternatively, if a court were to find this exclusive forum provision inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings described above, we may incur additional costs associated with resolving such matters in other jurisdictions, which could negatively affect our business, results of operations, and financial condition.
Risks related to accounting matters
We have identified material weaknesses in our internal control over financial reporting, and if our remediation of such material weaknesses is not effective, or if we are unable to develop and maintain an effective system of internal control over financial reporting or disclosure controls and procedures, we may not be able to accurately and timely report financial results or prevent fraud, and our ability to meet our reporting obligations and the trading price of our common stock could be negatively affected.
As described in Item 9A — Controls and Procedures and elsewhere in this Form 10-K, Management concluded our disclosure controls and procedures were effective as of December 31, 2025. However, Management identified a material weakness in our internal control over financial reporting for the fiscal year ended December 31, 2024. While this material weakness was remediated during the year ended December 31, 2025, effective internal control over financial reporting is necessary to provide reliable financial reports and to assist in the effective prevention of fraud. Any inability to provide reliable financial reports or prevent fraud could harm our business. We regularly review and update our system of internal control over financial reporting, disclosure controls and procedures, and corporate governance policies. In addition, we are required under the Sarbanes-Oxley Act of 2002 to report annually on our internal control over financial reporting. Any system of internal controls, however well designed and operated, is based in part on certain assumptions and can provide only reasonable, not absolute, assurances that the objectives of the system are met. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis. Accordingly, a material weakness increases the risk that the financial information we report contains material errors.
In the course of making our assessment of the effectiveness of internal control over financial reporting as of December 31, 2024, we identified one material weakness. The material weakness identified was in relation to not maintaining effective internal controls to verify that key inputs for our stock-based awards were entered correctly into the equity system early in 2024, which was attributable to an outdated internal policy with unclear guidance regarding appropriate inputs
The aforementioned material weaknesses did not result in any identified material misstatements to our financial statements, and there were only immaterial changes to previously released financial results.
To address our material weaknesses, we had implemented the remediation plans described in Item 9A — Controls and Procedures in this Form 10-K. Any failure to establish and maintain effective internal control over financial reporting and disclosure controls and procedures could adversely impact our ability to report our financial results on a timely and accurate basis. If our financial statements are not accurate, investors may not have a complete understanding of our operations or may lose confidence in our reported financial information. Likewise, if our financial statements are not filed on a timely basis as required by the SEC and The Nasdaq Stock Market LLC, we could face severe consequences from those authorities. In either case, it could result in a material adverse effect on our business or have a negative effect on the trading price of our common stock. Further, if we fail to remedy these deficiencies (or any other future deficiencies) or maintain the adequacy of our internal control over financial reporting and disclosure controls and procedures, we could be subject to regulatory scrutiny, civil or criminal penalties or shareholder litigation. We can give no assurance that the measures we have taken and plan to take in the future will remediate the material weaknesses identified or that any additional material weaknesses or restatements of our financial statements will not arise in the future due to a failure to implement and maintain adequate internal control over financial reporting or disclosure controls and procedures.
Further, in the future, if we cannot conclude that we have effective internal control over financial reporting or disclosure controls and procedures, or if our independent registered public accounting firm is unable to provide an unqualified opinion regarding the effectiveness of our internal control over financial reporting, investors could lose confidence in the reliability of our financial statements, which could lead to a decline in our stock price. Failure to comply with reporting requirements could also subject us to sanctions and/or investigations by the SEC, The Nasdaq Stock Market LLC or other regulatory authorities.
Changes in tax laws and regulations could adversely affect our financial condition and results of operations.
We are subject to regular examination by the U.S. Internal Revenue Service and state, local and foreign tax authorities. We regularly assess the likelihood of favorable or unfavorable outcomes resulting from these examinations to determine the adequacy of our expense for income taxes. Although we believe our tax estimates are reasonable, there can be no assurance that any final determination will not be materially different from the treatment reflected in our historical income tax expenses and accruals, which could materially and adversely affect our financial condition and results of operations.
Our ability to utilize our NOL carryforwards to offset future taxable income may be limited by a lack of sufficient profits in the jurisdictions where these NOLs were generated. We have accumulated significant NOLs in various tax jurisdictions from historical operating losses, but if we do not generate adequate taxable income in those jurisdictions before the NOLs expire, we may be unable to fully utilize them, resulting in higher effective tax rates and increased cash tax payments in profitable periods. Tax laws in certain jurisdictions impose restrictions on NOL usage, such as annual utilization caps or requirements for income in the same entity or jurisdiction, which could further constrain our ability to offset taxes.
Our ability to use NOL and tax credit carryforwards and certain built-in losses to reduce future tax payments is limited by provisions of the Internal Revenue Code, and it is possible that certain transactions or a combination of certain transactions may result in material additional limitations on our ability to use our NOL and tax credit carryforwards.
Section 382 and 383 of the Internal Revenue Code contain rules that limit the ability of a company that undergoes an ownership change, which is generally any change in ownership of more than 50% of its stock over a three-year period, to utilize its NOL and tax credit carryforwards and certain built-in losses recognized in years after the ownership change. These rules generally operate by focusing on ownership changes involving stockholders owning directly or indirectly 5% or more of the stock of a company and any change in ownership arising from a new issuance of stock by us. Generally, if an ownership change occurs, the yearly taxable income limitation on the use of NOL and tax credit carryforwards and certain built-in losses is equal to the product of the applicable long-term, tax-exempt rate and the value of our stock immediately before the ownership change. We may be unable to offset our taxable income with losses, or our tax liability with credits, before such losses and credits expire and therefore would incur larger federal income tax liability.
Risks related to disruptive events
Natural disasters, geopolitical unrest, war, terrorism, public health issues or other catastrophic events could disrupt the supply, delivery or demand of products, which could negatively affect our operations and performance.
Earthquakes, floods and other natural disasters, fire, power shortages, geopolitical unrest or other political conditions (including government shutdowns), wars and other military conflicts (such as the ongoing war in Ukraine, conflict in the Middle East and recent U.S. involvement in Venezuela), terrorist attacks and other hostile acts and other events beyond our control and the control of the third parties on which we depend could negatively affect our operations and performance. Any of these events, whether in the United States or abroad, may have a strong negative impact on the global economy, our employees, facilities, partners, suppliers, distributors or customers, and could decrease demand for our products, create delays and inefficiencies in our supply chain and make it difficult or impossible for us to deliver products to our customers. If any of these events result in the destruction or disruption of our data centers or our critical business or information technology systems it could severely affect our ability to conduct normal business operations and, as a result, our operating results would be adversely affected.
Tariffs and other trade policies could have a substantial impact on our business.
Our business is dependent upon the availability of supplies for our products. U.S. relations with the rest of the world remain uncertain with respect to taxes, trade policies, and tariffs, especially under an increasingly volatile political landscape within the United States and abroad. Throughout 2025 and during the first quarter of 2026, there have been significant tariffs imposed on imported goods within the United States and there are currently indications that future tariffs are likely to be imposed. While many of these tariffs have been suspended or reduced temporarily, the imposition of such tariffs may strain international trade relations and has led to foreign governments implementing retaliatory tariffs on goods imported from the United States.
Political tensions resulting from changes in U.S. trade policies could reduce trade volume, investment, technological exchange and other economic activities between major international economies, resulting in a material adverse effect on global economic conditions and the stability of global financial markets and could significantly reduce global trade.
Increases in tariffs, additional taxes or other trade restrictions and retaliatory measures may increasingly impact customer demand and customer investment in manufacturing equipment, increase our manufacturing costs, decrease margins, reduce the competitiveness of our products, or inhibit our ability to sell products or purchase necessary equipment and supplies, which could have a material adverse effect on our business, results of operations, or financial condition.
Unfavorable currency exchange rate fluctuations may impact our operating margins, or may cause us to raise prices for our products and services, which could result in reduced sales.
As we increase sales in international markets, any such international instability and reduction in global trade could negatively impact our expansion plans and international sales. Such risks may also affect our customers’ budgets and their policies which may adversely affect our sales revenue.
During the year ended December 31, 2025, approximately 20% of our revenue was derived from customers located outside the United States, primarily in Europe, Middle East, and Africa. Currency exchange rate fluctuations could have an adverse effect on our sales, cost of sales, and results of operations. Most sales to foreign customers are denominated in the currency of the country in which these products are sold or services are provided, and the currency received in payment for such sales could be less valuable as compared to the U.S. dollar at the time of receipt as a result of exchange rate fluctuations. Unfavorable currency fluctuations could require us to increase prices for our products and services to customers, which could result in lower net sales. Alternatively, if we do not adjust prices for our products and services in response to unfavorable currency fluctuations, our results of operations, including our margins, could be materially and adversely affected. We currently hold an immaterial amount of cash and cash equivalents denominated in foreign currencies. However, should we increase the amount of cash and cash equivalents denominated in foreign currencies held by us, our financial results and capital ratios may be materially impacted by movements in exchange rates. We could experience losses with respect to forward exchange contracts into which we may enter, and from time to time, we may enter into forward exchange contracts and cross-currency swap agreements to reduce currency exposure, but we cannot be certain that our efforts will be adequate to protect us against significant currency fluctuations.
Public health crises have adversely affected, and could in the future adversely affect, our business, financial condition. results of operations and cash flows.
We are subject to risks associated with public heath crises, including those related to pandemics and epidemics. The occurrence of such public health risks could materially affect our business, financial condition, results of operations and cash flows, including due to negative impacts to the global economy, disruptions to global supply chains and workforce participation, and volatility and disruption of financial markets. For example, the COVID-19 pandemic created significant volatility, uncertainty, and economic disruption, which had an adverse effect on our business operations, results of operations, cash flows and financial condition.
In particular, the financial or operational impacts as a result of public health crises have included, and may in the future include:
•The temporary closure of our manufacturing facilities and/or those of our outside manufacturers;
•Unavailability of supplies and other components for our products;
•Costs associated with protecting the health of our employees and adhering to any guidance or orders of various governmental authorities, such masking, testing, and social distancing requirements;
•Risks associated with remote work, including increased cybersecurity risk;
•Widespread staffing shortages;
•Outbreaks of disease in our facilities or those of our third-party service providers, which could require us or them to temporarily shut down business operations or cause a disruption to, or shortage in, our or their workforce;
•Significant volatility or reductions in demand for our products;
•Delays in shipments of our products, which could harm our customer relations and adversely impact our competitive position and sales;
•Restrictions on the ability of our personnel to access customers;
•Challenges to our capacity to manufacture, sell and support the use of our products; and
•Volatility in credit or financial markets.
The extent to which public health crises, including health pandemics and epidemics and other outbreaks, impact our business operations, financial performance and results of operations remains uncertain and will depend on many factors outside our control, including the timing, extent, trajectory and duration of the public health crisis, the emergence of new variants, the development, availability, distribution and effectiveness of vaccines and treatments, and the imposition of protective public safety measures. Additional future impacts on us may include material adverse effects on our manufacturing, supply chain and distribution channels, our ability to execute our strategic plans, and our profitability. The potential effects of public health crises may also impact and potentially heighten many of our other risk factors discussed in this “Risk Factors” section.
Global climate change and related legal and regulatory developments could negatively affect our business, financial condition and results of operations.
Climate change presents risks to us and to our customers, with the risks expected to increase over time. Our products and services are subject to and affected by environmental regulation by federal, state, and local authorities in the United States and regulatory authorities with jurisdiction over our international operations. Future regulations or voluntary actions on our part in response to climate change could result in costly changes to our facilities to reduce carbon emissions. They could also increase energy costs as a result of switching to less carbon-intensive, but more expensive, sources of energy to operate our facilities and to transport and ship products and samples. There can be no assurance that climate change or environmental regulation and response will not have a negative competitive impact on our ability to provide our products or that economic returns will match the investments that we are making in the development of new products and services. We will likely face increasing complexity related to product design, the use of regulated materials, energy consumption and efficiency, and the reuse, recycling, or disposal of products and their components at end-of-use or useful life. There continues to be a lack of consistent climate legislation among local, state, federal and international governmental authorities, which creates economic and regulatory uncertainty regarding future incentives for energy-efficiency and costs of compliance, which may impact the demand for our products and services, our costs associated with providing our products and services, and our results of operations and financial condition. In addition, the potential physical impacts of climate change on our operations are highly uncertain, which is enhanced by the particularities of the geographic
circumstances in areas in which we operate. These impacts may include changes in global weather patterns, which could include local changes in rainfall and storm patterns and intensities, water shortages, changing sea levels, and changing temperature averages or extremes. These impacts may also adversely affect our properties, our business, financial condition and results of operations.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 1C. CYBERSECURITY
We have a thorough process for identifying, assessing, and managing cybersecurity risks. We design and assess our cybersecurity risk management program leveraging the National Institute of Standards and Technology Cybersecurity Framework ("NIST") within our broader risk management framework. The cybersecurity team gathers insights from external experts and internal threat intelligence teams to support our cybersecurity risk management program. A dedicated team oversees cybersecurity risk management, led by professionals with deep expertise, including our Vice President of Technology, who has over 20 years of experience in technology and cybersecurity focused roles. Our executive leadership, supported by our cybersecurity team, oversees our enterprise risk management and regularly considers cybersecurity and other material risks. We promote a culture of security, sending out monthly cybersecurity awareness trainings to our employees.
Within our cybersecurity risk management system, our incident management team tracks and logs privacy and security incidents across the Company and third-party service providers. Significant incidents undergo review by a cross-functional group, with immediate escalation for potentially material incidents. We have an incident response retainer with a leading professional services network, ensuring immediate support in the event of a cybersecurity incident. We consult with outside counsel as needed, with final decisions made by the CFO and CEO.
The Board considers cybersecurity risk as part of its risk oversight function and has delegated to the Audit Committee oversight of steps the Company has taken to monitor or mitigate significant cybersecurity risks. The Audit Committee additionally ensures compliance with disclosure requirements and cooperation with law enforcement. Senior management regularly updates the Audit Committee on cyber risks and any material incidents.
While our business strategy and financial condition have not been materially affected by cybersecurity risks, we cannot guarantee future immunity. For more details, refer to Part I, Item 1A, “Risk Factors” in this Annual Report.
ITEM 2. PROPERTIES
Our material office and manufacturing leases are detailed below:
| | | | | | | | | | | | | | | | | | | | |
| Location | | Square Feet | | Principal Use | | Lease Expiration |
| Bothell, WA | | 75,241 | | Corporate headquarters, manufacturing, research and development, marketing, and administrative offices | | July 2031 |
| Woodinville, WA | | 13,578 | | Warehouse | | February 2030 |
| Manchester, CT | | 524 | | Research and development | | August 2026 |
| Indianapolis, IN | | 10,890 | | Manufacturing, research and development, and administrative offices | | June 2027 |
| Edmonton, AB | | 200 | | Research and development | | March 2026 |
| Ottawa, ON | | 200 | | Research and development | | December 2026 |
We consider the facilities to be in a condition suitable for their current uses. Due to the increasing requirements of customers or regulatory agencies, we may need to acquire additional space or upgrade and enhance existing space. We believe that adequate facilities will be available upon the conclusion of our leases.
ITEM 3. LEGAL PROCEEDINGS
From time to time, we may be subject to legal proceedings and claims in the ordinary course of business. We are not currently aware of any such proceedings or claims that we believe will have, individually or in the aggregate, a material adverse effect on our business, financial condition or results of operations.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market information for common stock
Our common stock is traded on the Nasdaq Stock Market under the trading symbol “BLFS.”
Stockholders and dividends
As of February 19, 2026, there were approximately 214 holders of record of our common stock. We have never paid cash dividends on our common stock and do not anticipate that any cash dividends will be paid in the foreseeable future. We anticipate that we will retain all earnings, if any, to support our operations. Any future determination as to the payment of dividends will be at the sole discretion of our Board and will depend on our financial condition, results of operations, capital requirements and other factors our Board deems relevant.
Performance graph
The following information is not deemed to be “soliciting material” or to be “filed” with the SEC or subject to Regulation 14A under the Exchange Act, or to the liabilities of Section 18 of the Exchange Act, and will not be deemed to be incorporated by reference into any filing under the Securities Act or the Exchange Act, except to the extent we specifically incorporate it by reference into such a filing.
The following graph shows the cumulative total stockholder return on our common stock with the cumulative total return of the S&P Small Cap 600 Index and our peer group, assuming an initial investment of $100 on December 31, 2020 and the reinvestment of all dividends.
Issuer repurchases of equity securities
Not applicable.
ITEM 6. RESERVED
Reserved.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Company Overview
We are a life sciences company that develops, manufactures, and markets bioproduction products and services which are designed to improve quality and de-risk biologic manufacturing, distribution, and transportation in the cell and gene therapy industry. Our products are used in basic and applied research and commercial manufacturing of biologic-based therapies. Customers use our products to maintain the health and function of biologic material during sourcing, manufacturing, and distribution.
Our current portfolio of bioproduction products and services is comprised of one revenue line that contains three main offerings:
•Cell processing and other products
◦Biopreservation media
◦Human platelet lysate media (“hPL”), cryogenic cryogenic and ultralow temperature containers, and automated cell-processing fill machines
◦Automated thawing devices
On October 6, 2025, the Company entered into the SAVSU Purchase Agreement by and between the Company and the SAVSU Buyer for the sale by the Company of all SAVSU Interests to the SAVSU Buyer. Upon the execution of the SAVSU Purchase Agreement, the SAVSU business is presented in the accompanying Consolidated Financial Statements as
a discontinued operation for all periods presented. See Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the divestiture.
On April 4, 2025, pursuant to the PanTHERA Purchase Agreement by and among the Company, the PanTHERA Sellers, the PanTHERA Buyer Sub, and Dr. Jason Acker, solely in his capacity as Sellers’ Representative, the Company acquired the remaining 90% of the issued and outstanding shares of common stock of PanTHERA not owned by the Company in the PanTHERA Transaction. See Note 2: Acquisition within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the transaction.
On November 14, 2024, the Company entered into the CBS Purchase Agreement with CBS Buyer and CBS for the sale by the Company of all of the issued and outstanding CBS Shares to CBS Buyer. Upon the execution of the CBS Purchase Agreement, the CBS business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented. See Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the divestiture.
On November 12, 2024, the Company entered into the SciSafe Purchase Agreement with the Sci Safe Buyer for the sale by Seller of all of the issued and outstanding SciSafe Shares to SciSafe Buyer. Upon the execution of the SciSafe Purchase Agreement, the SciSafe business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented. See Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the divestiture.
On April 17, 2024, the Company sold all of the issued and outstanding shares of common stock of Global Cooling to GCI Holdings pursuant to the Global Cooling Purchase Agreement. Upon the execution of the Global Cooling Purchase Agreement, the Global Cooling business is presented in the accompanying Consolidated Financial Statements as a discontinued operation for all periods presented. See Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for further details regarding the divestiture.
We currently operate as one bioproduction products and services business which supports several steps in the biologic material manufacturing and delivery process. Our portfolio of tools and services focuses on biopreservation media and cell processing products. We have in-house expertise in cryobiology and the broader CGT workflow, and continue to evaluate opportunities to maximize the value of our product platforms for our extensive customer base through organic growth innovations, partnerships, and acquisitions.
Segment reporting
Management views the Company's operations and makes decisions regarding how to allocate resources as one reportable segment and one reporting unit. For additional information on the Company's segment considerations, Note 15: Segment, customer, and geographic information within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
Critical accounting policies and estimates
We have identified the policies and estimates below as being critical to our business operations and the understanding of our results of operations. These policies require management’s most difficult, subjective, or complex judgments, often as a result of the need to make estimates about the effect of matters that are inherently uncertain. The impact of any associated risks related to these policies on our business operations are discussed throughout “Management’s Discussion and Analysis of Financial Condition,” including in the “Results of Operations” section, where such policies affect our reported and expected financial results. Although we believe that our estimates, assumptions, and judgments are reasonable, they are based upon information presently available. Actual results may differ significantly from these estimates under different assumptions, judgments, or conditions.
Revenue recognition
To determine revenue recognition for contractual arrangements that we determine are within the scope of Financial Accounting Standards Board (“FASB”) Topic 606, Revenue from Contracts with Customers, we perform the following five steps: (i) identify each contract with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to our performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy the relevant performance obligation. We only apply the five-step model to contracts when
it is probable that we will collect the consideration we are entitled to in exchange for the goods or services we transfer to the customer. Contracts with customers may contain multiple performance obligations. For such arrangements, the transaction price is allocated to each performance obligation based on the estimated relative standalone selling prices of the promised products or services underlying each performance obligation. The Company determines standalone selling prices based on the price at which the performance obligation is sold separately. If the standalone selling price is not observable through past transactions, the Company estimates the standalone selling price, taking into account available information such as market conditions and internally approved pricing guidelines related to the performance obligations. Payment terms and conditions vary, although terms generally include a requirement of payment within 30 to 60 days.
The Company primarily recognizes product revenues. Product revenues are generated from the sale of biopreservation media and cell processing tools. We recognize product revenue, including shipping and handling charges billed to customers, at a point in time when we transfer control of our products to our customers, which is upon shipment for substantially all transactions. Shipping and handling costs are classified as part of cost of product revenue in the Consolidated Statements of Operations.
Any remaining revenues earned, which primarily consisted of service revenues generated from various customer service agreements for the provision of warranty and other engineering services and equipment rental arrangement revenues, were not significant in any of the periods presented.
Intangible assets and goodwill
Intangible assets
Intangible assets with a definite life are amortized over their estimated useful lives using the straight-line method and the amortization expense is recorded within intangible asset amortization in the Consolidated Statements of Operations. If the estimate of a definite-lived intangible asset’s remaining useful life is changed, the remaining carrying amount of the intangible asset is amortized prospectively over the revised remaining useful life. Definite-lived intangible assets and their related estimated useful lives are reviewed at least annually to determine if any adverse conditions exist that would indicate the carrying value of these assets may not be recoverable.
Indefinite-lived intangibles are carried at the initially recorded fair value less any recognized impairment. Indefinite-lived intangibles are tested annually for impairment. Impairment assessments are conducted more frequently if certain conditions exist, including a change in the competitive landscape, any internal decisions to pursue new or different technology strategies, a loss of a significant customer, or a significant change in the marketplace, including changes in the prices paid for the Company’s products or changes in the size of the market for the Company’s products. If impairment indicators are present, the Company determines whether the underlying intangible asset is recoverable through estimated future undiscounted cash flows. If the asset is not found to be recoverable, it is written down to the estimated fair value of the asset based on the sum of the future discounted cash flows expected to result from the use and disposition of the asset.
Goodwill
We test goodwill for impairment on an annual basis, and between annual tests if events and circumstances indicate it is more likely than not that the fair value of our goodwill is less than its carrying value. Events that would indicate impairment and trigger an interim impairment assessment include, but are not limited to, current economic and market conditions, including a decline in the Company’s market capitalization, a significant adverse change in legal factors, business climate or operational performance of the business, and an adverse action or assessment by a regulator. Goodwill is tested for impairment in the fourth quarter of each year, or more frequently as warranted by events or changes in circumstances mentioned above. Accounting guidance also permits an optional qualitative assessment for goodwill to determine whether it is more likely than not that the carrying value of a reporting unit exceeds its fair value. If, after this qualitative assessment, we determine that it is not more likely than not that the fair value of a reporting unit is less than its carrying amount, then no further quantitative testing would be necessary. A quantitative assessment is performed if the qualitative assessment results in a more likely than not determination or if a qualitative assessment is not performed. The quantitative assessment considers whether the carrying amount of a reporting unit exceeds its fair value, in which case an impairment charge is recorded to the extent the reporting unit’s carrying value exceeds its fair value. The Company operates as one reporting unit as of the goodwill impairment measurement date in the fourth quarter of 2025.
Contingent consideration
We estimate the acquisition date fair value of the acquisition-related contingent consideration using various valuation approaches, including option pricing models and Monte Carlo simulations, as well as significant unobservable inputs, reflecting the Company’s assessment of the assumptions market participants would use to value these liabilities. The fair value of the contingent consideration is remeasured each reporting period, with any change in the value recorded in our Consolidated Statements of Operations as change in fair value of contingent consideration.
During the year ended December 31, 2023, all contingent consideration liabilities were written off upon the conclusion that we would not achieve certain revenue targets for earnouts.
Stock-based compensation
We measure and record compensation expense using the applicable accounting guidance for share-based payments related to stock options, time-based restricted stock, market-based restricted stock awards and performance-based awards granted to our directors and employees. The fair value of market-based restricted stock awards is estimated at the date of grant using the Monte Carlo Simulation model. The Monte Carlo Simulation valuation model incorporates assumptions as to stock price volatility, the expected life of options or awards, a risk-free interest rate and dividend yield. In valuing our market-based stock awards, significant judgment is required in determining the expected volatility of our common stock. Expected volatility for our market-based restricted stock awards is based on the historical volatility of our own stock and the stock of companies within our defined peer group. Further, our expected volatility may change in the future, which could substantially change the grant-date fair value of future awards and, ultimately, the expense we record. The fair value of restricted stock, including performance awards, without a market condition is estimated using the current market price of our common stock on the date of grant.
We expense stock-based compensation for stock options and restricted stock awards over the requisite service period. For awards with only a service condition, we expense stock-based compensation using the straight-line method over the requisite service period for the entire award. For awards with a market condition, we expense the grant date fair value over the vesting period regardless of the value that the award recipients ultimately receive. For awards with performance conditions, we begin expensing the grant date fair value over the requisite vesting period only when the performance condition is deemed probable.
We have, from time to time, modified the terms of restricted stock awards awarded to employees. We account for the incremental increase in the fair value over the original award on the date of the modification as an expense for vested awards or over the remaining service (vesting) period for unvested awards. The incremental compensation cost is the excess of the fair value of the modified award on the date of modification over the fair value of the original award immediately before the modification.
Provision for income taxes
The assessment regarding whether a valuation allowance is required considers both positive and negative evidence when determining whether it is more likely than not that deferred tax assets are recoverable. In making this assessment, significant weight is given to evidence that can be objectively verified. In its evaluation, the Company considered its cumulative loss and its forecasted losses in the near-term as significant negative evidence. Based upon a review of the four sources of income identified within Accounting Standard Codification ("ASC") 740, Accounting for Income Taxes, the Company determined that the Company’s recorded deferred tax liabilities as of December 31, 2025 would be a sufficient source of taxable income to realize all of its deferred tax assets except for a portion of its NOL carryforwards. As a result, a full valuation allowance on its deferred tax assets was recorded as of December 31, 2025. The Company will continue to assess the realizability of its assets going forward and will adjust the valuation allowance as needed.
The Company determines its uncertain tax positions based on a determination of whether and how much of a tax benefit taken by the Company in its tax filings or positions is more likely than not to be sustained upon examination by the relevant income tax authorities. The Company is generally subject to examination by U.S. federal and local income tax authorities for all tax years in which loss carryforward is available.
The Company applies judgment in the determination of the financial statement recognition and measurement of tax positions taken or expected to be taken in a tax return. As of December 31, 2025, the Company has an unrecognized tax
benefit of $1.2 million related to tax attributes being carried forward. The Company is generally subject to examination by U.S. federal and local income tax authorities for all tax years in which loss carryforward is available.
As of December 31, 2025, the Company had U.S. federal NOL carryforwards of approximately $168.4 million, which is available to reduce future taxable income. Approximately $38.6 million of NOLs will expire from 2026 through 2037, and approximately $129.8 million of NOLs will be carried forward indefinitely. The NOL carryforwards are subject to an annual limitation in the event of certain cumulative changes in the ownership interest. This limits the amount of tax attributes that can be utilized annually to offset future taxable income or tax liabilities. Subsequent ownership changes may further affect the limitation in future years.
Recent accounting standards update
See Note 1: Organization and significant accounting policies – Recent accounting pronouncements, within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report for more information.
Results of operations
The following discussion of the financial condition and results of operations should be read in conjunction with the accompanying Consolidated Financial Statements and the related footnotes thereto.
Revenue
Total revenue for the years ended December 31, 2025, 2024, and 2023 is presented below:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| Revenue | $ | 96,214 | | | $ | 74,647 | | | $ | 68,016 | | | $ | 21,567 | | | 29 | % | | $ | 6,631 | | | 10 | % |
Revenue
Total revenue was $96.2 million for the year ended December 31, 2025, representing an increase of $21.6 million, or 29%, compared with the year ended December 31, 2024. The increase in revenue was primarily driven by a $19.3 million, or 30%, increase in biopreservation media products from an increase in demand from customers with commercially approved therapies when compared to the prior year.
Total revenue was $74.6 million for the year ended December 31, 2024, representing an increase of $6.6 million, or 10%, compared with the year ended December 31, 2023. The increase in revenues was primarily driven by the $4.8 million, or 8%, increase in biopreservation media products due to the increase in customer demand when compared to the prior year. From the third and fourth quarters of 2023 through the first and second quarters of 2024, we experienced a decrease in our revenue from our customers destocking inventory levels in addition to decreases in broader biotech funding, which strongly recovered during the third and fourth quarters of the year ended December 31, 2024.
Cost of revenue and Gross margin
Total costs and operating expenses for years ended December 31, 2025, 2024, and 2023 were comprised of the following:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| Revenue | $ | 96,214 | | | $ | 74,647 | | | $ | 68,016 | | | $ | 21,567 | | | 29 | % | | $ | 6,631 | | | 10 | % |
| Cost of revenue | 34,096 | | | 24,551 | | | 26,698 | | | 9,545 | | | 39 | % | | (2,147) | | | (8 | %) |
| Gross profit | $ | 62,118 | | | $ | 50,096 | | | $ | 41,318 | | | $ | 12,022 | | | 24 | % | | $ | 8,778 | | | 21 | % |
| | | | | | | | | | | | | |
| Gross margin | 65 | % | | 67 | % | | 61 | % | | | | (2 | %) | | | | 6 | % |
In the year ended December 31, 2025, cost of revenue increased $9.5 million, or 39%, from the year ended December 31, 2024. This increase is driven by our 29% increase in overall sales volume in addition to lower yields on biopreservation bags. Additionally, scrap increased as a percentage of revenue in 2025 due to an inventory reserve in Q3 2025 and an increased rate of disposal of expired raw material and finished goods inventory.
Gross margin was 65% and 67% for the years ended December 31, 2025 and 2024, respectively. The decrease in gross margin can be attributed to an increase in costs of materials and overhead due to a less favorable product mix.
In the year ended December 31, 2024, cost of revenue decreased $2.1 million, or 8%, from the year ended December 31, 2023. This decrease can be attributed to a more favorable product mix and increased operational efficiencies.
Gross margin was 67% and 61% for the years ended December 31, 2024 and 2023, respectively. The increase in gross margin can be attributed to a more favorable product mix in our biopreservation media product line and a decrease in supply expenses.
Operating expenses
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| General and administrative | $ | 45,520 | | | $ | 40,627 | | | $ | 42,792 | | | $ | 4,893 | | | 12 | % | | $ | (2,165) | | | (5 | %) |
| Sales and marketing | 9,850 | | | 8,932 | | | 11,377 | | | 918 | | | 10 | % | | (2,445) | | | (21 | %) |
| Research and development | 7,566 | | | 4,751 | | | 5,515 | | | 2,815 | | | 59 | % | | (764) | | | (14 | %) |
| IPR&D expense | 15,521 | | | — | | | — | | | 15,521 | | | 100 | % | | — | | | — | % |
| Intangible asset amortization | 263 | | | 263 | | | 1,032 | | | — | | | — | % | | (769) | | | (75 | %) |
| Change in fair value of contingent consideration | — | | | — | | | (2,193) | | | — | | | — | % | | 2,193 | | | 100 | % |
| Total operating expenses | $ | 78,720 | | | $ | 54,573 | | | $ | 58,523 | | | $ | 24,147 | | | 44 | % | | $ | (3,950) | | | (7 | %) |
General and administrative
During the years ended December 31, 2025, 2024, and 2023, general and administrative (“G&A”) expense consisted primarily of personnel-related expenses, stock-based compensation, professional fees, such as accounting and consulting fees, and corporate insurance.
In the year ended December 31, 2025, G&A expenses increased by $4.9 million, or 12%, compared with the year ended December 31, 2024. The increase is primarily driven by increases in personnel expenses, including $5.1 million in stock compensation expenses, $0.8 million in severance expenses related to the departure of former executives, and $0.4 million in salaries. We also experienced increases of $0.5 million in acquisition costs and $0.4 million in lease expenses. The increases in G&A expenses for the year ended December 31, 2025 were primarily offset by a $2.7 million decrease in estimated sales tax expense as compared to prior years.
G&A expenses decreased $2.2 million, or 5%, during the year ended December 31, 2024 compared with the year ended December 31, 2023. The decrease was primarily driven by a decrease of $1.4 million in severance expenses related to the departure of the former CEO in the prior year in addition to a decrease of $3.8 million in consulting expenses. The decreases in G&A expenses for the year ended December 31, 2024 were offset by increases in personnel expenses, including $1.6 million increase in bonus expenses and a $1.5 million increase in salaries compared with the year ended December 31, 2023.
Sales and marketing
During the years ended December 31, 2025, 2024, and 2023, sales and marketing expense (“S&M”) consisted primarily of personnel-related costs, consulting, trade shows, advertising, and travel expenses.
S&M expense increased $0.9 million in the year ended December 31, 2025, or 10%, compared with the year ended December 31, 2024. The increase is primarily due to increases in personnel expenses of $0.6 million, including stock-based compensation expenses of $0.2 million.
S&M expense decreased $2.4 million, or 21% in the year ended December 31, 2024, compared with the year ended December 31, 2023. The decrease was primarily due to decreases in personnel expenses, including stock-based compensation expenses of $1.5 million and $0.5 million in salaries from reduced headcount. There was additionally decreases of $0.3 million in consulting expenses.
Research and development
During the years ended December 31, 2025, 2024, and 2023, R&D expense consisted primarily of personnel-related costs, consulting, research supplies, and milestone expenses related to third-party research agreements.
R&D expense increased $2.8 million in the year ended December 31, 2025, or 59%, compared with the year ended December 31, 2024. The increase is primarily due to increases in personnel expenses, including $1.1 million in salaries from an increased headcount, $0.9 million in stock-based compensation, and $0.5 million in bonus expenses.
R&D expenses decreased $0.8 million in the year ended December 31, 2024, or 14%, compared with the year ended December 31, 2023. The decrease is primarily due to a decrease of $0.6 million in research milestone costs related to our historical PanTHERA Development and License Agreement (as defined in Note 2: Acquisition within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report) in addition to a decrease of $0.5 million in stock-based compensation when compared to the prior year. The decrease in R&D expenses for the year ended December 31, 2024 was offset by a $0.3 million increase in product testing expenses compared with the year ended December 31, 2023.
IPR&D expense
In-process research and development ("IPR&D") expense during the year ended December 31, 2025 consists of the immediate $15.5 million expense of the IPR&D asset we acquired in the PanTHERA Transaction. For additional information on the details of the PanTHERA Transaction, see Note 2: Acquisition within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
Intangible asset amortization expense
Amortization expense consists of charges related to the amortization of intangible assets associated with previous acquisitions in which we acquired definite-lived intangible assets.
Change in fair value of contingent consideration
Change in fair value of contingent consideration consists of changes in estimated fair value of our potential earnouts related to previous acquisitions. The benefit recognized in the year ended December 31, 2023 related primarily to changes in our estimated probability of achieving earnout targets set forth within the purchase agreements. The related liability was written off during the year ended December 31, 2023 due to target revenues not being met or probable to achieve in future periods.
Other income and expenses
Total other income and expenses for the years ended December 31, 2025, 2024, and 2023 were comprised of the following:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| Interest income (expense), net | 2,706 | | | (766) | | | (1,406) | | | 3,472 | | | 453 | % | | 640 | | | 46 | % |
| Other income | 1,815 | | | 494 | | | 1,275 | | | 1,321 | | | 267 | % | | (781) | | | (61) | % |
| Change in fair value of investments | — | | | (4,074) | | | — | | | 4,074 | | | 100 | % | | (4,074) | | | — | % |
| Gain on settlement of Global Cooling escrow | — | | | — | | | 5,115 | | | — | | | — | % | | (5,115) | | | 100 | % |
| Total other income (expense), net | $ | 4,521 | | | $ | (4,346) | | | $ | 4,984 | | | $ | 8,867 | | | (204) | % | | $ | (9,330) | | | (187 | %) |
Interest income (expense), net
Interest expense incurred in the year ended December 31, 2025 related primarily to the Term Loan (as defined in Note 12: Long-term debt within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report), financed insurance premiums, and indirect tax liabilities. We also earn interest on cash held in our money market account and on our available-for-sale security investments. Decreases in interest expense, net during the year ended December 31, 2025 is attributed to the increases in interest income from our available-for-sale securities compared to the year ended December 31, 2024.
Other income
Other income consists of various non-cash income and expenses, primarily reflecting activity in the accretion or amortization of our available-for-sale securities and other investments. The increase in other income during the year ended December 31, 2025 is primarily due to increased income from the accretion of our available-for-sale securities in addition to the increase in the balance of our financial instruments measured at fair value.
Change in fair value of investments
Reflects fair value adjustments to our investment in iVexSol (as defined in Note 5: Investments within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report). As of June 30, 2024, we determined that the fair value of its equity interest was less than its carrying amount, and no longer recoverable, triggering an impairment charge of $4.1 million, which represented the entirety of the value of the investment.
Gain on settlement of Global Cooling escrow
Reflects the non-cash gain associated with our post-closing adjustments for indemnifications and negotiated terms in connection with our acquisition of Global Cooling, and subsequent release and cancellation of these shares of our common stock from the third-party escrow account established in connection with that transaction in 2023. For additional information, see Note 11 within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
Income Tax Expense (Benefit)
Income tax benefit for the years ended December 31, 2025, 2024, and 2023 was as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| Income tax expense (benefit) | $ | 49 | | | $ | (38) | | | $ | (64) | | | $ | 87 | | | (229) | % | | $ | 26 | | | (41 | %) |
| Effective tax rate | (0.4 | %) | | 0.4 | % | | 1.0 | % | | | | | | | | |
The income tax expense (benefit) recognized in the year ended December 31, 2025 primarily related to losses generated in 2025. Our effective tax rate for 2025 was lower than the U.S. statutory rate of 21% primarily due to the change in our valuation allowance.
Liquidity and capital resources
We believe our cash, cash equivalents, cash generated from operations, available-for-sale securities, and credit lines will satisfy, for at least the foreseeable future, our liquidity requirements, both globally and domestically, including the following: working capital needs, capital expenditures, contractual obligations, commitments, principal and interest payments on debt, and other liquidity requirements associated with our operations.
Significant cash and non-cash expenses related to acquisitions and divestitures
On October 6, 2025, we consummated the SAVSU Divestiture. In connection with the closing of the transaction, we received net proceeds of $23.9 million, including $2.5 million indemnity holdback which management expects to receive in full one year after the closing date. We also incurred additional expenses related to the SAVSU Divestiture, including $1.5 million to the brokers, attorneys, and other external parties for legal and other transaction services. We also recognized $1.3 million in stock compensation expense in connection with the acceleration of unvested shares for all former employees that remained with SAVSU upon the closing of this transaction in addition to providing a retention bonus for all former employees of $0.5 million. For additional information on the SAVSU Divestiture, see Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
On April 4, 2025, we consummated the PanTHERA Transaction. The aggregate purchase price of the acquisition was $16.8 million, which included $11.5 million in cash and 213,360 shares of our common stock. Additionally, pursuant to the PanTHERA Purchase Agreement, the PanTHERA Sellers are eligible to receive up to $7.2 million in cash or equivalent shares of the Company's common stock (as elected by the PanTHERA Sellers) over a three-year earnout period upon the achievement of certain revenue targets based on the Company's earnings derived from the acquired IRI GEN 2 cryopreservation technology in addition to the achievement of an operational milestone within the first year of the earnout period. For additional information on the PanTHERA Transaction, see Note 2: Acquisition within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
On November 14, 2024, we consummated the CBS Divestiture. In connection with the closing of the transaction, we received net proceeds of $3.4 million. We also incurred additional expenses related to the CBS Divestiture, including $0.1 million to the brokers, attorneys, and other external parties for legal and other transaction services. We also recognized $2.0 million in stock compensation expense in connection with the acceleration of unvested shares for all former employees that remained with CBS upon the closing of this transaction. For additional information on the CBS Divestiture, see Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
On November 12, 2024, we consummated the SciSafe Divestiture. In connection with the closing of this transaction, we received net proceeds of $71.3 million. We also incurred additional expenses related to SciSafe Divestiture, including $0.5 million to the brokers, attorneys, and other external parties for legal and other transaction services, and incurred $0.4 million in severance costs. We also paid the former stockholders of SciSafe approximately $3.3 million in cash to waive all rights with respect to certain potential earn-out payments and recognized $4.0 million in stock compensation expense in connection with the acceleration of unvested shares for all of our former employees that remained with SciSafe upon the closing of this transaction. For additional information on the SciSafe Divestiture, see Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
On April 17, 2024, we consummated the Global Cooling Divestiture. In connection with the closing of the transaction, we provided $6.7 million in cash funding to effectuate the Global Cooling Divestiture and paid $0.6 million to the brokers, attorneys, and other external parties. In addition, we recognized $6.1 million in cash expenditures from operations during the third quarter of 2024 to meet certain post-closing requirements, costs to sell Global Cooling, the assumption of certain liabilities and debt, and severance expenses related to the reduction in force ("RIF") implemented on the business of Global Cooling, which reduced our workforce by 47 employees. For additional information on the details of the Global Cooling Divestiture, the RIF and its related costs, see Note 3: Discontinued operations within the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
Change in cash, cash equivalents, and available-for-sale securities
On December 31, 2025, we had $120.2 million in cash, cash equivalents, and available-for-sale securities, compared to $105.4 million as of December 31, 2024, as follows:
| | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 |
| (In thousands, except percentages) | 2025 | | 2024 | | $ Change | | % Change |
| Cash and cash equivalents | $ | 33,038 | | | $ | 91,538 | | | $ | (58,500) | | | (64) | % |
| Available-for-sale securities | 87,139 | | | 13,826 | | | 73,313 | | | 530 | % |
| Maturities in less than one year | 55,889 | | | 9,198 | | | 46,691 | | | 508 | % |
| Maturities in greater than one year | 31,250 | | | 4,628 | | | 26,622 | | | 575 | % |
| Total cash, cash equivalents, and available-for-sale securities | $ | 120,177 | | | $ | 105,364 | | | $ | 14,813 | | | 14 | % |
The decrease in cash and cash equivalents of $58.5 million as of December 31, 2025 as compared with the year ended December 31, 2024 is primarily due to $73.3 million of net investments into available-for-sale securities during the current year, investments in capital expenditure of $9.5 million, and the IPR&D asset purchase in the PanTHERA Transaction of $10.2 million. This decrease was partially offset by $23.5 million in cash proceeds received from the SAVSU Divestiture and cash provided by operating activities of $20.1 million.
Our available-for-sale securities consist of U.S. government securities, corporate debt securities, and other debt securities. Management classifies investments at the time of purchase and reevaluates such classification at each balance sheet date. The increase in available-for-sale securities of $73.3 million primarily resulted from the investment of $114.1 million in available-for-sale securities, offset by $34.8 million in maturities and $6.8 million in cash proceeds.
Cash flows
We have elected to present the Consolidated Statements of Cash Flows on a consolidated basis rather than a continuing operations basis with effect to the divestitures of SAVSU, CBS, SciSafe, and Global Cooling. The discussions regarding changes in cash activity in this section are therefore reflective of consolidated results inclusive of operating results of the divested entities.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | 2025 vs. 2024 | | 2024 vs. 2023 |
| (In thousands, except percentages) | 2025 | | 2024 | | 2023 | | $ Change | | % Change | | $ Change | | % Change |
| Operating activities | $ | 20,115 | | | $ | 8,431 | | | $ | (12,498) | | | $ | 11,684 | | | 139 | % | | $ | 20,929 | | | 167 | % |
| Investing activities | (71,539) | | | 58,300 | | | 17,837 | | | (129,839) | | | (223 | %) | | 40,463 | | | 227 | % |
| Financing activities | (10,924) | | | (6,783) | | | 10,591 | | | (4,141) | | | (61) | % | | (17,374) | | | (164) | % |
| Net (decrease) increase in cash and cash equivalents | $ | (62,348) | | | $ | 59,948 | | | $ | 15,930 | | | $ | (122,296) | | | (204 | %) | | $ | 44,018 | | | 276 | % |
Operating activities
In the year ended December 31, 2025, our operating activities provided cash of $20.1 million, reflecting non-cash charges totaling $24.7 million primarily related to stock-based compensation, gain recognized on disposals of subsidiaries, the expense we incurred on the IPR&D asset purchased in the PanTHERA Transaction, depreciation, amortization, and changes in fair value of investments. Significant changes in operating assets and liabilities include an increase of prepaid expenses of $3.0 million, a decrease of accounts payable of $2.1 million, and a decrease of accrued expenses of $1.1 million.
In the year ended December 31, 2024, our operating activities provided cash of $8.4 million, reflecting non-cash charges totaling $28.6 million primarily related to stock-based compensation, gain recognized on disposals of subsidiaries, depreciation, amortization, and changes in fair value of investments. Significant changes in operating assets and liabilities include an increase of accounts receivable of $2.9 million, an increase of prepaid expenses of $2.4 million, and a decrease in accrued expenses of $6.5 million.
Investing activities
Our investing activities used $71.5 million of cash in the year ended December 31, 2025. This was primarily used for the $114.1 million of investments in available-for-sale securities, offset by $34.8 million in maturities of available-for-sale securities. We additionally invested $10.2 million in the IPR&D asset purchased in the PanTHERA Transaction. Our investing activity cash usage was offset by $23.5 million in proceeds from the SAVSU Divestiture.
In the year ended December 31, 2024, investing activities provided $58.3 million of cash. We had $73.4 million in proceeds from the divestitures of SciSafe and CBS, offset by cash payments of $13.0 million for the Global Cooling Divestiture. We additionally incurred $5.3 million in capital expenditures and purchases of assets held for rent to maintain and expand the Company's operations.
Financing activities
In the year ended December 31, 2025, cash used by financing activities was $10.9 million. The use of cash in financing activities was primarily related to $11.0 million in payments on our Term Loan and financed insurance premiums.
In the year ended December 31, 2024, cash used by financing activities was $6.8 million. The use of cash in financing activities was primarily related to $9.0 million in payments on our Term Loan, equipment loans, and financed insurance premiums, which was offset by proceeds from financed insurance premiums of $2.1 million.
Contractual obligations
Our cash flows from operations depend on a number of factors, including fluctuations in our operating results, accounts receivable collections, inventory management, and the timing of tax and other payments. As a result, the impact of contractual obligations on our liquidity and capital resources in future periods should be analyzed in conjunction with such factors. Despite these uncertainties, we believe that our balances of cash, cash equivalents, and available-for-sale securities in addition to our cash flows from operations are adequate to meet our liquidity requirements in the foreseeable future.
The following summarizes certain of our contractual obligations as of December 31, 2025 and the effect such obligations are expected to have on our cash flows in the next fiscal year:
Long-term debt, including interest
These amounts represent expected cash payments, including principal and interest. Debt obligations are described in Note 12: Long-term debt of the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. As of December 31, 2025, our total obligations were $5.0 million, all of which was short-term.
Lease obligations
We have various operating lease agreements for office space, warehouses, manufacturing, research equipment, machinery, and production locations as well as vehicles and other equipment. Lease obligations are described in Note 7: Leases of the Consolidated Financial Statements in Part II, Item 8 of this Annual Report. As of December 31, 2025, our total obligations were $12.9 million, of which $2.2 million was short-term.
Purchase obligations
Purchase obligations are defined as agreements to purchase goods or services that are enforceable and legally binding and that specify all significant terms, including fixed or minimum quantities to be purchased, fixed, minimum or variable pricing provisions and the approximate timing of the transactions. As of December 31, 2025, our total obligations were $14.5 million, of which $9.8 million was short-term.
Sales Tax
We remain in the process of evaluating our state sales tax liabilities for states in which we have economic nexus and collecting exemption documentation from our customers. It is probable that we will be subject to sales tax liabilities plus interest and penalties relating to historical activity in certain states. We have estimated a contingent liability for sales tax which is recorded in the Consolidated Balance Sheet. The liability includes significant judgments and estimates that may
change in the future, and the liability may exceed our current estimate. We may be subject to examination by the relevant state tax authorities, and we can provide no assurances that outcomes from these examinations will not have a significant effect on our operating results, financial condition, and cash flows.
Capital requirements
Our future capital requirements will depend on many factors, including the following:
•the expansion of our cell and gene therapy business
•the ability to sustain product revenue and profits of our cell and gene therapy products and services;
•the degree to which we implement additional automated production equipment throughout our facilities;
•our ability to acquire additional cell and gene therapy products and services;
•the scope of and progress made in our research and development activities; and
•the success of any proposed financing efforts.
Absent acquisitions of additional products, product candidates, or intellectual property, we believe our current cash, cash equivalents, and available-for-sale securities balances, in addition to our cash flows from operations, are adequate to meet our cash needs for the foreseeable future. We expect to incur continued spending related to our existing product lines and expansion of our commercial capabilities for the foreseeable future. Our future capital requirements may include, but are not limited to, purchases of property and equipment, internal development of products and services, the acquisition of additional cell and gene therapy products and technologies, and continued investment in our intellectual property portfolio.
We actively evaluate various strategic transactions on an ongoing basis, including acquiring complementary products, technologies or businesses that would augment our existing portfolio. We continue to seek to acquire such potential assets that may offer us the best opportunity to create value for our shareholders. In order to acquire such assets, we may need to seek additional financing to fund these investments. If our available cash balances and anticipated cash flow from operations are insufficient to satisfy our liquidity requirements, including because of any such acquisition-related financing needs or lower demand for our products, we may seek to sell common or preferred equity or convertible debt securities, enter into a credit facility or another form of third-party funding, or seek other debt funding. The sale of equity and convertible debt securities may result in dilution to our stockholders, and those securities may have rights senior to those of our common shares. If we raise additional funds through the issuance of preferred stock, convertible debt securities or other debt financing, these securities or other debt could contain covenants that would restrict our operations. Any other third-party funding arrangement could require us to relinquish valuable rights. We may require additional capital beyond our currently anticipated amounts. Additional capital may not be available on reasonable terms, if at all.
Risks and uncertainties
The preparation of financial statements requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities at the reporting date and revenues and expenses during the reporting periods. These estimates represent management's judgment about the outcome of future events. Macroeconomic factors, geopolitical unrest, inflation, changes in interest and foreign currency exchange rates, tariffs and retaliatory measures, war and other military conflict, and other risks and uncertainties have in the past and may continue to cause logistical challenges, increased input costs, or create constraints for our suppliers, distributors, or customers that could in turn decrease demand for our products, create delays and inefficiencies in our supply chain and make it difficult or impossible for us to deliver products to our customers. It is not possible to accurately predict the future impact of such events and circumstances. Actual results could differ from our estimates.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Foreign currency exchange risk
The Company's sales are primarily denominated in the U.S. dollar. Accordingly, our sales are not generally impacted by foreign currency rate changes. Any transactions denominated in a foreign currency, which were immaterial during the year ended December 31, 2025, incur gains or losses from the remeasurement and settlement of the balances and are reported in the Other income line item on the Consolidated Statements of Operations. Fluctuations in foreign currency exchange rates have not had a material impact on our results of operations for the periods presented.
Interest rate risk
Our exposure to market risk for changes in interest rates relates primarily to our investments in available-for-sale securities and our long-term debt. We invest our excess cash in investment grade short to intermediate-term fixed income securities. These securities may have their fair market value adversely affected due to a rise in interest rates, and we may suffer losses if forced to sell securities that have declined in market value due to changes in interest rates. Our long-term debt primarily bears interest at a fixed rate, with a variable component subject to an interest rate ceiling. Fluctuations in interest rates therefore do not materially impact our Consolidated Financial Statements from long-term debt. For additional information about our available-for-sale securities and long-term debt, see Notes 5: Investments and 12: Long-term debt to the Consolidated Financial Statements in Part II, Item 8 of this Annual Report.
ITEM 8. CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of Directors and Shareholders
BioLife Solutions, Inc.
Opinion on the financial statements
We have audited the accompanying consolidated balance sheets of BioLife Solutions Inc. (a Delaware corporation) and subsidiaries (the “Company”) as of December 31, 2025 and 2024, the related consolidated statements of operations, comprehensive loss, shareholders’ equity, and cash flows for each of the three years in the period ended December 31, 2025, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2025, in conformity with accounting principles generally accepted in the United States of America.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (“PCAOB”), the Company’s internal control over financial reporting as of December 31, 2025, based on criteria established in the 2013 Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”), and our report dated February 26, 2026 expressed an unqualified opinion.
Basis for opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical audit matters
The critical audit matters are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there are no critical audit matters.
/s/ GRANT THORNTON LLP
We have served as the Company's auditor since 2022.
Bellevue, Washington
February 26, 2026
BioLife Solutions, Inc.
Consolidated Balance Sheets
| | | | | | | | | | | |
| December 31, |
| (In thousands, except per share and share data) | 2025 | | 2024 |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 33,038 | | | $ | 91,538 | |
| Available-for-sale securities, current portion | 55,889 | | | 9,198 | |
Accounts receivable, trade, net of allowance for credit losses of $22 and $152 as of December 31, 2025 and December 31, 2024, respectively | 7,953 | | | 8,476 | |
| Inventories | 30,155 | | | 29,013 | |
| Prepaid expenses and other current assets | 9,526 | | | 5,952 | |
| Current assets, discontinued operations | — | | | 4,584 | |
| Total current assets | 136,561 | | | 148,761 | |
| | | |
| Property and equipment, net | 14,713 | | | 5,974 | |
| Operating lease right-of-use assets, net | 7,466 | | | 10,553 | |
| Other assets | 3,050 | | | 373 | |
| Available-for-sale securities, long term | 31,250 | | | 4,628 | |
| Equity investments | — | | | 995 | |
| Intangible assets, net | 4,257 | | | 5,565 | |
| Goodwill | 208,587 | | | 208,587 | |
| Long-term assets, discontinued operations | — | | | 14,051 | |
| Total assets | $ | 405,884 | | | $ | 399,487 | |
| | | |
| Liabilities and Shareholders’ Equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 1,486 | | | $ | 3,360 | |
| Accrued expenses and other current liabilities | 11,974 | | | 12,123 | |
| Sales taxes payable | 2,279 | | | 4,233 | |
| Lease liabilities, operating, current portion | 2,243 | | | 1,459 | |
| Debt, current portion | 4,997 | | | 10,943 | |
| Current liabilities, discontinued operations | — | | | 616 | |
| Total current liabilities | 22,979 | | | 32,734 | |
| | | |
| Lease liabilities, operating, long-term | 10,657 | | | 12,645 | |
| Debt, long-term | — | | | 4,997 | |
| Deferred tax liabilities | 189 | | | 124 | |
| Other long-term liabilities | 171 | | | — | |
| Long-term liabilities, discontinued operations | — | | | 78 | |
| Total liabilities | 33,996 | | | 50,578 | |
| | | |
| Commitments and contingencies (Note 11) | | | |
| | | |
| Shareholders’ equity: | | | |
Preferred stock, $0.001 par value; 1,000,000 shares authorized, Series A, 4,250 shares designated, and 0 shares issued and outstanding as of December 31, 2025 and December 31, 2024 | — | | | — | |
| | | | | | | | | | | |
Common stock, $0.001 par value; 150,000,000 shares authorized, 48,198,928 and 46,906,765 shares issued and outstanding as of December 31, 2025 and December 31, 2024, respectively | 48 | | | 47 | |
| Additional paid-in capital | 711,404 | | | 683,939 | |
| Accumulated other comprehensive loss, net of taxes | 132 | | | 24 | |
| Accumulated deficit | (339,696) | | | (335,101) | |
| Total shareholders’ equity | 371,888 | | | 348,909 | |
| Total liabilities and shareholders’ equity | $ | 405,884 | | | $ | 399,487 | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements
BioLife Solutions, Inc.
Consolidated Statements of Operations
| | | | | | | | | | | | | | | | | |
| Years Ended December 31 |
| (In thousands, except per share and share data) | 2025 | | 2024 | | 2023 |
| | | | | |
| Revenue | $ | 96,214 | | | $ | 74,647 | | | $ | 68,016 | |
| Cost of revenue | 34,096 | | | 24,551 | | | 26,698 | |
| Gross profit | 62,118 | | | 50,096 | | | 41,318 | |
| Operating expenses: | | | | | |
| General and administrative | 45,520 | | | 40,627 | | | 42,792 | |
| Sales and marketing | 9,850 | | | 8,932 | | | 11,377 | |
| Research and development | 7,566 | | | 4,751 | | | 5,515 | |
| IPR&D expense | 15,521 | | | — | | | — | |
| Intangible asset amortization | 263 | | | 263 | | | 1,032 | |
| Change in fair value of contingent consideration | — | | | — | | | (2,193) | |
| Total operating expenses | 78,720 | | | 54,573 | | | 58,523 | |
| Operating loss | (16,602) | | | (4,477) | | | (17,205) | |
| | | | | |
| Other income (expense): | | | | | |
| Interest income (expense), net | 2,706 | | | (766) | | | (1,406) | |
| Other income | 1,815 | | | 494 | | | 1,275 | |
| Change in fair value of investments | — | | | (4,074) | | | — | |
| Gain on settlement of Global Cooling escrow | — | | | — | | | 5,115 | |
| Total other income (expense), net | 4,521 | | | (4,346) | | | 4,984 | |
| | | | | |
| Loss from continuing operations before income tax expense (benefit) | (12,081) | | | (8,823) | | | (12,221) | |
| Income tax expense (benefit) | 49 | | | (38) | | | (64) | |
| Loss from continuing operations | (12,130) | | | (8,785) | | | (12,157) | |
| | | | | |
| Discontinued operations: | | | | | |
| Income (loss) from discontinued operations before income tax expense | 7,535 | | | (11,267) | | | (55,612) | |
| Income tax expense | — | | | 132 | | | 233 | |
| Income (loss) from discontinued operations | 7,535 | | | (11,399) | | | (55,845) | |
| | | | | |
| Net loss | $ | (4,595) | | | $ | (20,184) | | | $ | (68,002) | |
| | | | | |
| Loss per share - Basic: | | | | | |
| Continuing operations | $ | (0.25) | | | $ | (0.19) | | | $ | (0.28) | |
| Discontinued operations | $ | 0.16 | | | $ | (0.25) | | | $ | (1.27) | |
| Net loss | $ | (0.09) | | | $ | (0.44) | | | $ | (1.55) | |
| Loss per share - Diluted: | | | | | |
| Continuing operations | $ | (0.25) | | | $ | (0.19) | | | $ | (0.28) | |
| Discontinued operations | $ | 0.15 | | | $ | (0.25) | | | $ | (1.27) | |
| Net loss | $ | (0.10) | | | $ | (0.44) | | | $ | (1.55) | |
| Weighted average shares used to compute net loss per share attributable to common shareholders: | | | | | |
| Basic | 47,753,824 | | 46,067,073 | | 43,719,185 |
| Diluted | 48,645,084 | | 46,067,073 | | 43,719,185 |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements
BioLife Solutions, Inc.
Consolidated Statements Of Comprehensive Loss
| | | | | | | | | | | | | | | | | |
| Years Ended December 31 |
| (In thousands) | 2025 | | 2024 | | 2023 |
| | | | | |
| Net loss | $ | (4,595) | | | $ | (20,184) | | | $ | (68,002) | |
| | | | | |
| Other comprehensive income - foreign currency translation adjustment, net of tax | — | | | — | | | 278 | |
| Unrealized gain on available-for-sale securities, net of tax | 108 | | | 18 | | | 56 | |
| | | | | |
| Comprehensive loss | $ | (4,487) | | | $ | (20,166) | | | $ | (67,668) | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements
BioLife Solutions, Inc.
Consolidated Statements of Shareholders’ Equity
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands, except share data) | Series A
Preferred
Stock
Shares | | Series A
Preferred
Stock
Amount | | Common
Stock
Shares | | Common
Stock
Amount | | Additional
Paid-in
Capital | | Accumulated
Other
Comprehensive
Loss | | Accumulated
Deficit | | Total
Shareholders’
Equity |
| Balance, December 31, 2022 | — | | $— | | 42,832,231 | | $43 | | $611,739 | | $(679) | | $(246,915) | | $364,188 |
| Stock issued as consideration for SciSafe earnout | — | | — | | 116,973 | | — | | 2,263 | | — | | — | | 2,263 |
| Fees incurred for registration filings | — | | — | | — | | — | | (132) | | — | | — | | (132) |
| Stock-based compensation | — | | — | | — | | — | | 33,245 | | — | | — | | 33,245 |
| Stock option exercises | — | | — | | 239,043 | | — | | 507 | | — | | — | | 507 |
| Stock issued – on vested RSA units | | | | | 1,267,837 | | 1 | | (1) | | — | | — | | — |
| Settlement of Global Cooling escrow | | | | | (216,024) | | — | | (5,115) | | — | | — | | (5,115) |
| Common stock shares issued | — | | — | | 927,165 | | 1 | | 10,374 | | — | | — | | 10,375 |
| Other comprehensive income | — | | — | | — | | — | | — | | 334 | | — | | 334 |
| Net loss | — | | — | | — | | — | | — | | — | | (68,002) | | (68,002) |
| Balance, December 31, 2023 | — | | — | | 45,167,225 | | 45 | | 652,880 | | (345) | | (314,917) | | 337,663 |
| Stock-based compensation | — | | — | | — | | — | | 30,923 | | — | | — | | 30,923 |
| Stock option exercises | — | | — | | 90,250 | | — | | 202 | | — | | — | | 202 |
| Stock issued – on vested RSA units | — | | — | | 1,649,290 | | 2 | | (66) | | — | | — | | (64) |
| Other comprehensive income | — | | — | | — | | — | | — | | 369 | | — | | 369 |
| Net loss | — | | — | | | | | | | | | | (20,184) | | (20,184) |
| Balance, December 31, 2024 | — | | — | | 46,906,765 | | 47 | | 683,939 | | 24 | | (335,101) | | 348,909 |
| Stock-based compensation | — | | — | | — | | — | | 22,993 | | — | | — | | 22,993 |
| Stock option exercises | — | | — | | 10,000 | | — | | 18 | | — | | — | | 18 |
| Stock issued – on vested RSA units | — | | — | | 1,068,803 | | 1 | | (1) | | — | | — | | — |
| Common stock shares issued for PanTHERA Transaction | — | | — | | 213,360 | | | | 4,455 | | | | | | 4,455 |
| Other comprehensive income | — | | — | | — | | — | | — | | 108 | | — | | 108 |
| Net loss | — | | — | | — | | — | | — | | — | | (4,595) | | (4,595) |
| Balance, December 31, 2025 | — | | $— | | 48,198,928 | | $48 | | $711,404 | | $132 | | $(339,696) | | $371,888 |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements
BioLife Solutions, Inc.
Consolidated Statements of Cash Flows
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| (In thousands) | 2025 | | 2024 | | 2023 |
| Cash flows from operating activities | | | | | |
| Net loss | $ | (4,595) | | | $ | (20,184) | | | $ | (68,002) | |
| Adjustments to reconcile net loss to net cash provided by (used in) operating activities | | | | | |
| Impairment of intangible assets | — | | | — | | | 5,758 | |
| Impairment of long-lived assets | — | | | — | | | 9,727 | |
| Gain on settlement of Global Cooling escrow | — | | | — | | | (5,115) | |
| Depreciation | 2,253 | | | 5,160 | | | 7,114 | |
| Amortization of intangible assets | 2,434 | | | 3,501 | | | 5,181 | |
| Amortization of loan costs | — | | | — | | | 13 | |
| Stock-based compensation | 22,993 | | | 30,923 | | | 33,245 | |
| Non-cash lease expense | 1,875 | | | 424 | | | 404 | |
| Deferred income tax expense (benefit) | 65 | | | (64) | | | (62) | |
| Change in fair value of contingent consideration | — | | | — | | | (2,193) | |
| Change in fair value of equity investment and other | (983) | | | 4,074 | | | — | |
| Accretion of investments | (736) | | | (474) | | | (1,262) | |
| Loss on disposal of assets held for rent, net | 510 | | | 567 | | | 594 | |
| Loss on disposal of property and equipment, net | 11 | | | 154 | | | 633 | |
| IPR&D expense | 15,521 | | | — | | | — | |
| Net gain on disposal of subsidiaries | (10,324) | | | (15,877) | | | — | |
| | | | | |
| | | | | |
| Change in operating assets and liabilities | | | | | |
| Accounts receivable, trade, net | 264 | | | (2,899) | | | 15,351 | |
| Inventories | (1,268) | | | 781 | | | (8,552) | |
| Prepaid expenses and other current assets | (3,039) | | | (2,407) | | | 137 | |
| Accounts payable | (2,086) | | | 26 | | | (8,425) | |
| Accrued expenses and other current liabilities | (1,144) | | | 6,485 | | | 2,002 | |
| Sales taxes payable | (1,905) | | | (973) | | | 1,311 | |
| Warranty liability | 5 | | | (528) | | | (454) | |
| Other | 264 | | | (258) | | | 97 | |
| Net cash provided by (used in) operating activities | 20,115 | | | 8,431 | | | (12,498) | |
| | | | | |
| Cash flows from investing activities | | | | | |
| Purchases of property and equipment | (9,478) | | | (3,227) | | | (6,381) | |
| Purchases of assets held for rent | (886) | | | (2,068) | | | (4,856) | |
| Proceeds from sale of available-for-sale securities | 6,829 | | | 3,518 | | | 3,469 | |
| Maturities of available-for-sale securities | 34,764 | | | 18,400 | | | 52,700 | |
| Investment in available-for-sale securities | (114,063) | | | (18,415) | | | (27,095) | |
| Purchases of intangible assets | — | | | (300) | | | — | |
| Investment in IPR&D | (10,221) | | | — | | | — | |
| Investment in convertible promissory note | (2,000) | | | — | | | — | |
| Proceeds from sale of divested entities | 23,516 | | | 73,431 | | | — | |
| Payments on divestiture of Global Cooling | — | | | (13,039) | | | — | |
| Net cash (used in) provided by investing activities | (71,539) | | | 58,300 | | | 17,837 | |
| | | | | |
| Cash flows from financing activities | | | | | |
| Payments on term loan | (10,000) | | | (5,000) | | | (300) | |
| Payments on equipment loans | — | | | (1,608) | | | (198) | |
| | | | | |
| Issuance of common stock | — | | | — | | | 10,244 | |
| | | | | |
| Proceeds from exercise of common stock options | 18 | | | 202 | | | 507 | |
| Proceeds from financed insurance premium | — | | | 2,094 | | | 2,639 | |
| Payments on financed insurance premium | (975) | | | (2,397) | | | (2,365) | |
| Other | 33 | | | (74) | | | 64 | |
| Net cash (used in) provided by financing activities | (10,924) | | | (6,783) | | | 10,591 | |
| | | | | |
| Net (decrease) increase in cash and cash equivalents | (62,348) | | | 59,948 | | | 15,930 | |
| Cash and cash equivalents – beginning of period | 95,386 | | | 35,438 | | | 19,473 | |
| Effects of currency translation on cash and cash equivalents | — | | | — | | | 35 | |
| Cash and cash equivalents – end of period | $ | 33,038 | | | $ | 95,386 | | | $ | 35,438 | |
| Non-cash investing and financing activities | | | | | |
| Assets acquired under operating leases | $ | — | | | $ | 5,833 | | | $ | 880 | |
| Assets acquired under finance leases | $ | — | | | $ | — | | | $ | 1,682 | |
| Purchase of property and equipment not yet paid | $ | — | | | $ | 107 | | | $ | 359 | |
| Unrealized loss (gain) on available-for-sale securities | $ | 108 | | | $ | (18) | | | $ | (56) | |
| Unrealized gain on currency translation | $ | — | | | $ | — | | | $ | (12) | |
| Non-cash acquisition of PanTHERA | $ | 5,300 | | | $ | — | | | $ | — | |
| Cashless issuance of SciSafe earnout shares | $ | — | | | $ | — | | | $ | 2,263 | |
| Returned shares from settlement of Global Cooling escrow | $ | — | | | $ | — | | | $ | (5,115) | |
| Cash interest paid | $ | 762 | | | $ | 1,608 | | | $ | 1,927 | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Organization and significant accounting policies
Business
BioLife Solutions, Inc. (“BioLife”, “us”, “we”, “our”, or the “Company”) is a developer, manufacturer, and supplier of a portfolio of bioproduction products and services including proprietary biopreservation media and cell processing tools. Our CryoStor freeze media and HypoThermosol hypothermic storage media are optimized to preserve cells in the cell and gene therapy market. These novel biopreservation media products are serum-free and protein-free, fully defined, and are formulated to reduce preservation-induced cell damage and death. Our Sexton cell processing product line includes human platelet lysates (“hPL”) for cell expansion, reducing risk and improving downstream performance over fetal bovine serum, human serum, and other chemically defined media, CellSeal closed systems that are purpose-built rigid containers used in cell and gene therapy (“CGT”) that can be filled manually or with high throughput systems, and automated cell processing machines that bring multiple processes traditionally performed by manual techniques under a higher level of control to protect therapies from loss or contamination. Our ThawSTAR product line is comprised of a family of automated thawing devices for frozen cell and gene therapies packaged in cryovials and cryobags. These products help administer temperature-sensitive biologic therapies to patients by standardizing the thawing process and reducing the risks of contamination and overheating, which are inherent with the use of traditional water baths.
Use of estimates
The preparation of financial statements in conformity with generally accepted accounting principles in the United States (“U.S. GAAP”) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities as of the date of the financial statements and reported amounts of revenue and expenses during the reporting period. Actual results could differ from those estimates.
Estimates and assumptions by management affect the Company’s allowance for credit losses, the net realizable value of inventory, sales tax liabilities, valuation of market based stock awards, valuations related to investments and transaction activities, expected future cash flows including growth rates, discount rates, terminal values and other assumptions and estimates used to evaluate the recoverability of long-lived assets, estimated fair values of intangible assets and goodwill, amortization methods and periods, certain accrued expenses, share-based compensation, and the provision for income taxes.
The Company regularly assesses these estimates; however, actual results could differ materially from these estimates. Changes in estimates are recorded in the period in which they become known. The Company bases its estimates on historical experience and various other assumptions that it believes to be reasonable under the circumstances.
Basis of presentation and consolidation
The Consolidated Financial Statements include the accounts of the Company and its wholly owned subsidiaries. All intercompany accounts and transactions have been eliminated in consolidation.
The Company is presenting SAVSU, CBS, SciSafe, and Global Cooling as discontinued operations for all periods presented within the Consolidated Balance Sheets and Consolidated Statements of Operations. The Consolidated Statements Of Comprehensive Loss, Consolidated Statements of Shareholders' Equity, and Consolidated Statements of Cash Flows are presented on a consolidated basis for both continuing operations and discontinued operations. All amounts, percentages, and disclosures for all periods presented reflect only the continuing operations of the Company unless otherwise noted. See Note 3: Discontinued operations for additional details about the divestitures.
All long-lived assets are maintained in the United States of America.
Segment reporting
The Company views its operations and makes decisions regarding how to allocate resources and manages its business as one reportable segment and one reporting unit. The Company’s Chief Executive Officer, who is the chief operating decision maker ("CODM"), reviews financial information on an aggregate basis for purposes of allocating resources and evaluating financial performance.
Revenue recognition
To determine revenue recognition for contractual arrangements that we determine are within the scope of Financial Accounting Standards Board ("FASB") Accounting Standards Codification ("ASC") Topic 606, Revenue from Contracts with Customers, we perform the following five steps: (i) identify each contract with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to our performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy the relevant performance obligation. We only apply the five-step model to contracts when it is probable that we will collect the consideration we are entitled to in exchange for the goods or services we transfer to the customer. Contracts with customers may contain multiple performance obligations. For such arrangements, the transaction price is allocated to each performance obligation based on the observable and estimated relative standalone selling prices of the promised products or services underlying each performance obligation. The Company determines standalone selling prices based on the price at which the performance obligation is sold separately. If the standalone selling price is not observable through past transactions, the Company estimates the standalone selling price, taking into account available information such as market conditions and internally approved pricing guidelines related to the performance obligations. Payment terms and conditions vary, although terms generally include a requirement of payment within 30 to 60 days.
The Company primarily recognizes product revenues. Product revenues are generated from the sale of biopreservation media and cell processing tools. We recognize product revenue, including shipping and handling charges billed to customers, at a point in time when we transfer control of our products to our customers, which is upon shipment for substantially all transactions. Shipping and handling costs are classified as part of cost of product revenue in the Consolidated Statements of Operations.
Any remaining revenues earned, which primarily consisted of service revenues generated from various customer service agreements for the provision of warranty and other engineering services and equipment rental revenues, were not significant in any of the periods presented.
There was no estimated revenue expected to be recognized in the future related to performance obligations that are unsatisfied or partially unsatisfied as of the end of the reporting periods as of December 31, 2025. The Company elected not to disclose the value of the remaining unsatisfied performance obligation with a duration of one year or less as permitted by the practical expedient in Accounting Standards Update ("ASU") 2014-09, Revenue from Contracts with Customers.
For additional information on the Company's revenues by geographic region, product line, and other customer concentration information, see Note 15: Segment, customer, and geographic information.
Risks and uncertainties
The preparation of financial statements requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities at the reporting date and revenues and expenses during the reporting periods. These estimates represent management's judgment about the outcome of future events. Macroeconomic factors, geopolitical unrest, inflation, changes in interest and foreign currency exchange rates, tariffs and retaliatory measures, war and other military conflict, and other risks and uncertainties have in the past and may continue to cause logistical challenges, increased input costs, or create constraints for our suppliers, distributors, or customers that could in turn decrease demand for our products, create delays and inefficiencies in our supply chain and make it difficult or impossible for us to deliver products to our customers. It is not possible to accurately predict the future impact of such events and circumstances. Actual results could differ from our estimates.
Loss per share from continuing operations
Basic earnings per share is calculated by dividing net income by the weighted average number of shares of common stock during the reporting period. Diluted earnings per share is calculated using the weighted average number of shares of common stock plus the potentially dilutive effect of common equivalent shares outstanding determined under both the two-class method and the treasury stock method, whichever is more dilutive. In periods when we have a net loss, common stock equivalents are excluded from our calculation of earnings per share as their inclusion would have an antidilutive effect.
The following table presents computations of basic and diluted earnings per share:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| (In thousands, except share and earnings per share data) | 2025 | | 2024 | | 2023 |
| Basic and diluted loss from continuing operations per common share | | | | | |
| Numerator: | | | | | |
| Loss from continuing operations | $ | (12,130) | | | $ | (8,785) | | | $ | (12,157) | |
| | | | | |
| Denominator: | | | | | |
| Weighted-average common shares issued and outstanding - Basic | 47,753,824 | | 46,067,073 | | 43,719,185 |
| | | | | |
| | | | | |
| | | | | |
| Net loss per share from continuing operations attributable to common shareholders | | | | | |
| Basic | $ | (0.25) | | | $ | (0.19) | | | $ | (0.28) | |
| | | | | |
| | | | | |
| Anti-dilutive shares | 891,260 | | | 1,283,885 | | | 647,348 | |
Cash and cash equivalents
Cash equivalents consist primarily of interest-bearing money market accounts. We consider all highly liquid debt instruments purchased with an initial maturity of three months or less to be cash equivalents. We maintain cash balances that may exceed federally insured limits. We do not believe that this results in any significant credit risk.
Investments
Available-for-sale securities
Available-for-sale securities consist of U.S. government securities, corporate debt securities, and other debt securities. Management classifies investments at the time of purchase and reevaluates such classification at each balance sheet date. Investments with maturities beyond one year may be classified as short-term based on their liquid nature and because such marketable securities represent the investment of cash that is available for current operations. Available-for-sale securities are reported at fair value based on quoted market prices and other observable market data. Unrealized gains and losses are reported as a component of other comprehensive (loss) income, net of any related tax effect. Realized gains and losses are calculated using the specific identification method and recorded as interest income or expense. Any other-than-temporary impairments on investments are included in other income (expense).
Equity investments
We periodically invest in securities of private companies to promote business and strategic objectives. These investments were measured and recorded as follows:
Non-marketable equity securities are equity securities without a readily determinable fair value. In November of 2020, the Company invested $1.0 million in Class E Preferred Shares in PanTHERA CryoSolutions, Inc. (“PanTHERA”). In conjunction with this investment, the Company executed a development and license agreement with PanTHERA (the “PanTHERA Development and License Agreement”)under which the Company was to make milestone development payments up to $2.0 million in the event that certain milestones were met in exchange for exclusive, perpetual, worldwide marketing and distribution rights to the technology for use in CGT applications. As of December 31, 2024, the Company had paid $1.9 million in milestone development payments. The Preferred Stock investments in PanTHERA were carried at cost minus impairment, if any, plus or minus changes resulting from observable process changes in orderly transactions for identical or similar investments of the same issuer. As of December 31, 2024, the Company’s investment in Series E Preferred Stock in PanTHERA was valued at $1.0 million. During the second quarter of 2025, the Company acquired the remaining 90% of the outstanding shares of capital stock of PanTHERA pursuant to a Stock Purchase Agreement (the “PanTHERA Purchase Agreement”), by and among the Company, Casdin Partners Master Fund L.P. and each other person
listed on Schedule A thereto (the “PanTHERA Sellers”), 2699979 Alberta LTD., an Alberta corporation and a wholly owned subsidiary of the Company (“PanTHERA Buyer Sub”), PanTHERA and Dr. Jason Acker, solely in his capacity as Sellers’ Representative (the “PanTHERA Transaction”). The previously held equity interest in PanTHERA was added to the overall consideration paid to determine the costs to allocate to assets in the PanTHERA Transaction and the remaining liability for milestone development payments were extinguished. For additional details on the details of the asset acquisition, see Note 2: Acquisition.
In November of 2020, the Company elected to convert a convertible note into Series A-1 Preferred Stock and invest an additional $1.0 million in Series A-2 Preferred Stock in iVexSol. The Preferred Stock investments in iVexSol were carried at cost minus impairment, if any, plus or minus changes resulting from observable process changes in orderly transactions for identical or similar investments of the same issuer. For the period ending December 31, 2023, the fair value of the Company's shares was $4.1 million. During the second quarter ending June 30, 2024, the Company received communications that triggered substantial doubt about a going concern for the investment. As of June 30, 2024, the Company determined that the fair value of its equity interest was less than its carrying amount, and no longer recoverable, triggering an impairment charge of $4.1 million, which represented the entirety of the value of the investment.
Fair value
In accordance with FASB ASC Topic 820, Fair Value Measurements and Disclosures, (“ASC 820”), the Company measures its financial instruments at fair value on a recurring basis. The carrying values of certain financial instruments including cash and cash equivalents, accounts receivable, accounts payable, and accrued liabilities approximate fair value because of their short maturities. The carrying value of the Company's marketable debt securities, which are accounted for as available-for-sale, are classified within either Level 1 or Level 2 in the fair value hierarchy due to the use of quoted market prices or alternative pricing sources and models utilizing market observable inputs to determine their fair value. The carrying values of the Company's long-term debt, which is classified within Level 2 in the fair value hierarchy, approximates fair value as the borrowings with lenders are at interest rates that approximate market rates for comparable loans. ASC 820 clarifies that fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. As a basis for considering such assumptions, ASC 820 establishes a three-tier value fair hierarchy, which prioritizes the inputs used in measuring fair value as follows:
Level 1 – Observable inputs that reflect quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level 2 – Observable inputs other than quoted prices included in Level 1 for similar assets or liabilities, quoted prices in markets that are not active or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the related assets or liabilities.
Level 3 – Unobservable data points for the asset or liability, and include situations where there is little, if any, market activity for the asset or liability.
There were no remeasurements to fair value during the year ended December 31, 2025 of financial assets and liabilities that are not measured at fair value on a recurring basis.
For certain financial instruments, the Company may elect to utilize the fair value option ("FVO") in accordance with ASC Topic 825, Financial Instruments ("ASC 825"). The Level 3 financial instrument contains embedded features wherein the entire financial instrument was initially measured at its issue-date estimated fair value and will be subsequently remeasured at estimated fair value on a recurring basis at each reporting period date using a Monte Carlo simulation model. Significant unobservable inputs utilized in the valuation include the estimated probability of certain qualified settlement events, the estimated equity value of the investee, the projected revenue of the investee, the risk-adjust discount rate, and volatility rate. Significant changes in any of those inputs in isolation may result in significant changes in fair value measurement. The carrying value of the Company's Level 3 financial instrument is presented within Other assets in the Consolidated Balance Sheets. The Company elected to disclose changes in the estimated fair value of the Level 3 financial instrument as a component of Other income in the Consolidated Statements of Operations. For further information on the nature of this financial instrument, see Note 4: Fair value measurement.
Inventories
Inventories relate to the Company’s products. The Company values biopreservation media inventory at cost or, if lower, net realizable value, using the specific identification method. The Company values thaw inventory at cost or, if lower, net realizable value, using the average costing method. All other inventory is valued at cost or, if lower, net realizable value, using the first-in, first-out method. The Company reviews its inventories at least quarterly and records a provision for inventory that has become obsolete, inventory that has a cost basis in excess of its expected net realizable value, and inventory in excess of expected revenue volume to cost of product revenue. The Company bases its estimates on expected product revenue volume, production capacity and expiration dates of raw materials, work in process, and finished products. A change in the estimated timing or amount of demand for the Company’s products could result in additional provisions for excess inventory quantities on hand. Any significant unanticipated changes in demand or unexpected quality failures could have a significant impact on the value of inventory and reported operating results.
Accounts receivable
Accounts receivable consist of short-term amounts due from our customers and are stated at the amount we expect to collect. We establish an allowance for credit losses based on our assessment of the collectability of specific customer accounts. Accounts considered uncollectible are charged against the established allowance.
Accounts receivable are stated at principal amount, do not bear interest, and are generally unsecured. Accounts considered uncollectible are charged against the established allowance.
Property and equipment
Property and equipment are stated at cost and are depreciated using the straight-line method over estimated useful lives of three to ten years. Leasehold improvements are amortized using the straight-line method over the shorter of the estimated useful lives of the assets or the remaining lease term of the respective assets. Gains or losses on disposals of property and equipment are recorded within income from operations. Costs of repairs and maintenance are included as part of operating expenses unless they are incurred in relation to major improvements to existing property and equipment, at which time they are capitalized.
Long-Lived Assets
The Company reviews long-lived assets, including property and equipment, leased assets, and definite life intangible assets for impairment whenever events and changes in circumstances indicate that the carrying value of an asset may not be recoverable. In order to determine if assets have been impaired, assets are grouped and tested at the lowest level for which identifiable independent cash flows are available ("asset group"). An impairment loss is recognized when the sum of the projected undiscounted cash flows is less than the carrying value of the asset group. The measurement of the impairment loss to be recognized is based on the difference between the fair value and the carrying value of the asset group. Fair value can be determined using a market approach, income approach or cost approach.
Lease accounting
We determine if an arrangement is a lease at inception. Where an arrangement is a lease, we determine if it is an operating lease or a financing lease. At lease commencement, we record a lease liability and corresponding right-of-use (“ROU”) asset. Lease liabilities represent the present value of our future lease payments over the expected lease term which includes options to extend or terminate the lease when it is reasonably certain those options will be exercised. The present value of our lease liability is determined using our incremental collateralized borrowing rate at lease inception. ROU assets represent our right to control the use of the leased asset during the lease and are recognized in an amount equal to the lease liability for leases with an initial term greater than twelve months. Over the lease term we use the effective interest rate method to account for the lease liability as lease payments are made and the ROU asset is amortized in a manner that results in straight-line expense recognition. The Company has elected the practical expedient and does not separate lease components from non-lease components for its leases.
We elected to apply the practical expedient for short-term leases and accordingly do not apply lease recognition requirements for short-term leases with a duration less than twelve months. Instead, we recognize payments related to these arrangements in the Consolidated Statement of Operations as lease costs on a straight-line basis over the lease term.
Warranty
Certain equipment products contain standard warranty terms typically cover one year from the date of delivery. We accrue for standard warranty costs based on historical trends in warranty charges. The accrual is reviewed regularly and periodically adjusted to reflect changes in warranty cost over the period.
Income taxes
We account for income taxes using an asset and liability method which generally requires recognition of deferred tax assets and liabilities for the expected future tax effects of events that have been included in the financial statements or tax returns. Under this method, deferred tax assets and liabilities are recognized for the future tax effects of differences between tax bases of assets and liabilities, and financial reporting amounts, based upon enacted tax laws and statutory rates applicable to the periods in which the differences are expected to affect taxable income. We evaluate the likelihood of realization of deferred tax assets and provide an allowance where, in management’s opinion, it is more likely than not that the asset will not be realized. Our policy for interest and penalties is to recognize interest and penalties as a component of the provision for income taxes in the Consolidated Statement of Operations.
We determine any uncertain tax positions based on a determination of whether and how much of a tax benefit taken in the Company’s tax filings or positions is more likely than not to be sustained upon examination by the relevant income tax authorities.
Judgment is applied in the determination of the financial statement recognition and measurement of tax positions taken or expected to be taken in a tax return. As of December 31, 2025, the Company has an unrecognized tax benefit of $1.2 million related to tax attributes being carried forward. The Company is generally subject to examination by U.S. federal and local income tax authorities for all tax years in which loss carryforward is available.
Sales Taxes Payable
The Company records sales tax collected from customers on a net basis and therefore excludes it from total revenues as defined in ASC Topic 606. Cash collected from customers is recorded in accrued expenses on the Company's Consolidated Balance Sheet and then remitted to the proper taxing authority. In addition, refer to Note 11: Commitments and contingencies for discussion regarding an estimated sales tax liability the Company recorded in relation to historical activity in certain states. As of December 31, 2025, 2024, and 2023 total interest expenses assessed on sales tax liabilities were $0.1 million, $0.4 million, and $0.4 million, respectively.
Advertising
Advertising costs are expensed as incurred and totaled $0.4 million, $0.9 million, and $0.8 million for the years ended December 31, 2025, 2024, and 2023, respectively.
Research and development
Research and development costs are expensed as incurred, including any identifiable assets purchased from others for a particular research and development project outside of a business combination with no alternative future use.
Stock-based compensation
We measure and record compensation expense using the applicable accounting guidance for share-based payments related to stock options, time-based restricted stock, market-based restricted stock awards, and performance-based restricted stock awards granted to our directors and employees. The fair value of stock options, including performance awards, without a market-based condition is determined by using the Black-Scholes option-pricing model. The fair value of restricted stock awards with a market condition is estimated at the date of grant using the Monte Carlo Simulation model. The Black-Scholes and Monte Carlo Simulation valuation models incorporate assumptions as to stock price volatility, the expected life of options or awards, a risk-free interest rate and dividend yield. The fair value of restricted stock, including performance awards, without a market condition is estimated using the current market price of our common stock on the date of grant.
We expense stock-based compensation for stock options and restricted stock awards over the requisite service period. For awards with only a service condition, we expense stock-based compensation using the straight-line method over the requisite service period for the entire award. For awards with a market condition, we expense the grant date fair value over the vesting period regardless of the value that the award recipients ultimately receive. For awards with performance conditions, we begin expensing the grant date fair value over the requisite vesting period only when the performance condition is deemed probable.
We have, from time to time, modified the terms of restricted stock awards awarded to employees. We account for the incremental increase in the fair value over the original award on the date of the modification as an expense for vested awards or over the remaining service (vesting) period for unvested awards. The incremental compensation cost is the excess of the fair value of the modified award on the date of modification over the fair value of the original award immediately before the modification.
Business combinations
In accordance with ASC Topic 805, Business Combinations ("ASC 805"), the Company accounts for an acquisition as a business combination if the assets acquired and liabilities assumed in the transaction constitute a business. Such acquisitions are accounted for using the acquisition method, whereby the Company recognizes the identifiable tangible and intangible assets acquired and liabilities assumed, and any non-controlling interest and acquisition date fair value of any previously held equity interest in the acquired business is measured at the acquisition date fair values. Where the set of assets acquired and liabilities assumed do not constitute a business, it is accounted for as an asset acquisition where the individual assets and liabilities are recorded at their respective relative fair values corresponding to the consideration transferred. Should the Company have previously held equity interest in the acquiree of an asset acquisition, the Company has elected to include the fair value of the previously held equity interest within the total cost of the asset acquisition.
Goodwill
Goodwill represents the excess of the purchase price over the net amount of identifiable assets acquired and liabilities assumed in a business combination measured at fair value. Goodwill is not amortized but is tested for impairment at least annually. The Company reviews goodwill for impairment annually in the fourth quarter and whenever events or changes in circumstances indicate that the fair value of a reporting unit may be less than its carrying amount (a triggering event). The Company first assesses qualitative factors to determine whether it is more likely than not that the fair value of its reporting unit is less than its carrying amount as a basis for determining whether it is necessary to perform the quantitative goodwill impairment test described in FASB ASC Topic 350, Intangibles – Goodwill and Other ("ASC 350"). The more likely than not threshold is defined as having a likelihood of more than 50 percent. If, after assessing the totality of events or circumstances, the Company determines that it is not more likely than not that the fair value of a reporting unit is less than its carrying amount, then performing the quantitative goodwill impairment test is unnecessary and goodwill is considered to be unimpaired. However, if based on the qualitative assessment the Company concludes that it is more likely than not that the fair value of a reporting unit is less than its carrying amount, the Company will proceed with performing the quantitative goodwill impairment test. In performing the quantitative goodwill impairment test, the Company determines the fair value of its reporting unit and compares it to its carrying value. If the fair value of the reporting unit exceeds the carrying value of the net assets assigned to that unit, goodwill is not impaired. If the carrying value of the reporting unit exceeds its fair value, the Company records an impairment loss equal to the difference. The Company operates as one reporting unit as of the goodwill impairment measurement date in the fourth quarter of 2025. As of the testing date and the period after that date through the issuance date of our financial statements, the Company has observed no indicators of potential goodwill impairment at any point during the period based on its required assessment.
Intangible assets
Intangible assets with a definite life are amortized over their estimated useful lives using the straight-line method and the amortization expense is recorded within intangible asset amortization in the Consolidated Statements of Operations. If the estimate of a definite-lived intangible asset’s remaining useful life is changed, the remaining carrying amount of the intangible asset is amortized prospectively over the revised remaining useful life. Definite-lived intangible assets and their related estimated useful lives are reviewed at least annually to determine if any adverse conditions exist that would indicate the carrying value of these assets may not be recoverable.
Indefinite-lived intangibles are carried at the initially recorded fair value less any recognized impairment. Indefinite-lived intangibles are tested annually for impairment. Impairment assessments are conducted more frequently if certain conditions
exist, including a change in the competitive landscape, any internal decisions to pursue new or different technology strategies, a loss of a significant customer, or a significant change in the marketplace, including changes in the prices paid for the Company’s products or changes in the size of the market for the Company’s products. If impairment indicators are present, the Company determines whether the underlying intangible asset is recoverable through estimated future undiscounted cash flows. If the asset is not found to be recoverable, it is written down to the estimated fair value of the asset based on the sum of the future discounted cash flows expected to result from the use and disposition of the asset.
Recent accounting pronouncements
Recently adopted accounting pronouncements
In December 2023, FASB issued ASU No. 2023-09, Improvements to Income Tax Disclosures (“ASU 2023-09”), which requires additional disaggregated information about a reporting entity’s effective tax rate reconciliation as well as information increasing transparency of income taxes paid. The standard is intended to benefit investors by providing more detailed income tax disclosures that would be useful in making capital allocation decisions. The Company adopted this standard effective January 1, 2025 using a prospective approach. Refer to Note 14: Income taxes for further information.
Recently issued accounting pronouncements not yet adopted
On December 8, 2025, FASB issued ASU 2025‑11, Interim Reporting (Topic 270): Narrow‑Scope Improvements ("ASU 2025-11"), which clarifies and enhances guidance under ASC 270 on interim financial reporting by (i) clarifying the scope of ASC 270 such that it now explicitly applies only to entities that issue complete interim financial statements and related notes under U.S. GAAP, (ii) establishing clear guidance on the form of interim statements and notes, incorporating a comprehensive list of required interim disclosures drawn from across the ASC, and (iii) introducing a requirement to disclose material events and changes occurring after the end of the last annual period that could impact interim results. This guidance will be effective for interim periods beginning in 2028, though early adoption is permitted. The Company is currently evaluating the impact of this accounting standard on its Consolidated Financial Statements and related disclosures.
On December 4, 2025, FASB issued ASU 2025-10, Accounting for Government Grants Received by Business Entities ("ASU 2025-10"), which establishes guidance on the recognition, measurement, and presentation of government grants received by business entities. ASU 2025-10 is effective for annual periods beginning after December 15, 2028. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On November 25, 2025, FASB issued ASU 2025‑09, Derivatives and Hedging (Topic 815): Hedge Accounting Improvements ("ASU 2025-09"), which amends ASC 815 to align hedge accounting more closely with an entity’s economic risk management practices. Key amendments include (i) to allow designating a variable price component of a nonfinancial forecasted purchase or sale as the hedged risk, (ii) to allow grouping individual forecasted transactions with similar (not identical) risk exposures, (iii) a new model for hedging forecasted interest on variable-rate debt, enabling changes in index or tenor without designation, subject to simplifying assumptions, and (iv) additional clarifications related to hedge accounting of nonfinancial components, net written options, and dual-hedge strategies. This guidance is effective beginning in 2027, though early adoption is permitted. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On November 12, 2025, FASB issued ASU 2025‑08, Financial Instruments - Credit Losses (Topic 326): Purchased Loans ("ASU 2025-08"), which expands the scope of the “gross‑up” method, formerly applicable only to purchased credit‑deteriorated ("PCD") assets, to include acquired non‑PCD loans that meet certain criteria, now referred to as “purchased seasoned loans” (PSLs). Under this model, an allowance for expected credit losses is recognized at acquisition, offsetting the loan’s amortized cost basis, thereby eliminating the day-one credit‑loss expense previously required for non‑PCD assets. PSLs are defined as non‑PCD loans acquired either (i) through a business combination, or (ii) purchased more than 90 days after origination when the acquirer was not involved in origination. This guidance is effective on a prospective basis for loans acquired on or after the adoption date, for interim and annual reporting periods beginning in 2027, though early adoption is permitted. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On September 29, 2025, FASB issued ASU 2025-07, Derivatives Scope Refinements and Scope Clarification for Share-Based Noncash Consideration from a Customer in a Revenue Contract ("ASU 2025-07"), which excludes from derivative
accounting non-exchange-traded contracts with underlyings based on operations or activities specific to one of the parties to the contract. This guidance is effective for fiscal years and interim periods beginning after December 15, 2026, with early adoption permitted. These requirements may be applied prospectively or on a modified retrospective basis through a cumulative-effect adjustment to the opening balance of retained earnings. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On September 18, 2025, the FASB issued ASU 2025-06, Targeted Improvements to the Accounting for Internal-Use Software ("ASU 2025-06"), which modernizes the accounting guidance for the costs to develop software for internal use. The standard applies to costs incurred to develop or obtain software for internal use. ASU 2025-06 amends the existing standard that refers to various stages of a software development project to align better with current software development methods. Under the new standard, entities will commence capitalizing eligible costs when (i) management has authorized and committed to funding the software project, and (ii) it is probable that the project will be completed and the software will be used to perform the function intended. ASU 2025-06 guidance is effective for annual periods beginning after December 15, 2027. The guidance can be applied on a prospective basis, a modified basis for in-process projects, or on a retrospective basis. The Company is currently evaluating the impact of this accounting standard on its Consolidated Financial Statements and related disclosures.
On July 30, 2025, the FASB issued ASU 2025-05, Measurement of Credit Losses for Accounts Receivable and Contract Assets ("ASU 2025-05"), which provides a practical expedient that assumes current conditions as of the balance sheet date remain unchanged when developing forecasts for estimating expected credit losses. Under ASU 2025-05, an entity is required to disclose that it has elected to use the practical expedient and the election should be applied prospectively. ASU 2025-05 is effective for fiscal years, and interim periods within those years, beginning after December 15, 2025, with early adoption permitted. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On May 15, 2025, the FASB issued ASU 2025-04, Clarifications to Share-Based Consideration Payable to a Customer ("ASU 2025-04"), which clarifies the guidance on the accounting for share-based payment awards that are granted by an entity as consideration payable to its customer, with the intent to reduce diversity in practice and improve existing guidance by revising the definition of a “performance condition” and eliminating a forfeiture policy election for service conditions associated with share-based consideration payable to a customer. ASU 2025-04 also clarifies that the guidance in Topic 606 on the variable consideration constraint does not apply to share-based consideration payable to a customer "regardless of whether an award’s grant date has occurred." ASU 2025-04 is effective for fiscal years beginning after December 15, 2026 with updates to be applied on a retrospective or modified retrospective basis. Early adoption is permitted. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
On May 12, 2025, the FASB issued ASU 2025-03, Determining the Accounting Acquirer in the Acquisition of a Variable Interest Entity ("ASU 2025-03"), which revises the guidance in ASC 805 on identifying the accounting acquirer in a business combination in which the legal acquiree is a variable interest entity ("VIE"). ASU 2025-03 is intended to improve comparability between business combinations that involve VIEs and those that do not. Under ASU 2025-03, a reporting entity involved in a business combination effected primarily by the exchange of equity interests must consider the factors in ASC 805-10-55-12 through 55-15 to determine which entity is the accounting acquirer regardless of whether the legal acquiree is a VIE. More specifically, when considering those factors, the reporting entity can determine that a transaction in which the legal acquiree is a VIE represents a reverse acquisition (in which the legal acquirer is identified as the acquiree for accounting purposes). As a result, comparability is increased with business combinations in which the legal acquiree is a VIE. ASU 2025-03 is effective for fiscal years beginning after December 15, 2026, including interim periods within those fiscal years. Early adoption is permitted. The amendments in ASU 2025-03 must be applied prospectively to any business combination that occurs after the initial adoption date. The Company does not expect the application of this standard will have a material impact on its Consolidated Financial Statements and related disclosures.
In November 4, 2024, FASB issued ASU 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures: Disaggregation of Income Statement Expenses, which requires disclosure of disaggregated information about specific categories underlying certain income statement expense line items in the footnotes to the financial statements for both annual and interim periods. This ASU is effective for fiscal years beginning after December 15, 2026, and interim reporting periods beginning after December 15, 2027. Early adoption is permitted. The Company is currently evaluating the effects adoption of this guidance will have on the consolidated financial statements.
2. Acquisition
In November 2020, the Company invested approximately $1.0 million in Class E Preferred Shares in PanTHERA, representing an approximately 10% ownership interest. In conjunction with this investment, the Company executed the PanTHERA Development and License Agreement under which the Company made milestone development payments in exchange for exclusive, perpetual, and worldwide marketing and distribution rights to the technology for use in CGT applications.
On April 4, 2025, the Company acquired the remaining 90% of the outstanding shares of capital stock of PanTHERA from the PanTHERA Sellers. Through the PanTHERA Transaction, the Company obtained ownership of PanTHERA’s patented Ice Recrystallization Inhibitor ("IRI") GEN 2 cryopreservation technology that is expected to ultimately enhance the Company's core capabilities in biopreservation and within the CGT market upon achievement of commercial viability. This technology was valued to represent approximately 95% of the gross assets acquired in the PanTHERA Transaction. In accordance with ASC 805-50, due to the fair value of the technology representing substantially all of the gross assets acquired, the Company accounted for this transaction as an asset acquisition.
The IRI GEN 2 cryopreservation technology was under development as of the date of acquisition. It was therefore considered in-process research and development ("IPR&D") as of the date of acquisition and valued at $15.5 million. The Company analyzed the quantitative and qualitative factors relevant to the acquisition of the IPR&D IRI GEN 2 cryopreservation technology and determined that the asset should be immediately expensed in accordance with ASC 730-10 due to the technology not meeting alternative future use criteria. The expense was reported within the IPR&D expense line in the Consolidated Statements of Operations.
Pursuant to the PanTHERA Purchase Agreement, the PanTHERA Sellers are eligible to receive up to $7.2 million in cash or equivalent shares of the Company's common stock (as elected by the PanTHERA Sellers) over a three-year earnout period upon the achievement of certain revenue targets based on the Company's earnings derived from the acquired IRI GEN 2 cryopreservation technology in addition to the achievement of an operational milestone within the first year of the earnout period. The PanTHERA Purchase Agreement also contains an embedded change in control protective provision. As of December 31, 2025, the Company's management determined that the probability of the PanTHERA Sellers achieving the outlined revenue and operational targets to be remote. As a result, no earnout consideration for any period has been recognized.
The Company recognized the following in closing costs as of the closing date of the PanTHERA Transaction:
| | | | | |
| (In thousands) | |
| Cash paid at close of acquisition | $ | 9,545 | |
Indemnity escrow amount(1) | 350 | |
Transaction expenses(2) | 609 | |
Carrying value of equity interest(3) | 995 | |
Cash in lieu of stock payment(4) | 995 | |
Stock issued to PanTHERA Sellers(5) | 4,455 | |
| Gross closing cost consideration | 16,949 | |
Less: extinguishment of pre-existing liability(6) | (150) | |
| Total closing cost consideration | $ | 16,799 | |
(1) Represents the amount transferred to the escrow agent as of the closing date of the PanTHERA Transaction to cover any losses arising from a breach of representations as agreed upon within the PanTHERA Transaction. This amount will be held in escrow for 18 months following the closing date.
(2) Represents the costs incurred by the Company ($0.5 million) and on behalf of PanTHERA Sellers ($0.1 million) in connection with the PanTHERA Transaction, including fees to be paid to attorneys and other external parties.
(3) Represents the original investment amount by the Company in PanTHERA in November 2020. This was added to the overall consideration paid to determine the costs to allocate to assets acquired in the PanTHERA Transaction.
(4) Represents portion of common stock payout to PanTHERA Sellers that was elected to be distributed in cash rather than stock compensation.
(5) Represents market value of 213,360 shares of the Company's common stock issued to the PanTHERA Sellers as of the closing date of the PanTHERA Transaction in accordance with its terms.
(6) Represents pre-existing liability of the Company from milestone development payments under the PanTHERA Development and License Agreement.
3. Discontinued operations
As announced in the second quarter of 2023, the Company, management, and our board of directors (the "Board"), had determined that divesting Global Cooling and CBS (the "Freezer Business") would allow for the Company to optimize its product portfolio by focusing on its recurring higher margin revenue streams. Additionally, in November of 2024, the Company, management, and the Board determined the sale of SciSafe would further optimize the Company's product portfolio toward its proprietary high margin cell processing and other bioproduction products. In October of 2025, the Company, management, and the Board determined the sale of SAVSU would also fulfill its overall strategy to streamline its product portfolio to its cell processing products. The Company completed the sale of Global Cooling during the second quarter of 2024, the sales of CBS and SciSafe during the fourth quarter of 2024, and the sale of SAVSU during the fourth quarter of 2025. Accordingly, the results of these businesses are reported in the Loss from discontinued operations line in the Consolidated Statements of Operations. These changes have been applied to all periods presented.
Divestiture of SAVSU
On October 6, 2025, the Company entered into a Limited Liability Company Membership Interest Purchase Agreement (the “SAVSU Purchase Agreement”), by and between the Company and Peli BioThermal LLC, a Delaware limited liability company (“SAVSU Buyer”), for the sale by the Company of all of the issued and outstanding limited liability company membership interests (the “SAVSU Interests”) of SAVSU Cleo Technologies, LLC, a Delaware limited liability company ("SAVSU"), to SAVSU Buyer. The Company analyzed the quantitative and qualitative factors relevant to the sale of SAVSU and determined that the conditions for discontinued operations presentation were met during the fourth quarter of 2025.
The Company recognized a gain on disposal of SAVSU, calculated as follows:
| | | | | |
| (In thousands) | |
| Cash proceeds received from Buyer | $ | 23,276 | |
| Indemnity holdback | 2,500 | |
Net price adjustment(1) | 219 | |
| Costs to sell | (1,509) | |
Retention bonus paid by Company(2) | (541) | |
| Total proceeds | 23,945 | |
| Less: SAVSU carrying basis as of October 6, 2025 | 13,621 | |
| Net gain on disposal | $ | 10,324 | |
(1) As defined within the SAVSU Purchase Agreement, the final purchase price was subject to working capital adjustments upon the close of the disposal.
(2) As defined within the SAVSU Purchase Agreement, the Company agreed to provide a cash bonus to the divested employees with the condition that such employees remain with SAVSU for 90 days subsequent to the closing of the sale.
The Company recognized $1.3 million in stock compensation expense for the acceleration of unvested shares of all the Company's former employees that remained with SAVSU upon the closing of this transaction.
In accordance with ASC 350, upon the disposal of SAVSU, the Company assessed the goodwill to be allocated to the disposal group. The goodwill allocated to SAVSU was based on the relative fair value of SAVSU to the fair value of the Company as SAVSU was fully integrated into the Company's one reportable segment. The fair value of SAVSU was determined based on the enterprise value per the SAVSU Purchase Agreement. The fair value of the Company was determined by calculating the Company's market capitalization as of the disposal date plus any invested capital remaining of the Company, which included outstanding debt and financing lease liabilities, modified by an estimated market acquisition premium. Based on the calculation performed, the Company determined $3.7 million of goodwill was to be
allocated to SAVSU upon its disposal. The allocated goodwill was included in the carrying basis of SAVSU presented in the above table.
In addition, upon the closing of this transaction, the Company and SAVSU Buyer entered into a transition services agreement ("SAVSU TSA"), pursuant to which the Company will provide certain transition services, including payroll processing, bookkeeping and tax administration services, and information technology maintenance, among other administrative services, to SAVSU for 120 days. The SAVSU Purchase Agreement contains customary representations, warranties, covenants and indemnities of the parties thereto, including customary covenants that prevent the Company from competing with SAVSU, soliciting its employees or interfering with its business relationships for 5 years after the closing of the SAVSU Transaction. The SAVSU TSA has since expired pursuant to its terms on the stated expiration date. The Company has no other significant continuing involvement with SAVSU upon the expiration of its SAVSU TSA and related covenants.
Divestiture of Custom Biogenics
On November 14, 2024, the Company entered into a Stock Purchase Agreement (the "CBS Purchase Agreement"), by and among the Company, Standex International Corporation, a Delaware corporation, ("CBS Buyer") and Arctic Solutions, Inc., a Delaware corporation and a wholly owned subsidiary of the Company (doing business as Custom Biogenic Systems, or ("CBS"), for the sale by the Company of all of the issued and outstanding shares of common stock of CBS (the "CBS Divestiture"). The Company analyzed the quantitative and qualitative factors relevant to the sale of CBS and determined that the conditions for discontinued operations presentation were met during the fourth quarter of 2024.
During the year ended December 31, 2024, the Company recognized a loss on disposal of CBS of $3.4 million and $2.0 million in stock compensation expense for the acceleration of unvested shares of all the Company's former employees that remained with CBS upon the closing of this transaction.
In addition, upon the closing of this transaction, the Company and CBS entered into a transition service agreement (the "CBS TSA"), pursuant to which the Company is subject to customary covenants that prevent the Company from competing with CBS, soliciting its employees or interfering with its business relationships for two years after the closing of the CBS Divestiture. The Company has no other significant continuing involvement with CBS upon the expiration of its CBS TSA and related covenants.
Divestiture of SciSafe, Inc.
On November 12, 2024, the Company entered into a Stock Purchase Agreement (the "SciSafe Purchase Agreement"), by and among the Company, Subzero Purchaser Corp., a Delaware corporation ("SciSafe Buyer"), SciSafe, Inc., a Delaware corporation and an indirect, wholly owned subsidiary of the Company ("SciSafe Seller"), and SciSafe, Inc., a New Jersey corporation and an indirect wholly owned subsidiary of the Company ("SciSafe"), for the sale by Sci Safe Seller of all of the issued and outstanding shares of common stock of SciSafe to SciSafe Buyer (the "SciSafe Divestiture"). The Company analyzed the quantitative and qualitative factors relevant to the sale of SciSafe and determined that the conditions for discontinued operations presentation were met during the fourth quarter of 2024.
During the year ended December 31, 2024, the Company recognized a gain on disposal of SciSafe of $28.1 million and $4.0 million of stock-based compensation expense related to the acceleration of unvested shares for former employees who remained with SciSafe upon the closing of this transaction.
In addition, upon the closing of the SciSafe Divestiture, the Company and SciSafe entered into a transition services agreement ("SciSafe TSA"), pursuant to which the Company is subject to customary covenants that prevent the Company from competing with SciSafe, soliciting its employees or interfering with its business relationships for five years after the Closing Date. The Company has no other significant continuing involvement with SciSafe upon the expiration of its SciSafe TSA and related covenants.
In connection with the disposal of SciSafe, the Company remains liable and responsible for the full performance and observance of all of the provisions, covenants, and conditions in one of SciSafe's operating leases. In the case of a breach or violation of any provision of the lease by the SciSafe Buyer, the Company is deemed to be and shall constitute a default of the lease provisions. Simultaneously, the Company received indemnification pursuant any obligation owed by the Company under this operating lease. This indicates the Company undertakes the obligation to stand ready to perform over the term of the guarantee in the event of the specified triggering events noted above, or conditions, such as breach or
default, occur. However, the non-contingent aspect of the guarantee enables the Company to recover any losses from the SciSafe Buyer. As of December 31, 2025, the fair value of this guarantee is zero. The outstanding minimum lease payments equal approximately $2.2 million and the lease terminates in 2031.
Divestiture of Global Cooling, Inc.
On April 17, 2024, the Company sold all of the issued and outstanding shares of common stock (the "Global Cooling Divestiture") of Global Cooling, Inc., a Delaware corporation and wholly owned subsidiary of the Company ("Global Cooling"), to GCI Holdings, an Ohio limited liability company ("GCI Holdings") pursuant to a Stock Purchase Agreement, dated April 17, 2024, (the "Global Cooling Purchase Agreement"), by and between the Company and GCI Holdings. The Company analyzed the quantitative and qualitative factors relevant to the sale of Global Cooling and determined that the conditions for discontinued operations presentation were met during the second quarter of 2024.
During the year ended December 31, 2024, the Company recognized a loss on disposal of Global Cooling of $8.9 million, and $3.2 million of stock-based compensation expense related to the acceleration of unvested shares for employees impacted by the reduction in force, as well as former employees who remained with Global Cooling upon closing of the transaction.
As outlined in the Global Cooling Purchase Agreement, the Company is required to indemnify Global Cooling for certain preexisting legal contingencies. Prior to the Global Cooling Divestiture, two lawsuits were filed by previous customers related to Global Cooling's commercial freezer products seeking indemnification. The details of each case are described below.
Other than the Company's requirement to indemnify Global Cooling for certain preexisting legal contingencies, the Company has no other significant continuing involvement with Global Cooling.
Global Cooling legal contingencies
As of the year ended December 31, 2025, the Company recorded a loss contingency for a $1.4 million claim in relation to losses a previous customer claims to have incurred. The loss contingency was recorded under the discontinued operations of Global Cooling as outlined in the Global Cooling Purchase Agreement. During the third quarter of 2025, it became probable this loss would be settled within the next fiscal year, and the Company accrued for the loss contingency upon the determination of probability. During the fourth quarter of 2025, the claim was fully settled for $0.9 million, which was fully covered by the Company's insurance policy aside from the Company's insurance deductible. Legal expenses incurred on the claim were immaterial.
As of the year ended December 31, 2024, the Company recorded a loss contingency for a $4.0 million claim in relation to losses a previous customer claims to have incurred. The loss contingency was recorded under the discontinued operations of Global Cooling as outlined in the Global Cooling Purchase Agreement. During the fourth quarter of 2024, it became probable the loss would be settled within the next fiscal year, and the Company accrued for the loss contingency upon the determination of probability. There had been no changes in the status of this claim as of the year ended December 31, 2025. During the first quarter of 2026, the claim was fully settled for $2.5 million, which was fully covered by the Company's insurance policy aside from the Company's insurance deductible. Legal expenses incurred on the claim were immaterial.
Summarized financial data of discontinued operations
The tables below summarize financial data of discontinued operations as of the year ended December 31, 2024 and for the years ended December 31, 2025, 2024, and 2023. Interest expenses directly associated with the debt of a disposed entity are reported in discontinued operations below.
The table below summarizes the major classes of assets and liabilities of discontinued operations, which are summarized separately in the Consolidated Balance Sheets:
| | | | | | | | | | | | | |
| | | | | | | | | December 31, 2024 |
| (In thousands) | | | | | | | | | SAVSU |
| Cash and cash equivalents | | | | | | | | | $ | 3,848 | |
| Accounts receivable, net | | | | | | | | | 692 | |
| Prepaid expenses and other current assets | | | | | | | | | 44 | |
| Total current assets, discontinued operations | | | | | | | | | 4,584 | |
| | | | | | | | | |
| Assets held for rent, net | | | | | | | | | 6,103 | |
| Property and equipment, net | | | | | | | | | 111 | |
| Operating lease right-of-use assets, net | | | | | | | | | 121 | |
| Long-term deposits and other assets | | | | | | | | | 5 | |
| Intangible assets, net | | | | | | | | | 3,994 | |
| Goodwill | | | | | | | | | 3,717 | |
| Total assets, discontinued operations | | | | | | | | | 18,635 | |
| | | | | | | | | |
| Accounts payable | | | | | | | | | $ | 213 | |
| Accrued expenses and other current liabilities | | | | | | | | | 328 | |
| Sales taxes payable | | | | | | | | | 23 | |
| Lease liabilities, operating, current portion | | | | | | | | | 52 | |
| Total current liabilities, discontinued operations | | | | | | | | | 616 | |
| | | | | | | | | |
| Lease liabilities, operating, long-term | | | | | | | | | 78 | |
| Total liabilities, discontinued operations | | | | | | | | | $ | 694 | |
All divested entities had no remaining balances as of December 31, 2025. The divested entities Global Cooling, SciSafe, and CBS did not have any remaining balances as of December 31, 2024.
The key components of income (loss) from discontinued operations were as follows:
| | | | | | | | | | | | | |
| | | | | | | | | December 31, 2025 |
| (In thousands) | | | | | | | | | SAVSU |
| Revenue | | | | | | | | | $ | 5,977 | |
| Cost of revenue | | | | | | | | | 4,235 | |
| Gross profit | | | | | | | | | 1,742 | |
| Operating expenses | | | | | | | | | (4,418) | |
| Intangible asset amortization | | | | | | | | | (118) | |
| Other income, net | | | | | | | | | 5 | |
| Gain on disposal | | | | | | | | | 10,324 | |
| Income before income taxes | | | | | | | | | 7,535 | |
| Income tax expense | | | | | | | | | — | |
| Income from discontinued operations, net of income taxes | | | | | | | | | $ | 7,535 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2024 |
| (In thousands) | Global Cooling | | SciSafe | | CBS | | SAVSU | | Total |
| Revenue | $ | 7,157 | | | $ | 18,440 | | | $ | 12,141 | | | $ | 7,607 | | | $ | 45,345 | |
| Cost of revenue | 8,389 | | | 16,357 | | | 10,600 | | | 6,346 | | | 41,692 | |
| Gross profit | (1,232) | | | 2,083 | | | 1,541 | | | 1,261 | | | 3,653 | |
| Operating expenses | (9,418) | | | (11,467) | | | (4,967) | | | (3,753) | | | (29,605) | |
| Intangible asset amortization | — | | | (764) | | | — | | | (160) | | | (924) | |
| Other (expense) income, net | (25) | | | (183) | | | (110) | | | 50 | | | (268) | |
| (Loss) gain on disposal | (8,897) | | | 28,139 | | | (3,365) | | | — | | | 15,877 | |
| (Loss) income before income taxes | (19,572) | | | 17,808 | | | (6,901) | | | (2,602) | | | (11,267) | |
| Income tax expense | 10 | | | 122 | | | — | | | — | | | 132 | |
| (Loss) income from discontinued operations, net of income taxes | $ | (19,582) | | | $ | 17,686 | | | $ | (6,901) | | | $ | (2,602) | | | $ | (11,399) | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2023 |
| (In thousands) | Global Cooling | | SciSafe | | CBS | | SAVSU | | Total |
| Revenue | $ | 35,826 | | | $ | 18,014 | | | $ | 13,576 | | | $ | 7,839 | | | $ | 75,255 | |
| Cost of revenue | 36,682 | | | 17,283 | | | 12,632 | | | 5,553 | | | 72,150 | |
| Gross profit | (856) | | | 731 | | | 944 | | | 2,286 | | | 3,105 | |
| Operating expenses | (20,162) | | | (5,316) | | | (7,153) | | | (8,362) | | | (40,993) | |
| Intangible asset impairment charges | (7,175) | | | — | | | (8,310) | | | — | | | (15,485) | |
| Intangible asset amortization | (131) | | | (907) | | | (623) | | | (160) | | | (1,821) | |
| Other expense, net | (90) | | | (100) | | | (214) | | | (14) | | | (418) | |
| Loss before income taxes | (28,414) | | | (5,592) | | | (15,356) | | | (6,250) | | | (55,612) | |
| Income tax expense | 4 | | | 180 | | | 9 | | | 40 | | | 233 | |
| Loss from discontinued operations, net of income taxes | $ | (28,418) | | | $ | (5,772) | | | $ | (15,365) | | | $ | (6,290) | | | $ | (55,845) | |
Below is a summary of incurred depreciation, amortization, interest expenses, capital expenditures, and other noncash related costs for discontinued operations:
| | | | | | | | | | | | | |
| | | | | | | | | December 31, 2025 |
| (In thousands) | | | | | | | | | SAVSU |
| Depreciation | | | | | | | | | $ | 1,404 | |
| Amortization | | | | | | | | | 1,127 | |
| Stock-based compensation | | | | | | | | | 1,701 | |
| Interest expense, net | | | | | | | | | 2 | |
| Capital expenditures | | | | | | | | | 946 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2024 |
| (In thousands) | Global Cooling | | SciSafe | | CBS | | SAVSU | | Total |
| Depreciation | $ | — | | | $ | 2,402 | | | $ | 4 | | | $ | 2,112 | | | $ | 4,518 | |
| Amortization | — | | | 764 | | | — | | | 1,506 | | | 2,270 | |
| Stock-based compensation | 4,191 | | | 6,410 | | | 3,790 | | | 1,231 | | | 15,622 | |
| Interest (expense) income, net | (42) | | | (50) | | | (114) | | | 47 | | | (159) | |
| Capital expenditures | — | | | 2,200 | | | 720 | | | 2,129 | | | 5,049 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2023 |
| (In thousands) | Global Cooling | | SciSafe | | CBS | | SAVSU | | Total |
| Depreciation | $ | 397 | | | $ | 2,636 | | | $ | 471 | | | $ | 2,947 | | | $ | 6,451 | |
| Amortization | 131 | | | 907 | | | 623 | | | 1,506 | | | 3,167 | |
| Stock-based compensation | 4,734 | | | 2,759 | | | 2,503 | | | 3,346 | | | 13,342 | |
| Interest expense, net | 131 | | | 13 | | | 219 | | | 44 | | | 407 | |
| Capital expenditures | — | | | 4,659 | | | 750 | | | 5,353 | | | 10,762 | |
4. Fair value measurement
The following tables set forth the Company’s financial assets and liabilities measured at fair value on a recurring basis as of December 31, 2025 and 2024, based on the three-tier fair value hierarchy:
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2025 |
| (In thousands) | Level 1 | | Level 2 | | Level 3 | | Total |
| Assets: | | | | | | | |
| Cash equivalents: | | | | | | | |
| Money market accounts | $ | 31,241 | | | $ | — | | | $ | — | | | $ | 31,241 | |
| Available-for-sale securities: | | | | | | | |
| U.S. government securities | 20,749 | | | — | | | — | | | 20,749 | |
| Corporate debt securities | 6,700 | | | 45,180 | | | — | | | 51,880 | |
| Other debt securities | — | | | 14,510 | | | — | | | 14,510 | |
| Convertible promissory note | — | | | — | | | 2,983 | | | 2,983 | |
| Total | $ | 58,690 | | | $ | 59,690 | | | $ | 2,983 | | | $ | 121,363 | |
| | | | | | | | | | | | | | | | | | | |
| December 31, 2024 |
| (In thousands) | Level 1 | | Level 2 | | | | Total |
| Assets: | | | | | | | |
| Cash equivalents: | | | | | | | |
| Money market accounts | $ | 89,119 | | | $ | — | | | | | $ | 89,119 | |
| Available-for-sale securities: | | | | | | | |
| U.S. government securities | 1,494 | | | — | | | | | 1,494 | |
| Corporate debt securities | 398 | | | 8,602 | | | | | 9,000 | |
| Other debt securities | — | | | 3,332 | | | | | 3,332 | |
| Total | $ | 91,011 | | | $ | 11,934 | | | | | $ | 102,945 | |
In July 2025, the Company invested $2.0 million cash in a convertible promissory note issued by an unrelated third-party company. The convertible promissory note accrues interest at an annual rate of 10%. The principal and accrued paid in
kind ("PIK") interest are payable at maturity on October 1, 2028. The carrying value of the convertible promissory note is approximately $3.0 million as of December 31, 2025. There have been no transfers of assets or liabilities between the fair value measurement levels. We had no financial assets that utilize Level 3 inputs of measurement as of December 31, 2024.
5. Investments
Available-for-sale securities
The Company’s portfolio of available-for-sale marketable securities consists of the following:
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2025 |
| Amortized
Cost | | Gross unrealized | | Estimated
Fair Value |
| (In thousands) | | Gains | | Losses | |
| Available-for-sale securities, current portion | | | | | | | |
| U.S. government securities | $ | 9,946 | | | $ | 33 | | | $ | — | | | $ | 9,979 | |
| Corporate debt securities | 34,783 | | | 41 | | | — | | | 34,824 | |
| Other debt securities | 11,066 | | | 20 | | | — | | | 11,086 | |
| Total short-term | 55,795 | | | 94 | | | — | | | 55,889 | |
| | | | | | | |
| Available-for-sale securities, long-term | | | | | | | |
| U.S. government securities | 10,756 | | | 14 | | | — | | | 10,770 | |
| Corporate debt securities | 17,038 | | | 23 | | | (5) | | | 17,056 | |
| Other debt securities | 3,418 | | | 6 | | | — | | | 3,424 | |
| Total long-term | 31,212 | | | 43 | | | (5) | | | 31,250 | |
| | | | | | | |
| Total available-for-sale securities | $ | 87,007 | | | $ | 137 | | | $ | (5) | | | $ | 87,139 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2024 |
| Amortized
Cost | | Gross unrealized | | Estimated
Fair Value |
| (In thousands) | | Gains | | Losses | |
| Available-for-sale securities, current portion | | | | | | | |
| U.S. government securities | $ | 1,493 | | | $ | 1 | | | $ | — | | | $ | 1,494 | |
| Corporate debt securities | 5,775 | | | 14 | | | (1) | | | 5,788 | |
| Other debt securities | 1,912 | | | 4 | | | — | | | 1,916 | |
| Total short-term | 9,180 | | | 19 | | | (1) | | | 9,198 | |
| | | | | | | |
| Available-for-sale securities, long-term | | | | | | | |
| Corporate debt securities | 3,210 | | | 4 | | | (2) | | | 3,212 | |
| Other debt securities | 1,412 | | | 5 | | | (1) | | | 1,416 | |
| Total long-term | 4,622 | | | 9 | | | (3) | | | 4,628 | |
| | | | | | | |
| Total available-for-sale securities | $ | 13,802 | | | $ | 28 | | | $ | (4) | | | $ | 13,826 | |
The following table summarizes the contractual maturities of the short-term and long-term available-for-sale investments as of December 31, 2025:
| | | | | | | | | | | |
| (In thousands) | Amortized
Cost | | Estimated
Fair Value |
| Due in one year or less | $ | 55,795 | | | $ | 55,889 | |
| Due after one year through five years | 31,212 | | | 31,250 | |
| Total | $ | 87,007 | | | $ | 87,139 | |
The following tables present information about the available-for-sale investments that had been in a continuous unrealized loss position for less than 12 months:
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2025 |
| Less than 12 months | | Total |
| (In thousands) | Fair Value | | Unrealized Losses | | Fair Value | | Unrealized Losses |
| Corporate debt securities | $ | 7,519 | | | $ | (5) | | | $ | 7,519 | | | $ | (5) | |
| | | | | | | |
| Total | $ | 7,519 | | | $ | (5) | | | $ | 7,519 | | | $ | (5) | |
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2024 |
| Less than 12 months | | Total |
| (In thousands) | Fair Value | | Unrealized Losses | | Fair Value | | Unrealized Losses |
| Corporate debt securities | $ | 2,091 | | | $ | (3) | | | $ | 2,091 | | | $ | (3) | |
| Other debt securities | 1,030 | | | (1) | | | 1,030 | | | (1) | |
| Total | $ | 3,121 | | | $ | (4) | | | $ | 3,121 | | | $ | (4) | |
As of December 31, 2025 and 2024, all available-for-sale securities investments presented above with unrealized losses have been in an unrealized loss position for less than 12 months.
As of December 31, 2025 and 2024, none of our available-for-sale marketable securities exhibited risk of credit loss and therefore no allowance for credit losses was recorded.
6. Inventories
Inventories consist of the following as of December 31, 2025 and 2024:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Raw materials | $ | 10,561 | | | $ | 11,768 | |
| Work in progress | 6,371 | | | 4,082 | |
| Finished goods | 13,223 | | | 13,163 | |
| Total | $ | 30,155 | | | $ | 29,013 | |
7. Leases
We have various operating lease agreements for office space, warehouses, manufacturing, production locations, and other equipment. Our real estate leases had original lease terms of three to eleven years and remaining lease terms of two to six years. We exclude options that are not reasonably certain to be exercised from our lease terms, ranging from one to five years. Our lease payments consist primarily of fixed rental payments for the right to use the underlying leased assets over the lease terms, with all other lease payments consisting of variable lease costs. For certain leases, we receive incentives from our landlords, such as rent abatements, which effectively reduce the total lease payments owed for these leases.
We did not have any financing lease arrangements as of December 31, 2025 and 2024.
The table below presents certain information related to the weighted average discount rate and weighted average remaining lease term for the Company’s leases as of December 31, 2025 and 2024:
| | | | | | | | | | | |
| 2025 | | 2024 |
| Weighted average discount rate - operating leases | 6.4 | % | | 6.3 | % |
| Weighted average remaining lease term in years - operating leases | 5.2 | | 6.2 |
The components of lease expense for the years ended December 31, 2025, 2024, and 2023 were as follows:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| (In thousands) | 2025 | | 2024 | | 2023 |
| Operating lease costs | $ | 2,323 | | | $ | 1,940 | | | $ | 1,572 | |
| Short-term lease costs | 106 | | | 51 | | | 250 | |
| Total operating lease costs | 2,429 | | | 1,991 | | | 1,822 | |
| | | | | |
| Variable lease costs | 1,099 | | | 1,103 | | | 797 | |
| Total lease expense | $ | 3,528 | | | $ | 3,094 | | | $ | 2,619 | |
Maturities of our lease liabilities as of December 31, 2025 are as follows:
| | | | | |
| (In thousands) | Operating Leases |
| 2026 | $ | 2,967 | |
| 2027 | 2,843 | |
| 2028 | 2,642 | |
| 2029 | 2,724 | |
| 2030 | 2,551 | |
| Thereafter | 1,391 | |
| Total lease payments | 15,118 | |
| Less: interest | (2,218) | |
| Total present value of lease liabilities | $ | 12,900 | |
8. Property and equipment
Property and equipment consist of the following as of December 31, 2025 and 2024:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Property and equipment | | | |
| Leasehold improvements | $ | 3,507 | | | $ | 3,527 | |
| Furniture and computer equipment | 265 | | | 267 | |
| Manufacturing and other equipment | 4,607 | | | 3,919 | |
| Construction in-progress | 11,268 | | | 2,419 | |
| Subtotal | 19,647 | | | 10,132 | |
| Less: Accumulated depreciation | (4,934) | | | (4,158) | |
| Property and equipment, net | $ | 14,713 | | | $ | 5,974 | |
Depreciation expense for property and equipment was $0.8 million, $0.6 million, and $0.7 million for the years ended December 31, 2025, 2024, and 2023, respectively.
9. Accrued expenses and other current liabilities
Accrued expenses and other current liabilities consist of the following as of December 31, 2025 and 2024:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Accrued expenses | $ | 5,846 | | | $ | 7,192 | |
| | | |
| Accrued compensation | 5,933 | | | 4,828 | |
| Deferred revenue, current | 195 | | | 103 | |
| Total accrued expenses and other current liabilities | $ | 11,974 | | | $ | 12,123 | |
10. Goodwill and intangible assets
Goodwill
| | | | | |
| (In thousands) | Goodwill |
Goodwill as of December 31, 2024(1) | $ | 212,304 | |
| Less: Goodwill allocated to SAVSU in divestiture | (3,717) | |
| Balance as of December 31, 2025 | $ | 208,587 | |
(1) As discussed in Note 3: Discontinued operations, the Company allocated a portion of its goodwill to SAVSU in accordance with ASC 350. The goodwill balance presented above as of December 31, 2024 represents the balance of the reporting unit before the effect of the divestiture. The goodwill presented in the Consolidated Balance Sheets as of December 31, 2025 and 2024 is on a continuing operations basis giving effect to the divestitures.
Intangible assets
Intangible assets, net consisted of the following as of December 31, 2025 and 2024:
| | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands, except weighted average useful life) | December 31, 2025 | | |
| Intangible assets: | Gross Carrying Value | | Accumulated Amortization | | Net Carrying Value | | Weighted Average Useful Life
(in years) |
| Tradenames | $ | 2,794 | | | $ | (1,268) | | | $ | 1,526 | | | 5.9 |
| Technology - acquired | 7,822 | | | (5,091) | | | 2,731 | | | 2.9 |
| Total intangible assets | $ | 10,616 | | | $ | (6,359) | | | $ | 4,257 | | | 3.7 |
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2024 | | |
| Intangible assets: | Gross Carrying Value | | Accumulated Amortization | | Net Carrying Value | | Weighted Average Useful Life
(in years) |
| Tradenames | $ | 2,794 | | | $ | (1,004) | | | $ | 1,790 | | | 6.9 |
| Technology - acquired | 7,922 | | | (4,147) | | | 3,775 | | | 3.9 |
| Total intangible assets | $ | 10,716 | | | $ | (5,151) | | | $ | 5,565 | | | 4.7 |
Amortization expense for finite-lived intangible assets was $1.3 million, $1.2 million, and $2.0 million for the years ended December 31, 2025, 2024, and 2023, respectively. As of December 31, 2025, the Company expects to record the following amortization expense:
| | | | | |
| (In thousands) | |
| For the Years Ending December 31, | Estimated
Amortization
Expense |
| 2026 | $ | 1,206 | |
| 2027 | 1,016 | |
| 2028 | 742 | |
| 2029 | 530 | |
| 2030 | 411 | |
| Thereafter | 352 | |
| Total | $ | 4,257 | |
11. Commitments and contingencies
Employment agreements
We have employment agreements with certain key employees. None of these employment agreements is for a definitive period, but rather each will continue indefinitely until terminated in accordance with its terms. The agreements provide for a base annual salary, payable in monthly (or shorter) installments. Under certain conditions and for certain of these officers, we may be required to pay additional amounts upon terminating the officer or upon the officer resigning for good reason.
Litigation
From time to time, the Company is subject to various legal proceedings that arise in the ordinary course of business, none of which are currently material to the Company’s business. The Company’s industry is characterized by frequent claims and litigation, including claims regarding intellectual property. As a result, the Company may be subject to various legal proceedings from time to time. The results of any future litigation cannot be predicted with certainty, and regardless of the outcome, litigation can have an adverse impact on the Company because of defense and settlement costs, diversion of management resources and other factors. Management is not aware of any pending or threatened litigation.
Indemnification
As permitted under Delaware law and in accordance with the Company’s bylaws, the Company is required to indemnify its officers and directors for certain errors and occurrences while the officer or director is or was serving in such capacity. The Company is also party to indemnification agreements with its directors. The Company believes the fair value of the indemnification rights and agreements is minimal. Accordingly, the Company has not recorded any liabilities for these indemnification rights and agreements as of December 31, 2025.
Purchase obligations
Purchase obligations are defined as agreements to purchase goods or services that are enforceable and legally binding and that specify all significant terms, including fixed or minimum quantities to be purchased, fixed, minimum, or variable pricing provisions and the approximate timing of the transactions. As of December 31, 2025, our total short-term obligations were $9.8 million.
Non-income related taxes
Companies are required to collect and remit sales tax from certain customers if the company is determined to have nexus in a particular state. Upon the determination of nexus, which varies by state, companies are additionally required to maintain detailed record of specific product and customer information within each jurisdiction in which it has established nexus to appropriately determine their sales tax liability, requiring technical knowledge of each jurisdiction’s tax case law. During the year ended December 31, 2022, the Company determined that a sales tax liability related to the periods of 2021 through
2022 is probable. The estimated liability was determined to be approximately $2.3 million and $4.2 million as of December 31, 2025 and December 31, 2024, respectively. Due to the variety of jurisdictions in which this estimated liability relates to and our ongoing assessment of sales taxes owed, we cannot predict when final liabilities will be satisfied. We will reevaluate the estimated liability and timing of satisfaction each reporting period.
Settlement of Global Cooling Escrow
On May 3, 2021, the Company acquired Global Cooling pursuant to an Agreement and Plan of Merger, dated as of March 19, 2021 (the “GCI Merger Agreement”). Pursuant to the GCI Merger Agreement, the aggregate consideration paid to former stockholders of Global Cooling (collectively, the “Global Cooling Stockholders”) was 6,646,870 newly issued shares of common stock (the “GCI Merger Consideration”) were provided with the requirement that the GCI Merger Consideration otherwise payable to Global Cooling Stockholders were subject to reduction for indemnification obligations. Approximately 9% of the GCI Merger Consideration (the "GCI Escrow Shares") otherwise issuable to the GCI Stockholders were deposited into a segregated escrow account (the “GCI Escrow Account”) in accordance with an escrow agreement entered into in connection with the closing of the transactions contemplated by the GCI Merger Agreement (the “GCI Escrow Agreement”). Of the GCI Escrow Shares, an amount equal to 5% of the GCI Merger Consideration were considered general escrow shares (the “General Escrow Shares”). The General Escrow Shares were eligible to be held in escrow for a period of up to 18 months after the closing of the Global Cooling acquisition as the sole and exclusive source of payment for any indemnification claims made by the Company.
On September 28, 2022, the Company asserted an indemnification claim pursuant to the GCI Merger Agreement. On June 5, 2023, the Company entered into a Settlement Agreement with the representatives of the Global Cooling Stockholders, pursuant to which the parties agreed to release 65% of the General Escrow Shares, totaling 216,024 shares, to the Company from the GCI Escrow Account. These shares were returned to the Company and subsequently cancelled. As a result of the settlement, the Company recorded a $5.1 million gain recognizing the return of the shares during the second quarter of 2023.
12. Long-term debt
Term Loan
On September 20, 2022, the Company and certain of its subsidiaries entered into the Loan and Security Agreement, dated September 20, 2022, by and among Silicon Valley Bank, a division of First-Citizens Bank & Trust Company (“Bank”), the Company, SAVSU, CBS, SciSafe Holdings, Inc., a Delaware corporation (“SciSafe Parent”), and Sexton Biotechnologies, Inc., a Delaware corporation (“Sexton,” and together with the Company, SAVSU, CBS and SciSafe Parent, “Borrower”), as amended by that certain Waiver and First Amendment to Loan and Security Agreement, dated February 26, 2024, that certain Consent and Second Amendment to Loan and Security Agreement, dated April 17, 2024 (the “Second Amendment”), and that certain Consent and Third Amendment to Loan and Security Agreement, dated November 11, 2024 (the “Third Amendment”) that certain Consent and Fourth Amendment to Loan and Security Agreement, dated April 4, 2025 (the “Fourth Amendment”), that certain Consent to Loan and Security Agreement dated July 29, 2025 (the “Fifth Amendment”) and that certain Consent to Loan and Security Agreement, dated October 6, 2025 (the “Sixth Amendment”, and the foregoing collectively, the “Loan Agreement”), which provides for a term loan in an aggregate maximum principal amount of up to $60 million in the increments and upon the dates and milestones described below (the “Term Loan”). The Term Loan matures on June 1, 2026. The Loan Agreement permitted the Company to borrow up to $30 million upon the initial closing of the transactions contemplated by the Loan Agreement (the “Term Loan Closing”), and provided options to borrow (i) up to $10 million between the Term Loan Closing and June 30, 2023, (ii) up to $10 million upon the achievement of certain revenue milestones by the Company, and (iii) an additional $10 million at the discretion of the lender. The Company borrowed $20 million at the Term Loan Closing and accounts for the Term Loan at cost. As of December 31, 2023, the Company had not drawn additional funding nor had it met the revenue milestones outlined within the Loan Agreement. The Company had until December 31, 2023 to draw an additional $10 million, subject to approval from the lender, and therefore has no additional opportunities under the Loan Agreement. Payments on the borrowing were interest-only through June 2024, with additional criteria allowing for interest-only payments to continue through June 2025, which the Company did not pursue. Tranches borrowed under the Loan Agreement bear interest at the Wall Street Journal prime rate plus 0.5%. However, the interest rate is subject to a ceiling that restricts the interest rate for each tranche from exceeding 1.0% above the overall rate applicable to each tranche at their respective funding dates and has a balloon payment due at the earliest of term loan maturity, repayment of the Term Loan in full, or termination of the Loan Agreement at $1.2 million. As of December 31, 2025, the implied interest rate of the Term Loan is 9.3% and the implied value of the Term Loan is $6.1 million. The Loan Agreement contains customary representations and warranties as well as
customary affirmative and negative covenants. As of December 31, 2025, the Company is in compliance with the covenants set forth in the Loan Agreement.
On April 17, 2024, the Company entered into the Second Amendment by and among Bank, the Company, SAVSU, CBS, SciSafe Parent, Global Cooling and Sexton (the Company, SAVSU, CBS, SciSafe Parent, Global Cooling and Sexton, collectively, the “Second Amendment Borrower”). Pursuant to the Second Amendment and subject to the conditions set forth therein, Bank consented to the Global Cooling Divestiture and released its security interests in the assets of Global Cooling and the shares of common stock of Global Cooling arising under the Loan Agreement. In addition, effective as of the closing of the Global Cooling Divestiture, the Second Amendment amended the Loan Agreement to remove Global Cooling as a party to the Loan Agreement and provide for a non-refundable termination fee in the amount of $500,000 payable by the Second Amendment Borrower to Bank in the event that the Loan Agreement is terminated prior to the Term Loan Maturity Date (as defined in the Loan Agreement) for any reason. The Second Amendment also contains customary representations and warranties of the Second Amendment Borrower and provides for a release of Bank by the Second Amendment Borrower for any claims existing or arising through the date of the Second Amendment, including, without limitation, those arising out of or in any manner connected with or related to the Loan Agreement.
On November 11, 2024, the Company entered into the Third Amendment by and among Bank, the Company, SAVSU, CBS, SciSafe Parent and Sexton (the Company, SAVSU, CBS, SciSafe Parent and Sexton, collectively, the “Third Amendment Borrower”). Pursuant to the Third Amendment and subject to the conditions set forth therein, Bank consented to the SciSafe Divestiture as required pursuant to the Loan Agreement. In addition, effective as of the closing of the SciSafe Divestiture, the Third Amendment amended the Loan Agreement to provide for a non-refundable termination fee in the amount of $750,000 payable by the Third Amendment Borrower to Bank in the event that the Loan Agreement is terminated prior to the Term Loan Maturity Date for any reason. The Third Amendment also made certain other ministerial changes to the Loan Agreement, contains customary representations and warranties of the Third Amendment Borrower and provides for a release of Bank by the Third Amendment Borrower for any claims existing or arising through the date of the Third Amendment, including, without limitation, those arising out of or in any manner connected with or related to the Loan Agreement.
On April 4, 2025, the Company entered into the Fourth Amendment, by and among Bank, the Company, SAVSU, and Sexton (the Company, SAVSU and Sexton, collectively, the “Fourth Amendment Borrower”). Pursuant to the Fourth Amendment and subject to the conditions set forth therein, Bank consented to the PanTHERA Transaction and the dissolution of SciSafe as required pursuant to the Loan Agreement. The Fourth Amendment also made certain other ministerial changes to the Loan Agreement, contains customary representations and warranties of the Fourth Amendment Borrower and provides for a release of Bank by the Fourth Amendment Borrower for any claims existing or arising through the date of the Fourth Amendment, including, without limitation, those arising out of or in any manner connected with or related to the Loan Agreement.
On July 29, 2025, the Company entered into the Fifth Amendment, by and among Bank, the Company, SAVSU, and Sexton (the Company, SAVSU and Sexton, collectively, the “Fifth Amendment Borrower”). Pursuant to the Fifth Amendment and subject to the conditions set forth therein, Bank consented to adding PanTHERA as a borrower to the Loan Agreement and granted Bank a security interest in the assets of PanTHERA. The Fifth Amendment also made certain other ministerial changes to the Loan Agreement, contains customary representations and warranties of the Fifth Amendment Borrower and provides for a release of Bank by the Fifth Amendment Borrower for any claims existing or arising through the date of the Fifth Amendment, including, without limitation, those arising out of or in any manner connected with or related to the Loan Agreement.
On October 6, 2025, the Company entered into Sixth Amendment by and among Bank and the Company, SAVSU, Sexton, and PanTHERA (collectively, the “Sixth Amendment Borrower”). Pursuant to the Sixth Amendment and subject to the conditions set forth therein, the Bank consented to the purchase of the convertible promissory note from the iPSC developer and the SAVSU Transaction as required by the Loan Agreement and released its security interests in the assets of SAVSU and the outstanding membership interests of SAVSU arising under the Loan Agreement. The Sixth Amendment contains customary representations and warranties of the Sixth Amendment Borrower and provides for a release of the Bank by the Sixth Amendment Borrower for any claims existing or arising through the date of the Sixth Amendment, including, without limitation, those arising out of or in any manner connected with or related to the Loan Agreement.
Long-term debt consisted of the following as of December 31, 2025 and 2024:
| | | | | | | | | | | | | | | | | | | | | | | |
| | | | | December 31, |
| (In thousands) | Maturity Date | | Interest Rate | | 2025 | | 2024 |
Term Loan(1) | Jun-26 | | 7.0 | % | | $ | 5,000 | | | $ | 15,000 | |
| Insurance premium financing | Various | | 8.3 | % | | — | | | 975 | |
| Total debt, excluding unamortized debt issuance costs | | | | | 5,000 | | | 15,975 | |
| Less: unamortized debt issuance costs | | | | | (3) | | | (35) | |
| Total debt | | | | | 4,997 | | | 15,940 | |
| Less: current portion of debt | | | | | (4,997) | | | (10,943) | |
| Total long-term debt | | | | | $ | — | | | $ | 4,997 | |
As of December 31, 2025, the scheduled maturities of loans payable for each of the next five years and thereafter were as follows:
| | | | | |
| (In thousands) | Amount |
| 2026 | $ | 4,997 | |
| |
| |
| |
| |
| |
| Total | $ | 4,997 | |
13. Stock-based compensation
Stock compensation plans
Our stock-based compensation programs are long-term retention programs that are intended to attract, retain, and provide incentives for talented employees, officers, and directors, and to align stockholder and employee interests. Compensation expense associated with equity-based awards is recognized on a straight-line basis over the requisite service period, with awards generally vesting over a 4 year period, and forfeitures recognized as incurred. We have the following stock-based compensation plans and programs:
During 2013, we adopted the 2013 Performance Incentive Plan (the “2013 Plan”), which allowed us to grant options or restricted stock awards to all employees, including executive officers, outside consultants and non-employee directors. An aggregate of 3.1 million shares of common stock was initially reserved for issuance under the 2013 Plan. In May 2017, July 2020, June 2021, and June 2022, the shareholders approved an increase in the number of shares available for issuance to 4.1 million shares, 5.0 million shares, 6.5 million shares, and 8.5 million shares, respectively. As of April 25, 2023, the 2013 Plan expired as to future awards in accordance with its terms. As of December 31, 2025, there were outstanding options to purchase 117,000 shares of the Company’s common stock and approximately 70,000 unvested restricted stock awards outstanding under the 2013 Plan.
On July 21, 2023, our stockholders approved the 2023 Omnibus Performance Incentive Plan (the "2023 Plan"). The 2023 Plan allows us to grant equity awards to employees, directors, and outside consultants. An aggregate of 4.2 million shares of common stock were initially reserved for issuance under the 2023 Plan, plus any shares subject to awards under the 2013 Plan that were outstanding as of July 21, 2023, and which are subsequently forfeited or lapsed and not issued under the 2013 Plan. As of December 31, 2025, there were approximately 1.7 million unvested restricted stock awards outstanding under the 2023 Plan.
Issuance of shares
When options are exercised, it is the Company’s policy to issue new shares.
Stock option activity
Service vesting-based stock options
The following is a summary of service vesting-based stock option activity for the year ended December 31, 2025 and 2024, and the status of service vesting-based stock options outstanding as of December 31, 2025 and 2024:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2025 | | 2024 | | 2023 |
| Shares | | Wtd. Avg.
Exercise Price | | Shares | | Wtd. Avg.
Exercise Price | | Shares | | Wtd. Avg.
Exercise Price |
| Outstanding as of beginning of year | 127,000 | | $ | 2.19 | | | 217,250 | | $ | 2.21 | | | 456,293 | | $ | 2.17 | |
| Exercised | (10,000) | | 1.83 | | | (90,250) | | 2.23 | | | (239,043) | | 2.12 | |
| | | | | | | | | | | |
| Outstanding at end of year | 117,000 | | $ | 2.22 | | | 127,000 | | $ | 2.19 | | | 217,250 | | $ | 2.21 | |
| | | | | | | | | | | |
| Stock options exercisable at year end | 117,000 | | $ | 2.22 | | | 127,000 | | $ | 2.19 | | | 217,250 | | $ | 2.21 | |
We did not recognize stock compensation expense related to service-based options during the years ended December 31, 2025, 2024, and 2023. As of December 31, 2025, there was $2.6 million of aggregate intrinsic value of outstanding and exercisable service-based vesting stock options. Intrinsic value is the total pretax intrinsic value for all “in-the-money” options (i.e., the difference between the Company’s closing stock price on the last trading day of the year and the exercise price, multiplied by the number of shares) that would have been received by the option holders had all option holders exercised their options on December 31, 2025. This amount will change based on the fair market value of the Company’s stock. Intrinsic value of service vesting-based awards exercised during the years ended December 31, 2025, 2024, and 2023 was $0.2 million, $1.8 million, and $3.9 million, respectively. There were no service based-vesting options granted during the years ended December 31, 2025, 2024, and 2023. The weighted average remaining contractual life of service vesting-based options outstanding and exercisable as of December 31, 2025 is 0.4 years. There were no unrecognized compensation costs for service vesting-based stock options as of December 31, 2025.
The following table summarizes information about service vesting-based stock options exercisable and outstanding as of December 31, 2025:
| | | | | | | | | | | | | | | | | | | | |
| Range of Exercise Prices | | Number Outstanding at
December 31, 2025 | | Weighted Average
Remaining Contractual Life (in years) | | Weighted Average Exercise
Price |
$1.00 - 1.50 | | 2,000 | | 0.85 | | $ | 1.49 | |
| $1.51 - 2.00 | | 105,000 | | 0.30 | | 1.90 | |
| $2.51 - 8.60 | | 10,000 | | 1.92 | | 5.69 | |
| | 117,000 | | 0.44 | | $ | 2.22 | |
Restricted stock
Service vesting-based restricted stock
The following is a summary of service vesting-based restricted stock activity for the years ended December 31, 2025 and 2024, and the status of unvested service vesting-based restricted stock outstanding as of December 31, 2025 and 2024:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2025 | | 2024 | | 2023 |
| Shares | | Wtd. Avg. Grant
Date Fair Value | | Shares | | Wtd. Avg. Grant
Date Fair Value | | Shares | | Wtd. Avg. Grant
Date Fair Value |
| Outstanding as of beginning of year | 1,295,640 | | $ | 16.00 | | | 2,312,898 | | $ | 18.32 | | | 1,879,215 | | $ | 28.94 | |
| Granted | 470,824 | | | 25.64 | | | 484,886 | | | 20.65 | | | 1,907,101 | | | 13.12 | |
| Vested | (599,358) | | | 18.58 | | | (1,323,729) | | | 21.57 | | | (1,237,221) | | | 24.97 | |
| Forfeited | (61,532) | | 17.35 | | | (178,415) | | 17.09 | | | (236,197) | | 25.88 | |
| Non-vested at year end | 1,105,574 | | $ | 18.62 | | | 1,295,640 | | $ | 16.00 | | | 2,312,898 | | $ | 18.32 | |
The aggregate fair value of the service vesting-based awards granted during the years ended December 31, 2025, 2024, and 2023 was $11.4 million, $10.0 million, and $25.0 million, respectively. The aggregate fair value of the service vesting-based awards that vested during the years ended December 31, 2025, 2024, and 2023 was $11.2 million, $27.7 million, and $20.5 million, respectively.
We recognized stock compensation expense of $10.6 million, $9.6 million, and $12.8 million related to service vesting-based awards during the years ended December 31, 2025, 2024, and 2023, respectively. As of December 31, 2025, there was $16.8 million in unrecognized compensation costs related to service vesting-based awards. We expect to recognize those costs over 2.6 years.
Performance-based restricted stock
On March 8, 2024, the Company granted a performance-based restricted stock award ("PSA") for 109,512 shares to an executive. The shares granted contain performance conditions based on Company metrics related to future performance. The performance-based award was structured to vest between 0% and 200% of the number of restricted shares granted to the recipient based on the achievement of certain financial metrics related to future performance. The grant date fair value of this award was $17.36 per share. The fair value of this award is being expensed on a straight-line basis over the requisite service period ending on December 31, 2025. The PSA has been modified since its grant date as follows:
| | | | | |
| (in millions) | Fair Value |
| Fair value of PSA as of grant date | $ | 1.9 | |
| Q4 2024 change in estimated achievement probability | 0.8 | |
| Q1 2025 modification | 1.6 | |
| Cumulative stock compensation expense related to PSA as of December 31, 2025 | (4.3) | |
| Remaining unrecognized non-cash compensation costs related to PSA | $ | — | |
During the fourth quarter of the year ended December 31, 2024, it was determined the probability of attainment of the performance condition increased to greater than 100% of shares granted. In accordance with ASC Topic 718, Compensation - Stock Compensation ("ASC 718") we recognized a cumulative catch up in stock compensation expense to reflect the increased probability the PSA would vest in excess of the shares originally granted. The fair value of this award is being expensed on a straight-line basis in accordance with the estimated quantity of shares expected to vest over the requisite service period ending on December 31, 2025.
During the first quarter of the year ended December 31, 2025, the Board approved a modification to the metrics underlying the PSA to give effect to the adjusted operating results of the Company following the divestitures that occurred during the
year ended December 31, 2024. The modification increased the probability that the performance-based award would vest in excess of the shares originally granted to its maximum amount of 200%. In accordance with ASC 718, this modification resulted in an incremental compensation cost of $1.6 million, which will be recognized over the remainder of the service period of the award.
We recognized stock compensation expense related to the PSA of $3.1 million and $1.2 million for the years ended December 31, 2025 and 2024, respectively. There were no unrecognized compensation costs for the PSA as of December 31, 2025.
There were no performance-based restricted stock awards granted or vested during the year ended December 31, 2025. The aggregate fair value of the service vesting-based awards granted during the year ended December 31, 2024 was $1.9 million. No performance-based restricted stock awards vested during the year ended December 31, 2024.
No performance-based restricted stock awards were granted or vested during the year ended December 31, 2023.
Market-based restricted stock
The following is a summary of market-based restricted stock activity under our 2023 Plan for the years ended December 31, 2025 and 2024 and the status of market-based restricted stock outstanding as of December 31, 2025 and 2024:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2025 | | 2024 | | 2023 |
| Shares | | Wtd. Avg. Grant Date Fair
Value | | Shares | | Wtd. Avg. Grant Date Fair
Value | | Shares | | Wtd. Avg. Grant Date Fair
Value |
| Outstanding as of beginning of year | 495,686 | | $ | 25.69 | | | 509,166 | | $ | 26.50 | | | 271,044 | | $ | 30.64 | |
| Granted | 451,801 | | 32.91 | | | 312,081 | | 26.61 | | | 268,738 | | 25.19 | |
| Vested | (470,287) | | 25.19 | | | (325,561) | | 27.84 | | | (30,616) | | 51.65 | |
| | | | | | | | | | | |
| Non-vested at year end | 477,200 | | $ | 33.02 | | | 495,686 | | $ | 25.69 | | | 509,166 | | $ | 26.50 | |
The following is a summary of key inputs to our market-based restricted stock awards as of December 31, 2025:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | Fair Value Assumptions | | | | |
| Grant Date | | Target Shares | | Vesting Range | | Market Condition Period | | FV of Award
(in millions) | | Volatility | | Risk Free Rate | | Dividend Rate | | Attainment % | | Vested Shares |
| 2022 TSR | 2/24/2022 | | 240,428 | | 0% - 200% | | 1/1/2022 - 12/31/2023 | | $ | 6.7 | | | 63 | % | | 1.5 | % | | — | % | | 125 | % | | 300,529 |
2023 TSR(1) | 1/3/2023 | | 268,738 | | 0% - 200% | | 1/1/2023 - 12/31/2024 | | $ | 6.8 | | | 78 | % | | 4.4 | % | | — | % | | 175 | % | | 470,287 |
2024 TSR(2) | 3/8/2024 | | 239,464 | | 0% - 200% | | 1/1/2024 - 12/31/2025 | | $ | 6.3 | | | 80 | % | | 4.6 | % | | — | % | | N/A | | N/A |
| 2025 TSR | 3/18/2025 | | 250,252 | | 0% - 200% | | 1/1/2025 - 12/31/2026 | | $ | 9.8 | | | 60 | % | | 4.1 | % | | — | % | | N/A | | N/A |
(1) Of the $6.8 million fair value of the 2023 Total Shareholder Return ("TSR") award being expensed on a straight-line basis over the grant date to the vesting date, $1.6 million of expense was recognized in 2023 to reflect accelerations in the vesting period of certain awards.
(2) Of the $6.3 million fair value of the 2024 TSR award being expensed on a straight-line basis over the grant date to the vesting date, $0.3 million of expense was recognized in 2024 to reflect accelerations in the vesting period of certain awards.
Each of the market-based restricted stock awards outlined above were granted to the Company's executives. These restricted stock awards contain a market condition based on TSR. The market-based restricted stock awards vest at a range determined by the Compensation Committee of the Board in comparison of the Company's TSR to the TSR of a group of the Company's peers. The fair value of these awards is determined using a Monte Carlo simulation with various assumptions. These assumptions include historical volatility, dividend yield, and a risk-free interest rate. The historical
volatility is based on the most recent 2-year period for the Company and correlated with the components of the peer group. The stock price projection for the Company and the components of the peer group assumes a 0% dividend yield. This is mathematically equivalent to reinvesting dividends in the issuing entity over the performance period. The risk-free interest rate is based on the yield on the U.S. Treasury Strips as of the measurement date with a maturity consistent with the 2-year term associated with the market condition of these awards. The fair value of these awards is expensed on a straight-line basis over the grant date to the vesting date.
When the TSR period for each award has elapsed, the Company determines the TSR attainment percentage to award each recipient based on the targeted amount of shares granted.
We recognized stock compensation expense of $7.6 million, $4.5 million, and $7.1 million related to market-based restricted stock awards for the years ended December 31, 2025, 2024, and 2023. As of December 31, 2025, there was $5.5 million in unrecognized non-cash compensation costs related to market-based restricted stock awards expected to vest. We expect to recognize those costs over 1 year.
The aggregate fair value of the market-based awards granted during the years ended December 31, 2025, 2024, and 2023 was $6.3 million, $6.3 million, and $6.5 million, respectively. The aggregate fair value of the market-based awards that vested during the years ended December 31, 2025, 2024, and 2023 was $11.8 million, $5.7 million, and $0.7 million, respectively.
Total stock compensation expense
We recorded total stock compensation expense for the years ended December 31, 2025, 2024, and 2023, as follows:
| | | | | | | | | | | | | | | | | |
| 2025 | | 2024 | | 2023 |
| Research and development costs | $ | 2,499 | | | $ | 1,558 | | | $ | 2,076 | |
| Sales and marketing costs | 1,792 | | | 1,620 | | | 3,158 | |
| General and administrative costs | 15,875 | | | 10,814 | | | 13,213 | |
| Cost of revenue | 1,105 | | | 1,291 | | | 1,456 | |
| Total | $ | 21,271 | | | $ | 15,283 | | | $ | 19,903 | |
(1) During the years ended December 31, 2025 and 2024, $21 thousand and $18 thousand, respectively, of stock compensation expenses were capitalized to internal use software. No stock compensation expenses were capitalized into internal use software during the year ended December 31, 2023.
14. Income taxes
The Company prospectively adopted ASU 2023-09 as of January 1, 2025.
The Company's loss from continuing operations before income tax expense (benefit) included a loss of $0.1 million from foreign sources as of December 31, 2025. The Company's loss from continuing operations before income tax expense (benefit) did not contain any foreign components as of December 31, 2024 and 2023.
The following table presents U.S. and foreign components of loss from continuing operations before income tax expense (benefit):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| (In thousands) | 2025 | | 2024 | | 2023 |
| Domestic | $ | (11,954) | | | $ | (8,823) | | | $ | (12,221) | |
| Foreign | (127) | | | — | | | — | |
| Loss from continuing operations before income tax expense (benefit) | $ | (12,081) | | | $ | (8,823) | | | $ | (12,221) | |
Income tax expense (benefit) from continuing operations consists of the following:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| (In thousands) | 2025 | | 2024 | | 2023 |
| Current: | | | | | |
| | | | | |
| State | $ | 100 | | | $ | 26 | | | $ | 6 | |
| Foreign | (126) | | | — | | | — | |
| Total current tax provision | (26) | | | 26 | | | 6 | |
| | | | | |
| Deferred: | | | | | |
| Federal | 38 | | | (64) | | | (70) | |
| State | 37 | | | — | | | — | |
| | | | | |
| Total deferred tax provision | 75 | | | (64) | | | (70) | |
| | | | | |
| Income tax expense (benefit) | $ | 49 | | | $ | (38) | | | $ | (64) | |
The Company's loss from continuing operations before income tax expense (benefit) did not contain any foreign components as of December 31, 2024, and 2023.
The tax benefit for the years ended December 31, 2024 contained excess tax benefits from stock-based compensation of $0.4 million. The tax benefit for the year ended December 31, 2023 did not contain excess tax benefits from stock-based compensation.
The Company has paid the following in income taxes, net of refunds received:
| | | | | | | | | |
| Year Ended December 31, |
| (In thousands) | 2025 | | | | |
| Current: | | | | | |
| | | | | |
| State | $ | 64 | | | | | |
| Total income taxes paid | $ | 64 | | | | | |
The following table presents a reconciliation of the federal statutory rate and the Company's effective tax rate for the year ended December 31, 2025, in accordance with ASU 2023-09:
| | | | | | | | | | | |
| Year Ended December 31, |
| 2025 |
| (In thousands, except percentages) | Amount | | Percentage |
| At statutory rate | $ | (2,537) | | | 21.0 | % |
| State income taxes, net of federal effect | 85 | | | (0.7 | %) |
| Change in valuation allowance | (2,174) | | | 18.0 | % |
| Nontaxable or nondeductible items | | | |
| IPR&D expense | 3,259 | | | (26.9 | %) |
| Sec. 162(m) limitation on executive compensation | 3,378 | | | (27.9 | %) |
| Stock compensation | (1,789) | | | 14.8 | % |
| | | |
| | | |
| Other nondeductible items | 98 | | | (0.8 | %) |
| | | |
| Tax credits | | | |
| R&D credit | (210) | | | 1.6 | % |
| Worldwide changes in unrecognized tax benefit | 22 | | | (0.2 | %) |
| Other | (130) | | | 1.0 | % |
| Foreign tax effects | | | |
| Canada | 47 | | | (0.3 | %) |
| Income tax expense | $ | 49 | | | (0.4 | %) |
A reconciliation of income taxes computed using the U.S. federal statutory rate to that reflected in operations follows:
| | | | | | | | | | | | | |
| | | Year Ended December 31, |
| | | 2024 | | 2023 |
| Federal statutory tax | | | 21.0 | % | | 21.0 | % |
| State tax, net of federal benefit | | | 7.3 | % | | 6.4 | % |
| Stock compensation | | | 4.8 | % | | (8.3 | %) |
| Sec. 162(m) limitation on executive compensation | | | (5.2 | %) | | (10.7 | %) |
| Fair value change in contingent consideration | | | — | % | | 3.8 | % |
| Tax credits | | | 5.3 | % | | 6.6 | % |
| Change in valuation allowance | | | (28.3 | %) | | (24.2 | %) |
| Gain on escrow settlement | | | — | % | | 8.8 | % |
| Other | | | (4.5 | %) | | (2.4 | %) |
| Total | | | 0.4 | % | | 1.0 | % |
In 2025, state and local income taxes in Pennsylvania and Texas comprise the majority of the state and local income taxes, net of federal tax.
The principal components of the Company’s net deferred tax liabilities are as follows as of December 31, 2025 and 2024:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Deferred tax assets related to: | | | |
| Net operating loss carryforwards | $ | 37,443 | | | $ | 39,079 | |
| Capital loss carryforward | 3,164 | | | 7,011 | |
| Tax credit carryforward | 2,801 | | | 2,695 | |
| Stock-based compensation | 1,498 | | | 2,141 | |
| Accruals and reserves | 1,566 | | | 1,883 | |
| Inventory | 1,111 | | | 1,410 | |
| Fixed assets | (143) | | | 568 | |
| Lease liabilities | 3,219 | | | 3,540 | |
| Capitalized research and development | 2,540 | | | 3,222 | |
| Fair value change in investments | 314 | | | 556 | |
| Other | 28 | | | 1,084 | |
| Total deferred tax assets | 53,541 | | | 63,189 | |
| | | |
| Deferred tax liabilities related to: | | | |
| Intangibles | (1,108) | | | (2,326) | |
| Right-of-use assets | (1,863) | | | (2,654) | |
| | | |
| | | |
| Total deferred tax liabilities | (2,971) | | | (4,980) | |
| | | |
| Net deferred tax assets before valuation allowance | 50,570 | | | 58,209 | |
| Less: valuation allowance | 50,759 | | | 58,333 | |
| Net deferred tax liabilities | $ | (189) | | | $ | (124) | |
| | | |
| | | |
Realization of deferred tax assets is dependent upon the generation of future taxable income, if any, the timing and amount of which are uncertain. The assessment regarding whether a valuation allowance is required on deferred tax assets considers the evaluation of both positive and negative evidence when concluding whether it is more likely than not that deferred tax assets are realizable. The valuation allowance recorded as of December 31, 2025 and 2024 primarily relates to deferred tax assets for net operating loss carryforwards.
The changes in the valuation allowance for deferred tax assets were as follows:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Balance at beginning of period | $ | 58,333 | | | $ | 49,517 | |
| Net deferred tax assets divested | (2,990) | | | (7,723) | |
| Charged to income tax expense | (4,584) | | | 16,539 | |
| Balance at end of period | $ | 50,759 | | | $ | 58,333 | |
During the year ended December 31, 2025, the One Big Beautiful Bill Act ("OBBBA") was enacted, introducing significant changes to U.S. tax law. Among other items, the OBBBA allows the deduction of U.S. research and development expenses from taxable income. As previously required under the Tax Cuts and Jobs Act, the Company capitalized research and development expenditures in the years ended December 31, 2024 and 2023. With the enactment of OBBBA, the Company began deducting domestic Section 174 costs during the year ended December 31, 2025.
As of December 31, 2025, the Company had U.S. federal net operating loss (“NOL”) carryforwards of approximately $168.4 million. Approximately $38.6 million of NOL will expire from 2025 through 2037, and approximately $129.8 million of NOL will be carried forward indefinitely. The NOL carryforwards may become subject to an annual limitation in the event of certain cumulative changes in the ownership interest. This limited the amount of tax attributes that can be
utilized annually to offset future taxable income or tax liabilities. Subsequent ownership changes may further affect the limitation in future years.
The Company determines its uncertain tax positions based on a determination of whether and how much of a tax benefit taken by the Company in its tax filings or positions is more likely than not to be sustained upon examination by the relevant income tax authorities.
A reconciliation of the beginning and ending balances of uncertain tax positions in the years ended December 31, 2025 and 2024 is as follows:
| | | | | | | | | | | |
| (In thousands) | 2025 | | 2024 |
| Balance at beginning of period | $ | 1,155 | | | $ | 954 | |
| Increase related to prior year tax positions | (98) | | | — | |
| Increase related to current year tax positions | 120 | | | 201 | |
| Balance at end of period | $ | 1,177 | | | $ | 1,155 | |
The Company is generally subject to examination by U.S. federal and local income tax authorities for all tax years in which loss carryforward is available, which includes 2005 through 2025.
15. Segment, customer, and geographic information
The Company views its operations and makes decisions regarding how to allocate resources and manages its business as one reportable segment and one reporting unit. The Company’s Chief Executive Officer, Mr. Roderick de Greef, who is the CODM, reviews the Company’s operations on a consolidated basis for purposes of allocating resources and evaluating financial performance. As a single reportable segment entity, the Company’s segment performance measure is consolidated net loss from continuing operations.
Significant segment expenses are presented in the Company’s Consolidated Statements of Operations. Additional significant segment expenses that are not separately presented in the Company’s Consolidated Statements of Operations include Shared-based compensation and Depreciation expense. These are presented in the Consolidated Statement of Cash Flows, and Note 13: Stock-based compensation and Note 8: Property and equipment.
Other expense items not individually significant in net loss from continuing operations are changes in inventory values due to changes in its carrying basis, costs associated with the Company’s acquisitions and or divestitures in the period these take place, and gain or loss on disposal of fixed assets. The information provided to the Company’s CODM for purposes of making decisions and assessing segment performance excludes asset information.
Concentrations of risk
Significant customers are those that represent more than 10% of the Company’s total revenue or gross accounts receivable balances for the periods and as of each balance sheet date presented. For each significant customer, revenue as a percentage
of total revenue and gross accounts receivable as a percentage of total gross accounts receivable as of the periods presented were as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Accounts Receivable | | Revenue |
| December 31, | | Years Ended December 31, |
| 2025 | | 2024 | | 2025 | | 2024 | | 2023 |
| Customer A | 12 | % | | * | | * | | * | | * |
| Customer B | 22 | % | | * | | * | | * | | * |
| Customer C | * | | 23 | % | | * | | 15 | % | | 13 | % |
| Customer D | * | | 19 | % | | 15 | % | | 17 | % | | 16 | % |
| Customer E | * | | 16 | % | | 14 | % | | * | | * |
*less than 10%
The following is a summary of revenue by major product family representing over 10% of the Company's total revenue:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| Revenue by major product | 2025 | | 2024 | | 2023 |
| CryoStor | 82 | % | | 80 | % | | 82 | % |
The following table represents the Company’s total revenue by geographic area (based on the location of the customer):
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| Revenue by customers’ geographic locations | 2025 | | 2024 | | 2023 |
| United States | 80 | % | | 75 | % | | 81 | % |
| Europe, Middle East, Africa (EMEA) | 14 | % | | 19 | % | | 12 | % |
| Other | 6 | % | | 6 | % | | 7 | % |
| Total revenue | 100 | % | | 100 | % | | 100 | % |
All of the Company's long-lived assets, totaling $22.2 million, are located within the United States.
In the year ended December 31, 2025, two suppliers accounted for 12% and 11% of purchases, respectively. In the year ended December 31, 2023, one supplier accounted for 27% of purchases. In the year ended December 31, 2024, no suppliers accounted for more than 10% of purchases.
As of December 31, 2025, three suppliers accounted for 28%, 18%, and 17% of accounts payable, respectively. As of December 31, 2024, no suppliers accounted for more than 10% of our accounts payable.
16. Employee benefit plan
The Company sponsors 401(k) defined contribution plans for its employees. These plans provide for pre-tax and post-tax contributions for all employees. Employee contributions are voluntary. Employees may contribute up to 100% of their annual compensation to these plans, as limited by an annual maximum amount as determined by the Internal Revenue Service. The Company matches employee contributions in amounts to be determined at the Company’s sole discretion. The Company made contributions of $0.6 million, $0.5 million, and $0.4 million to the plans for the years ended December 31, 2025, 2024, and 2023.
17. Subsequent events
The Company has evaluated events subsequent to December 31, 2025 through the date of this filing to assess the need for potential recognition or disclosure. Based upon this evaluation, it was determined that no subsequent events occurred that require recognition or disclosure in the Consolidated Financial Statements.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
(a)Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer ("CEO") and Chief Financial Officer ("CFO"), evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2025. The term “disclosure controls and procedures” means controls and other procedures of a company that are designed to provide reasonable assurance that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the U.S. SEC rules and forms. Management recognizes that a control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Further, management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Based on the evaluation of our disclosure controls and procedures, our CEO and CFO have concluded that our disclosure controls and procedures were effective as of December 31, 2025.
(b)Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rule 13a-15(f) of the Exchange Act) and the preparation of financial statements for external purposes in accordance with United States Generally Accepted Accounting Policies (“U.S. GAAP”). Using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) in Internal Control—Integrated Framework (2013 framework), management of the Company under supervision and participation of the CEO and CFO, conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2025.
Based on our assessment under the framework in Internal Control—Integrated Framework (2013 framework), our management concluded that our internal control over financial reporting was effective as of December 31, 2025.
The Company’s independent registered public accounting firm, Grant Thornton, LLP, has audited the Company’s internal control over financial reporting as of December 31, 2025. Its report on the audit of internal control over financial reporting appears below.
(c)Remediation of Material Weaknesses in Internal Control over Financial Reporting
In fiscal year 2024, management identified a material weakness in our internal control over financial reporting as of December 31, 2024, regarding ineffective internal controls to verify that key inputs for the Company's stock-based awards were entered correctly into the equity system early in 2024. This weakness was attributed to an outdated internal policy with unclear guidance regarding the appropriate inputs. During the quarter ended March 31, 2025, Management made significant changes to its internal control over financial reporting to remediate the material weakness, as follows:
•Updated the Stock-Based Compensation Policy to include clearer guidance and considerations on how to identify the appropriate inputs.
•Trained accounting team members on the updated policy and execution.
(d)Changes in Internal Control Over Financial Reporting
For the year ended December 31, 2025:
•Management remediated the prior year material weaknesses related to ineffective internal controls to verify that key inputs for the Company's stock-based awards were entered correctly into the equity system as described in section (c).
•The Company implemented internal controls related to the acquisition of PanTHERA, the purchase of the convertible promissory note, and the divestiture of SAVSU Technologies, Inc.
Other than the changes noted above, there have been no other changes in our internal control over financial reporting during the fiscal quarter ended December 31, 2025 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
(e)Attestation Report of the Independent Registered Public Accounting Firm
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board of Directors and Shareholders
BioLife Solutions, Inc.
Opinion on internal control over financial reporting
We have audited the internal control over financial reporting of BioLife Solutions, Inc. (a Delaware corporation) and subsidiaries (the “Company”) as of December 31, 2025, based on criteria established in the 2013 Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”). In our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2025, based on criteria established in the 2013 Internal Control—Integrated Framework issued by COSO.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (“PCAOB”), the consolidated financial statements of the Company as of and for the year ended December 31, 2025, and our report dated February 26, 2026 expressed an unqualified opinion on those financial statements.
Basis for opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management’s Annual Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and limitations of internal control over financial reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and
directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ GRANT THORNTON LLP
Bellevue, Washington
February 26, 2026
ITEM 9B. OTHER INFORMATION
Rule 10b5-1 Trading Arrangements
During the quarter ended December 31, 2025, none of our officers or directors adopted or terminated any Rule 10b5-1 trading arrangements (as such term is defined in Item 408 of Regulation S-K).
Non-Rule 10b5-1 Trading Arrangements
During the quarter ended December 31, 2025, none of our officers or directors adopted or terminated any non-Rule 10b5-1 trading arrangement (as such term is defined in Item 408 of Regulation S-K).
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
None.
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Reference is made to the information regarding directors and nominees, code of ethics, corporate governance matters, and disclosure relating to compliance with Section 16(a) of the Securities Exchange Act of 1934 appearing under the captions “Election of Directors” and “Delinquent Section 16(a) Reports” in the Company’s Proxy Statement for its 2026 Annual Meeting of Stockholders (the “2026 Annual Meeting”), which information is incorporated in this Annual Report on Form 10-K by reference.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this item will be set forth under the caption “Executive Compensation” in the Company’s Proxy Statement for the 2026 Annual Meeting, and is incorporated herein by reference.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT RELATED STOCKHOLDER MATTERS
The information required by this item will be set forth under the captions “Security Ownership of Certain Beneficial Owners and Management Related Stockholder Matters” in the Company’s Proxy Statement for the 2026 Annual Meeting, and is incorporated herein by reference.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required by this item will be set forth under the captions “Certain Relationships and Related Transactions, and Director Independence” in the Company’s Proxy Statement for the 2026 Annual Meeting, and is incorporated herein by reference.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required by this item will be set forth under the caption Principal Accountant Fees And Services in the Company’s Proxy Statement for the 2026 Annual Meeting, and is incorporated herein by reference.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a)The following documents are filed as part of this Annual Report on Form 10-K:
(1)Financial Statements (Included Under Item 8): The Index to the Financial Statements is included in this Annual Report on Form 10-K and is incorporated herein by reference.
(2)Financial Statement Schedules: Schedules to the Financial Statements have been omitted because the information required to be set forth therein is not applicable or is shown in the accompanying Financial Statements or notes thereto.
(b)Exhibits
| | | | | | | | |
| Exhibit Number | | Document |
| 2.1†* | | Agreement and Plan of Merger, dated as of March 19, 2021, by and among the Company, BLFS Merger Subsidiary, Inc., Global Cooling, Inc. and Albert Vierling and William Baumel, in their capacity as the representatives of the stockholders of Global Cooling, Inc. (included as Exhibit 2.1 to the current report on Form 8-K filed by the Company with the SEC on March 25, 2021) |
| 2.2† | | Agreement and Plan of Merger, dated as of August 9, 2021, by and among the Company, BLFS Merger Sub, Inc., Sexton Biotechnologies, Inc. and Fortis Advisors LLC, in their capacity as the representatives of the stockholders of Sexton Biotechnologies, Inc. (incorporated by reference to Exhibit 2.6 to Company's report on Form 10-K filed by the Company with the SEC on March 31, 2022) |
| 2.3†*** | | |
| 2.4†*** | | |
| 2.5†*** | | |
| 3.1 | | |
| 3.2 | | |
| 3.3 | | |
| 3.4 | | |
| 4.1 | | |
| 10.1** | | |
| 10.2** | | |
| 10.3** | | |
| | | | | | | | |
| 10.4** | | |
| 10.5** | | |
| 10.6** | | |
| 10.7** | | |
| 10.8** | | |
| 10.9** | | |
| 10.10** | | |
| 10.11 | | |
| 10.12 | | |
| 10.13 | | |
| 10.14 | | |
| 10.15* | | |
| 10.16* | | |
| 10.17* | | Consent and Second Amendment to Loan and Security Agreement, dated April 17, 2024, by and among Silicon Valley Bank, BioLife Solutions, Inc., SAVSU Technologies, Inc., Arctic Solutions, Inc., SciSafe Holdings, Inc., Global Cooling, Inc., and Sexton Biotechnologies, Inc. (incorporated by reference to Exhibit 10.1 to the Company’s report on Form 8-K filed by the Company with the SEC on April 23, 2024) |
| 10.18† | | Consent and Third Amendment to Loan and Security Agreement, dated November 11, 2024, by and among Silicon Valley Bank, BioLife Solutions, Inc., SAVSU Technologies, Inc., Arctic Solutions, Inc., SciSafe Holdings, Inc., and Sexton Biotechnologies, Inc. (incorporated by reference to Exhibit 10.1 to the Company’s report on Form 8-K filed by the Company with the SEC on November 12, 2024) |
| 10.19†* | | |
| 10.20* | | |
| 10.21†* | | |
| 10.22** | | |
| 10.23** | | |
| | | | | | | | |
| 10.24** | | |
| 10.25** | | |
| 10.26** | | |
| 10.27** | | |
| 10.28** | | |
| 10.29** | | |
| 19.1 | | |
| 21.1 | | |
| 23.1 | | |
| 31.1 | | |
| 31.2 | | |
| 32.1 | | |
| 32.2 | | |
| 97.1 | | |
| 101.INS | | Inline XBRL Instance Document (filed herewith) |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema (filed herewith) |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase (filed herewith) |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase (filed herewith) |
| 101.LAB | | Inline XBRL Taxonomy Extension Label Linkbase (filed herewith) |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase (filed herewith) |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
| | | | | |
| * | Certain portions of this exhibit have been redacted pursuant to Item 601(b)(10) or Item 601(b)(2) of Regulation S-K. An unredacted copy of this exhibit will be furnished supplementally to the Securities and Exchange Commission upon request. |
| ** | Management contract or compensatory plan or arrangement. |
| † | Certain exhibits and schedules to this exhibit have been omitted pursuant to Item 601(a)(5) of Regulation S-K. The Registrant agrees to provide a copy of any omitted exhibit or schedule to the Securities and Exchange Commission or its staff upon request. |
(c)Excluded financial statements:
None.
ITEM 16. FORM 10-K SUMMARY
The Company has elected not to include a summary pursuant to this Item 16.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | |
| Date: | February 26, 2026 | BIOLIFE SOLUTIONS, INC. |
| | |
| | /s/ RODERICK DE GREEF |
| | Roderick de Greef |
| | Chief Executive Officer (principal executive officer) and Chairman of the Board of Directors |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | | | | | |
| Date: | February 26, 2026 | /s/ RODERICK DE GREEF |
| | Roderick De Greef |
| | Chief Executive Officer (principal executive officer) and Chairman of the Board of Directors |
| | |
| Date: | February 26, 2026 | /s/ TROY WICHTERMAN |
| | Troy Wichterman |
| | Chief Financial Officer (principal financial officer and principal accounting officer) |
| | |
| Date: | February 26, 2026 | /s/ CATHERINE COSTE |
| | Catherine Coste |
| | Director |
| | |
| Date: | February 26, 2026 | /s/ AMY DUROSS |
| | Amy DuRoss |
| | Director |
| | |
| Date: | February 26, 2026 | /s/ RACHEL ELLINGSON |
| | Rachel Ellingson |
| | Director |
| | |
| Date: | February 26, 2026 | /s/ JOYDEEP GOSWAMI |
| | Joydeep Goswami |
| | Director |
| | |
| Date: | February 26, 2026 | /s/ TONY HUNT |
| | Tony Hunt |
| | Director |
| | |
| Date: | February 26, 2026 | /s/ TIM MOORE |
| | Tim Moore |
| | Director |